4 1 point Which Lewis structure would the phosphide ion have in calcium phosphide? X O...
Fantastic news! We've Found the answer you've been seeking!
Question:



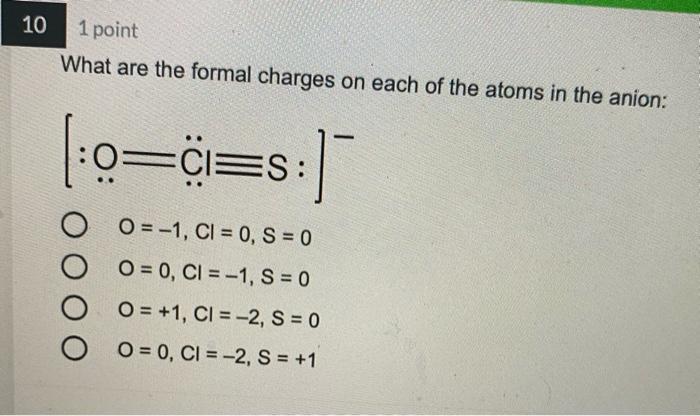
Transcribed Image Text:
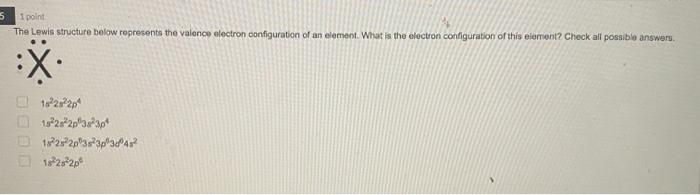
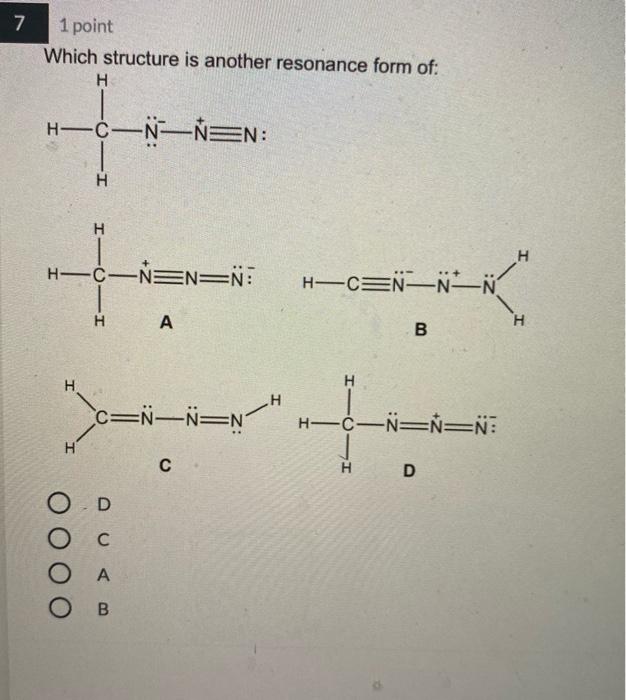
4 1 point Which Lewis structure would the phosphide ion have in calcium phosphide? ·X O |:X: ° [:X:]ª ° [³X³]¯ 3- 5 1 point The Lewis structure below represents the valence electron configuration of an element. What is the electron configuration of this element? Check all possible answers. :X. 18²28²2p² 1822s 2p 3s 3p 1s 2s 2p 3s 3p 3d 4² 18²2s²2p 7 1 point Which structure is another resonance form of: H H——NN=N: H H-C-NEN=N: H A H H -H N=N―N=0 ター O D C Ο Α OB C H-CNN N B H H—C—N=N=N: D H H 8 1 point Which is a reasonable Lewis structure for the OF* ion? |:0—F:)* | Q=F ]* |:0—F:]* [:0—F:]* A В с D 0 000 ACDB А O c О в 9 1 point In the Lewis structure of the nitrite anion, NO₂, if all atoms obey the octet rule, there must be: O2 N=0 bonds 2 N-O bonds 1 N-O bond and 1 NEO bond 1 N-O bond and 1 N=O bond 10 1 point What are the formal charges on each of the atoms in the anion: |:0=c=s: s:] O O=-1, CI = 0, S = 0 O=0, CI = -1, S = 0 O=+1, CI = -2, S = 0 O=0, CI = -2, S = +1 4 1 point Which Lewis structure would the phosphide ion have in calcium phosphide? ·X O |:X: ° [:X:]ª ° [³X³]¯ 3- 5 1 point The Lewis structure below represents the valence electron configuration of an element. What is the electron configuration of this element? Check all possible answers. :X. 18²28²2p² 1822s 2p 3s 3p 1s 2s 2p 3s 3p 3d 4² 18²2s²2p 7 1 point Which structure is another resonance form of: H H——NN=N: H H-C-NEN=N: H A H H -H N=N―N=0 ター O D C Ο Α OB C H-CNN N B H H—C—N=N=N: D H H 8 1 point Which is a reasonable Lewis structure for the OF* ion? |:0—F:)* | Q=F ]* |:0—F:]* [:0—F:]* A В с D 0 000 ACDB А O c О в 9 1 point In the Lewis structure of the nitrite anion, NO₂, if all atoms obey the octet rule, there must be: O2 N=0 bonds 2 N-O bonds 1 N-O bond and 1 NEO bond 1 N-O bond and 1 N=O bond 10 1 point What are the formal charges on each of the atoms in the anion: |:0=c=s: s:] O O=-1, CI = 0, S = 0 O=0, CI = -1, S = 0 O=+1, CI = -2, S = 0 O=0, CI = -2, S = +1
Expert Answer:
Answer rating: 100% (QA)
4 which lewis stuetuu would the phosphide ion have in ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The ground-state electron configuration of an atom is 1s 2 2s 2 2p 6 3s 2 3p 4 . What is the valence-shell configuration of the atom in the same group, but in Period 5?
-
A Lewis structure obeying the octet rule can be drawn for O2 as follows: Use the molecular orbital energy- level diagram for O2 to show that the above Lewis structure corresponds to an exicted state....
-
There are two contributing resonance structures for an anion called acetaldehyde enolate, whose condensed molecular formula is CH2CHO-. Draw the two resonance contributors and the resonance hybrid,...
-
1. (Adverse Selection) Consider a labor market model with many identical firms hiring workers. The firms produce a homogeneous product with a constant-returns-to-scale technology and act as price...
-
Selected information concerning the operation of Kern Company for the year ended December 31, 2014, is available as follows: Units produced .............. 10,000 Units sold ............... 9,000...
-
The preparation and analysis of comparative balance sheets and income statements are commonly applied tools of financial statement analysis and interpretation. Required: a. Discuss the inherent...
-
Which type of audit involves a review of general and application controls, with a focus on determining if there is compliance with policies and adequate safe guarding of assets? a. information...
-
Coleman and Rizzuto, architects, have been using a simplified costing system in which all professional labor costs are included in a single direct cost category, professional labor; and all over head...
-
Read the scenario carefully. Answer the question. Justify your answer by referencing the Criminal Code. Your justification should include knowing the offence, offence classification, the arrest...
-
On Monday morning, you have $3000 in cash on hand. For the next seven days, the following cash requirements must be met: Monday, $5000; Tuesday, $6000; Wednesday, $9000; Thursday, $2000; Friday,...
-
Use the table of water density at different temperatures to determine the theoretical density of water based on the temperature recorded in Part II. Enter it below, including all seven decimal places...
-
What role does ethical leadership play in shaping the ethical climate of organizations, influencing employee perceptions of fairness, integrity, and accountability, and how do ethical considerations...
-
Q6: Evaluate A=-sinx 0 sinx 0 siny cosx -siny 0 Q7: Differentiate a* w.r.t x, where a is constant
-
A line goes through the points (-5, -8) and (5, 2). Find its slope. Enter your answer as a simplified improper fraction, if necessary. Do not include "m =" in your answer. Provide your answer below:
-
Managing Systems Support and Security Before You Begin - Assignment Specific Your supervisor, Carlie Davis, needs to present management with the total cost of your in-house developed SIM system....
-
Ask your grandparents about their plans for income, security, and their retirement years clearly describe this plan. have their plans worked out if their plans have been successful what factors do...
-
Question 17 A projectile is fired from a catapult at an angle of 6 = 30. Not yet answered Marked out of 4.00 Flag question If the initial speed of the projectile is 5 m/s, calculate the time it takes...
-
Below is a sample of the data in the file NFLAttendance which contains the 32 teams in the National Football League, their conference affiliation, their division, and their average home attendance....
-
According to your calculations, a reaction should yield 5.67 g of oxygen, O 2 . What do you expect the volume to be at 23C and 0.894 atm?
-
Predict the geometry of the following ions, using the electron-pair repulsion model. a. BrO 3 b. AsO 4 3 c. CO 3 2 d. H 3 S +
-
A solution is made up by dissolving 18.0 g Na 2 CO 3 10H 2 O in 100.0 g of water. What is the molality of Na 2 CO 3 in this solution?
-
Flying High plc plans to expand rapidly over the next five years and is considering the following forms of finance to support that expansion. (a) A five-year 10 million floating-rate term loan from...
-
Convertibles are great because they offer a lower return than straight debt and we just dish out shares rather than have to find cash to redeem the bonds executive at Myopic plc. Comment on this...
-
What do you understand by the term monetary policy?

Study smarter with the SolutionInn App


