30) Substance A to E below are listed with several of their properties. The identities of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
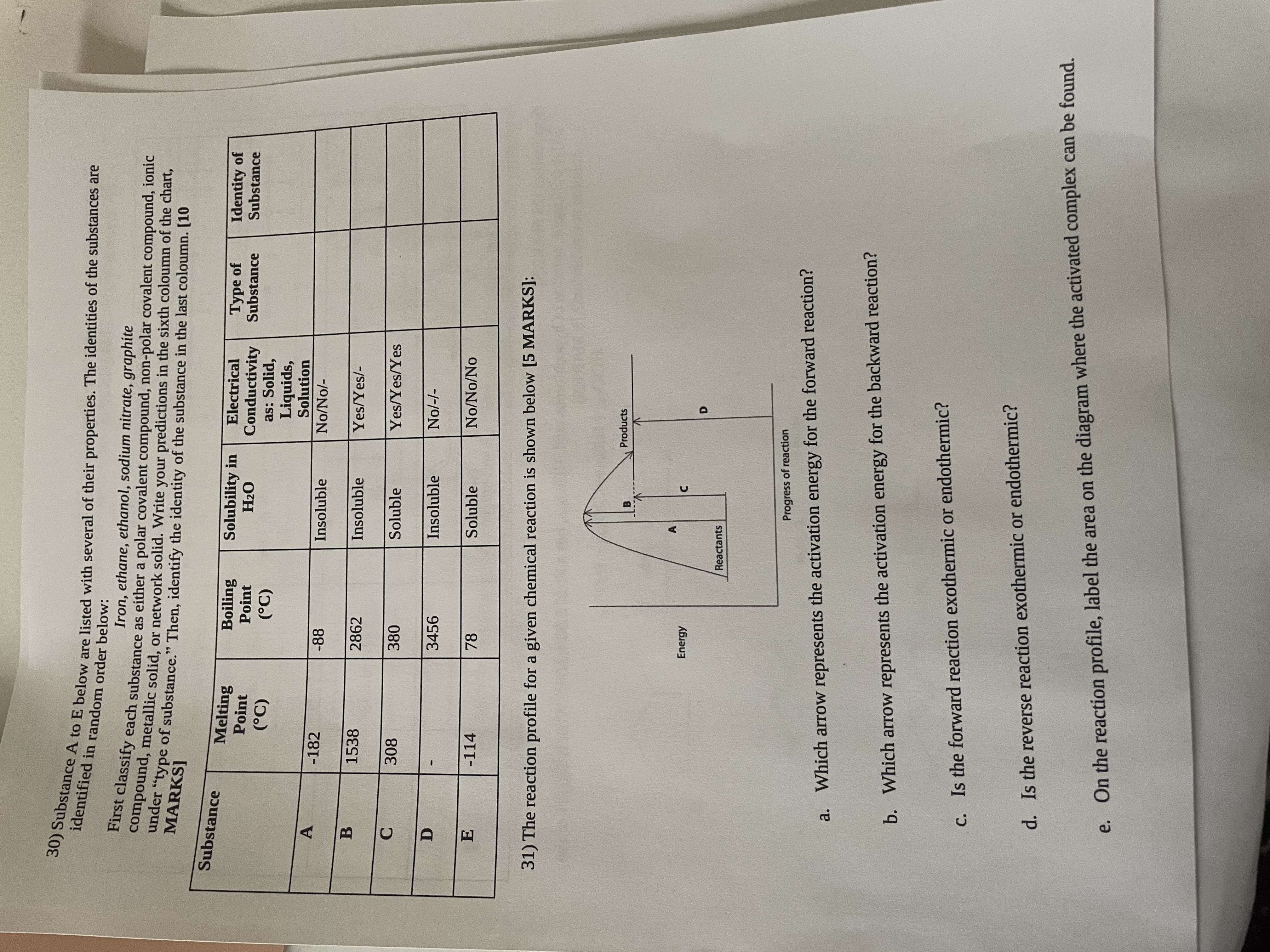
30) Substance A to E below are listed with several of their properties. The identities of the substances are identified in random order below: Iron, ethane, ethanol, sodium nitrate, graphite compound, metallic solid, or network solid. Write your predictions in the sixth coloumn of the chart, First classify each substance as either a polar covalent compound, non-polar covalent compound, ionic under "type of substance." Then, identify the identity of the substance in the last coloumn. [10 MARKS] Substance A B C D E Melting Point (C) -182 1538 308 -114 Boiling Solubility in Point HO (C) -88 2862 380 3456 78 Insoluble Energy Insoluble Soluble Insoluble Soluble Reactants Electrical Type of Conductivity Substance as: Solid, Liquids, Solution B No/No/- Yes/Yes/- Yes/Yes/Yes 31) The reaction profile for a given chemical reaction is shown below [5 MARKS]: No/-/- No/No/No Products D Progress of reaction a. Which arrow represents the activation energy for the forward reaction? b. Which arrow represents the activation energy for the backward reaction? c. Is the forward reaction exothermic or endothermic? d. Is the reverse reaction exothermic or endothermic? Identity of Substance e. On the reaction profile, label the area on the diagram where the activated complex can be found. 30) Substance A to E below are listed with several of their properties. The identities of the substances are identified in random order below: Iron, ethane, ethanol, sodium nitrate, graphite compound, metallic solid, or network solid. Write your predictions in the sixth coloumn of the chart, First classify each substance as either a polar covalent compound, non-polar covalent compound, ionic under "type of substance." Then, identify the identity of the substance in the last coloumn. [10 MARKS] Substance A B C D E Melting Point (C) -182 1538 308 -114 Boiling Solubility in Point HO (C) -88 2862 380 3456 78 Insoluble Energy Insoluble Soluble Insoluble Soluble Reactants Electrical Type of Conductivity Substance as: Solid, Liquids, Solution B No/No/- Yes/Yes/- Yes/Yes/Yes 31) The reaction profile for a given chemical reaction is shown below [5 MARKS]: No/-/- No/No/No Products D Progress of reaction a. Which arrow represents the activation energy for the forward reaction? b. Which arrow represents the activation energy for the backward reaction? c. Is the forward reaction exothermic or endothermic? d. Is the reverse reaction exothermic or endothermic? Identity of Substance e. On the reaction profile, label the area on the diagram where the activated complex can be found.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these algorithms questions
-
As discussed in the chapter, the principles underlying activity-based costing have evolved into the broader approach known as activity-based management. One of the common practices of activity-based...
-
The following investments occurred in 2013 for Mole Company. a. Mole purchased 5% of the common stock on Brandon Company, a business which has a history of paying large quarterly dividends. Mole...
-
Josie Inc. collects cash from customers two ways: 1. Accrued Revenue. Some customers pay Josie after Josie has performed service for the customer. During 2017, Josie made sales of $50,000 on account...
-
It is suggested that terrorism is directed towards both victims and an audience. How do these means, in the view of terrorists, impact the ability of terrorists to reach their goals and objectives?
-
In Figure an aluminum wire, of length L1 = 60.0cm, cross-sectional area 1.00 x 10-2 cm2, and density 2.60 g/cm3, is joined to a steel wire, of density 7.80 g/cm3 and the same cross-sectional area....
-
An ideal gas is heated from \(25^{\circ} \mathrm{C}\) to \(145^{\circ} \mathrm{C}\). The mass of the gas is \(2 \mathrm{~kg}\). Determine: (i) specific heats, (ii) change in internal energy, (iii)...
-
A sum of money invested at 2% per 6-month period (semiannually), will double in amount in approximately how many years?
-
Briefly describe any FOUR advantages of using comments in a program.
-
Write a paper on Strategic Management and Strategic Competitiveness in which described the followings: Give your opinion on the corporation's greatest strengths and most significant weaknesses....
-
Analyze the effectiveness of the leadership model that Anheuser Busch is currently using. Consider the current leadership styles in place at the organization, the effectiveness of the leadership...
-
An array of 8 solar thermal panels produces about 24,000 Btu of heat energy per hour. How much heat energy would a similar array of 6 panels produce per hour?
-
KORBIN COMPANY Comparative Balance Sheets December 31 Assets Current assets Long-term investments Plant assets, net Total assets Liabilities and Equity Current liabilities Common stock Other paid-in...
-
4. As the chapter states, GDP does not include the value of used goods that are resold. Why would including such transactions make GDP a less informative measure of economic well-being? 5. Below are...
-
How could behaviors influence leadership effectiveness and organizational cultures? How could each toxic leadership behavior have leadership consequences? Three recommended actions to prevent...
-
On July 9, 2010 Alfredo Matanglawin procured a life insurance designating his wife and children as irrevocable beneficiaries. Three years later Alfredo Matanglawin filed a petition in court to amend...
-
Classify each of the following activities as proper or prohibited under the various consumer statutes you have studied. a. Calling a hospital room to talk to a debtor who is a patient there. b....
-
Ethanol, C 2 H 5 OH, reacts with sodium metal because the hydrogen atom attached to the oxygen atom is slightly acidic. Write a balanced equation for the reaction of sodium with ethanol to give the...
-
Curium was first synthesized by bombarding an element with alpha particles. The products were curium-242 and a neutron. What was the target element?
-
In the presence of excess thiocyanate ion, SCN, the following reaction is first order in chromium(III) ion, Cr3+; the rate constant is 2.0 106/s. Cr3+(aq) + SCN(aq) Cr(SCN)2+(aq) If 85.0% reaction...
-
Hannah Gilpin is the controller of Blakemore Auto Glass, a division of Eastern Glass and Window. Her division has been under pressure to improve its divisional operating income. Currently, divisions...
-
As the new controller, reply to the following comment made by your plant manager: When I employ a proper accounting software, which can process all my daily accounting records and provide me with all...
-
What guidelines do management accountants use?

Study smarter with the SolutionInn App


