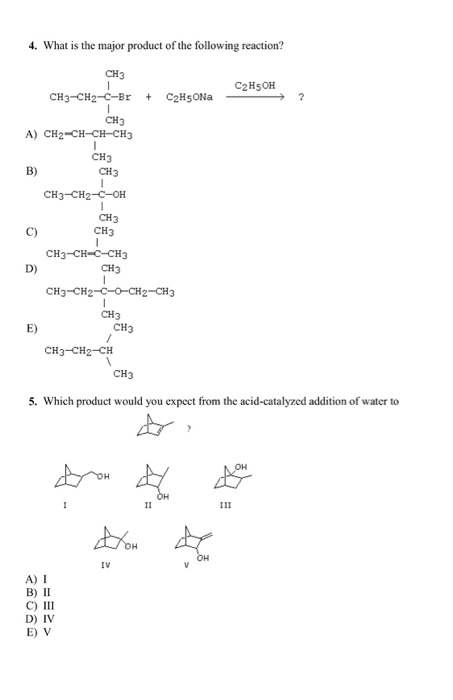
4. What is the major product of the following reaction? CH3 I CH3-CH2-C-Br + CH5ONa I...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
4. What is the major product of the following reaction? CH3 I CH3-CH2-C-Br + C₂H5ONa I CH3 A) CH2-CH-CH-CH3 I CH3 B) D) E) CH3-CH2-C-OH I CH3 CH3 1 CH3 CH3-CH-C-CH3 CH3 I CH3-CH2-C-O-CH2-CH3 I CH3 CH3-CH2-CH A) I C) III D) IV E) V I CH3 5. Which product would you expect from the acid-catalyzed addition of water to CH3 DOH IV OH V OH C₂H5OH III 4. What is the major product of the following reaction? CH3 I CH3-CH2-C-Br + C₂H5ONa I CH3 A) CH2-CH-CH-CH3 I CH3 B) D) E) CH3-CH2-C-OH I CH3 CH3 1 CH3 CH3-CH-C-CH3 CH3 I CH3-CH2-C-O-CH2-CH3 I CH3 CH3-CH2-CH A) I C) III D) IV E) V I CH3 5. Which product would you expect from the acid-catalyzed addition of water to CH3 DOH IV OH V OH C₂H5OH III
Expert Answer:
Answer rating: 100% (QA)
4 What is the major product of the following reaction CH3 CH3 A CH2 CH... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
What organic products A-H would you expect from each of the following reactions? MeLi (1) NaH HELO A (2) NH4Cl,H20 OMS Ni,B (P-2), H2 NaBH MeOH (1) LAH (2) aq. H.SO4 MsCI PyT
-
What products would you expect from the following reactions? (a) (b) (c) (d) (e) OMe OMe
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
(a) Use a graphing utility to graph the curve given by (b) Describe the graph and confirm your result analytically. (c) Discuss the speed at which the curve is traced as t increases from -20 to 20. x...
-
PennTech Corporation has been producing two precision bearings, components T79 and B81, for use in production in its central Pennsylvania plant. Data regarding these two components follow. PennTechs...
-
After you pick up a spare, your bowling ball rolls without slipping back toward the ball rack with a linear speed of vi = 2.62 m/s (FIGURE 10-34). To reach the rack, the ball rolls up a ramp that...
-
An Eagleton Institute poll asked men if they agreed with this statement: Abortion is a private matter that should be left to women to decide without government intervention. Among the men who were...
-
For several years, Dolphin Co.s sales have been on a cash only basis. On January 1, 2009, however, Dolphin Co. began offering credit on terms of n/30. The amount of the adjusting entry to record the...
-
Janice Huffman has decided to start Perez Cleaning, a residential housecleaning service, company. She is able to rent cleaning equipment at a cost of $790 per month. Labor costs are expected to be...
-
Draw a normal curve and identify the approximate location of the following scores. (a) You have the most frequent score. (b) You have a low-frequency score, but the score is higher than most. (c) You...
-
In July 2022 Henry saw a notice in his local newsagents advertising a 'room in a shared flat'. He telephoned and arranged to see the registered freehold owner, Rose, who did not live in the flat....
-
What are two aspects of project planning that will ensure success ?Explain
-
Match each table with its equation. 1 t Y y = 2 y = x y = VI Y = x Submit Question b. P d. Input -2 Input 2 1 Input -2 0 0 1 2 3 1 2 3 2 3 Input -1 0 1 2 3 Input -2 -1 0 1 2 3 Input -2 -1 0 1 4 9...
-
Write a complete C++ program that asks the user to type two positive integers. Validate the input. It should show a horizontal bar of X symbols whose length is given by the shorter of the two...
-
In the following network, node S transmits packets that pass through a network of 16 routers, and arrive at the destination D. The bit rate of all links is R = 10 Mbit/sec. The packet size is 100...
-
Collaboration in educational institutions usually involves cooperation among students, faculties, sponsors, and universities. Unlike the corporate world, higher education institutions have generally...
-
GOOD DAY sells a special type of flashlights which does not use dry-cell batteries but operates with "sun energy". The product would incur a total variable cost of P18 per unit to manufacture and...
-
Find the reduced echelon form of each of the matrices given in Problems 120. c 1 26 + 4
-
Show how you would convert 3-methylbenzenamine into each of the following compounds: (a) methylbenzene; (b) 1-bromo-3-methylbenzene; (c) 3-methylphenol; (d) 3-methylbenzonitrile; (e)...
-
Use the Michael Addition methods , with other reactions if necessary, to synthesize each of the following compounds. In each case, your starting materials should include one aldehyde or ketone and...
-
In each of the following pairs of compounds, indicate which is the stronger acid and which is the stronger base? (a) CH3SH, CH3OH; (b) HS-, HO-; (c) H3S+, H2S.
-
a. Suppose that General Hospital has a current ratio of 0.5. Which of the following actions would improve (increase) this ratio? Use cash to pay off current liabilities. Collect some of the current...
-
What is the role of internal control in an organization?
-
What are the elements and principles of the COSO framework?

Study smarter with the SolutionInn App


