CHE 308 Chemical Process Analysis Sessional Class Work 1 Time: 60 min In a sulfuric acid...
Fantastic news! We've Found the answer you've been seeking!
Question:
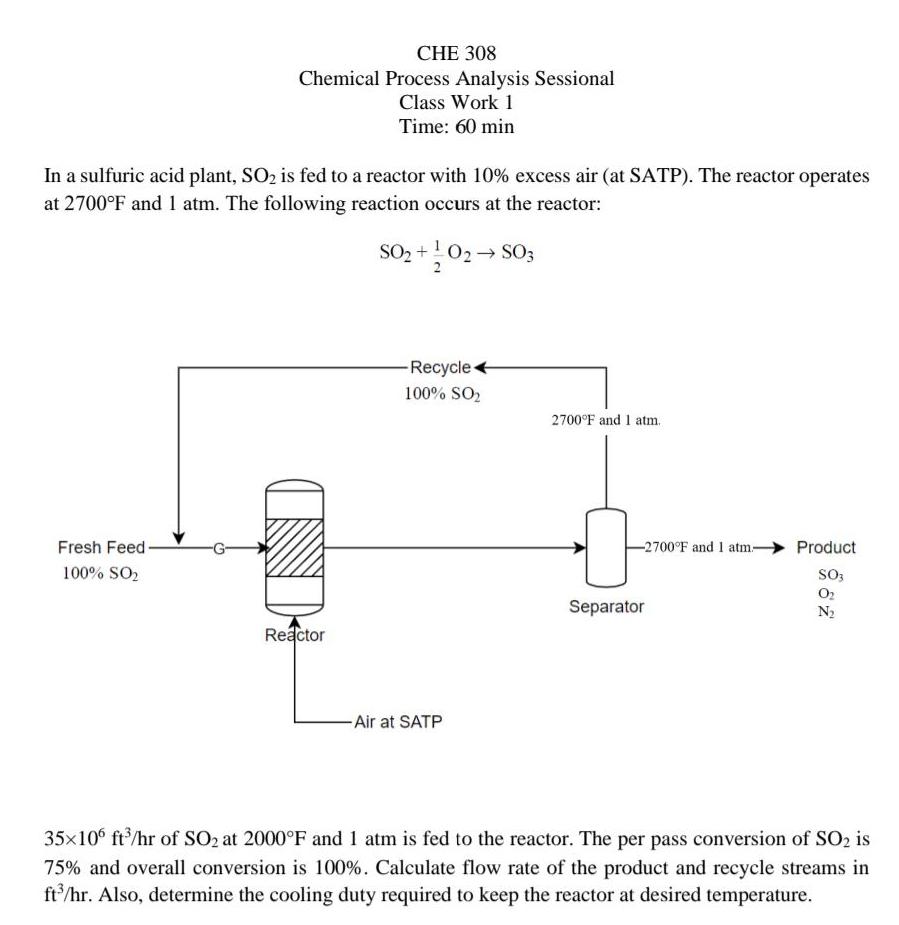
Transcribed Image Text:
CHE 308 Chemical Process Analysis Sessional Class Work 1 Time: 60 min In a sulfuric acid plant, SO2 is fed to a reactor with 10% excess air (at SATP). The reactor operates at 2700°F and 1 atm. The following reaction occurs at the reactor: SO2 +!02 SO3 Recycle + 100% SO2 2700°F and 1 atm. Fresh Feed- -2700°F and 1 atm- Product 100% SO, So3 Separator O2 N2 Reactor -Air at SATP 35x10° ft/hr of SO2 at 2000°F and 1 atm is fed to the reactor. The per pass conversion of SO2 is 75% and overall conversion is 100%. Calculate flow rate of the product and recycle streams in ft'/hr. Also, determine the cooling duty required to keep the reactor at desired temperature. CHE 308 Chemical Process Analysis Sessional Class Work 1 Time: 60 min In a sulfuric acid plant, SO2 is fed to a reactor with 10% excess air (at SATP). The reactor operates at 2700°F and 1 atm. The following reaction occurs at the reactor: SO2 +!02 SO3 Recycle + 100% SO2 2700°F and 1 atm. Fresh Feed- -2700°F and 1 atm- Product 100% SO, So3 Separator O2 N2 Reactor -Air at SATP 35x10° ft/hr of SO2 at 2000°F and 1 atm is fed to the reactor. The per pass conversion of SO2 is 75% and overall conversion is 100%. Calculate flow rate of the product and recycle streams in ft'/hr. Also, determine the cooling duty required to keep the reactor at desired temperature.
Expert Answer:
Answer rating: 100% (QA)
Ansuen t So2 s fed to de actor with 1o Pxcess ay Pracar oper... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The equilibrium constant for the reaction SO3 SO2 + O has the following values: Determine the average heat of dissociation using graphical method. T 800 K 900 K 1000 K 1105K | 0.0319 | 0.153 | 0.540...
-
Air at 1 atm, 60 F is compressed to 4 atm, after which it is expanded through a nozzle back to the atmosphere. The compressor and the nozzle are both reversible and adiabatic and kinetic energy...
-
A chemical reactor process whose production rate is a function of catalyst addition is shown in block diagram form in Figure P10.7 [10]. The time delay is T = 50 s, and the time constant T is...
-
Find the eccentricity and the distance from the pole to the directrix of the conic. Then identify the conic and sketch its graph. Use a graphing utility to confirm your results. r = 6 2 + cos 0
-
On one of my first dates with my wife, we went to see the movie Space balls and paid $5 per ticket. A: Halfway through the movie, my wife said: What on earth were you thinking? This movie sucks! I...
-
In 2008, Amphenol Industrial purchased a new quality inspection system for $550,000. The estimated salvage value was $50,000 after 8 years. Currently the expected remaining life is 3 years with an...
-
Refer to Samsungs financial statements in Appendix A. Compute its debt ratio as of December 31, 2015, and December 31, 2014. Data From Samsung Financial Statement Appendix A Samsung Electronics Co.,...
-
Using the payback period and unadjusted rate of return to evaluate alternative investment opportunities Louis Gallo owns a small retail ice cream parlor. He is considering expanding the business and...
-
(b) If the game below is repeated twice, the following strategies form an SPNE when = 1: Strategies Player 1 period 1: Play C period 2: Play B if (C,Y) in stage 1. Play A otherwise. Player 2 period...
-
Consider the calculation of the continuation value. Suppose Firm C has a WACC of 7%. Firm C's investment banker prepares a DCF valuation of Firm C using the WACC ap- proach. In her DCF valuation,...
-
Describe the two factors that make social, local, and mobile marketing different from traditional online marketing. Why are social, mobile, and local marketing efforts interconnected? 2. What are the...
-
Describe various opportunity costs of attending a four-year college (assuming a full-time schedule).Given these opportunity costs, why do people choose a four-year college experience? In your own...
-
A student must choose a program of four courses from a menu of courses consisting of English, Algebra, Geometry, History, Art, and Latin. This program must contain English and at least one...
-
Three identical resistors with resistances of 6.0 22 are connected as shown in the diagram below to a battery with an emf of 18.0V and zero internal resistance. (a) Find the equivalent resistance of...
-
Arm and Hammer Construction (AHC) is considering a change in its capital structure. The company has $40 million in debt carrying a rate of 6%, and its stock price is $80 per share with 2 million...
-
Dr. Richard Ferber, a pediatric sleep expert, has developed a method to help children, 6 months of age or older, sleep through the night. Often called "Ferberizing," it calls for parents to wait for...
-
Explain whether the following statements are facts: Canberra is 320 kilometres from Sydney Depreciation expense for Company X for 2008 was $1294000. (This is the amount reported on the income...
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
Although nitrogen dioxide (NO2) is a stable compound, there is a tendency for two such molecules to combine to form dinitrogen tetroxide (N2O4). Why? Draw four resonance structures of N2O4, showing...
-
Write structural formulas for the following organic compounds: (a) 3-methylhexane. (b) 1,3,5-trichlorocyclohexane. (c) 2,3-dimethylpentane. (d) 2-bromo-4-phenylpentane. (e) 3,4,5-trimethyloctane.
-
Two atoms have the electron configurations 1s22s22p6 and 1s22s22p63s1. The first ionization energy of one is 2080 kJ/mol, and that of the other is 496 kJ/mol. Match each ionization energy with one of...
-
One difference between accounting for a governmental (not-for-profit) unit and a commercial (for-profit) enterprise is that a governmental unit should a. Not record depreciation expense in any of its...
-
Carson Citys general fund issued purchase orders of $630,000 to vendors for supplies. Which of the following entries should the city make to record this transaction? Debit Credit Q. ENCUMBRANCES...
-
Belle Valley incurred $100,000 of salaries and wages for the month ended March 31, 20X2. How should this be recorded on that date? c. Expenditures Salaries and Wages Debit Vouchers Payable Salaries...

Study smarter with the SolutionInn App


