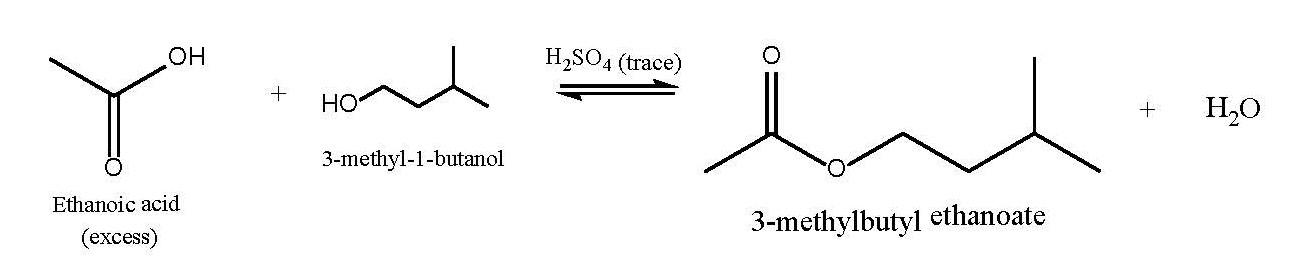
3-methyl-1-butanol (also called isoamyl alcohol or isopentylalcohol) was mixed with an excess of acetic acid (ethanoic acid
Question:
3-methyl-1-butanol (also called isoamyl alcohol or isopentylalcohol) was mixed with an excess of acetic acid (ethanoic acid isits systematic name) and a trace of sulfuric acid (which serves asa catalyst). This reaction is an equilibrium reaction, so it isexpected that not all the starting materials will be consumed. Theequilibrium should lie quite far to the right due to the excess ofacetic acid used, but not completely.
After an appropriate length of time, isolation of the desiredproduct from the reaction mixture was begun by adding a volume of5% aqueous sodium bicarbonate (NaHCO3 has aneffective pKa of 7) roughly equal to the volume of the reactionmixture. Bubbling occurred and a mixture consisting of two layersresulted—a basic aqueous layer and an organic layer.
The layers were separated, and the aqueous layer wasremoved.
The addition of aqueous sodium bicarbonate to the layer oforganic materials and separation of the layers was repeated twice.Each time the predominantly aqueous layers were removed, they werecombined in the same collection flask.
The organic layer that remained after the three bicarbonateextractions were dried and then subjected to distillation to obtaina pure sample of 3-methylbutyl ethanoate (isoamyl acetate).
- List all the chemical species likely to be present at the endof the reaction but before adding aqueous NaHCO3. Notethat the H2SO4 was not consumed (sinceit is a catalyst).