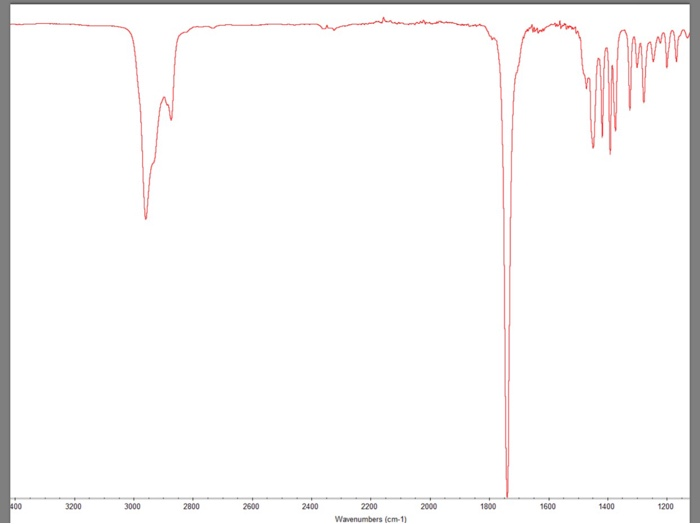
400 3200 3000 2800 2600 2400 2200 Wavenumbers (cm-1) 2000 1800 1600 P 1400 1200 What...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
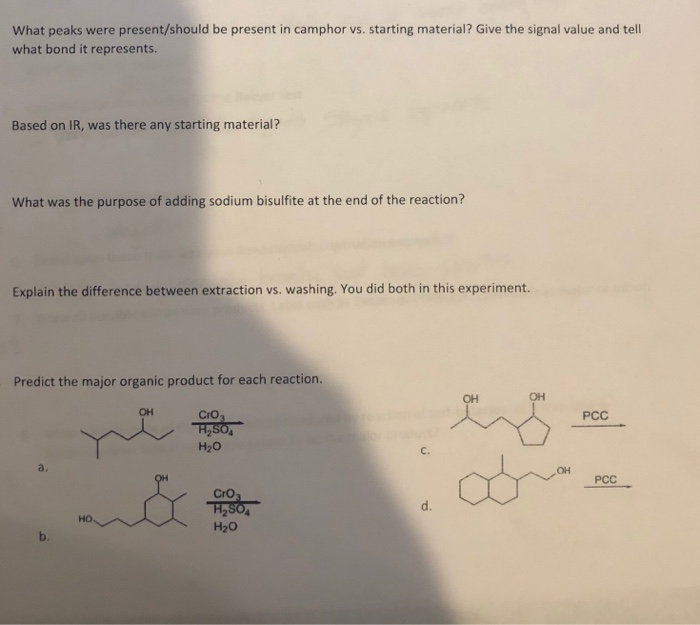
400 3200 3000 2800 2600 2400 2200 Wavenumbers (cm-1) 2000 1800 1600 P 1400 1200 What peaks were present/should be present in camphor vs. starting material? Give the signal value and tell what bond it represents. Based on IR, was there any starting material? What was the purpose of adding sodium bisulfite at the end of the reaction? Explain the difference between extraction vs. washing. You did both in this experiment. Predict the major organic product for each reaction. a. HO OH CrO H₂SO4 H₂O CrO3 H₂SO4 H₂O C. d. OH OH PCC PCC 400 3200 3000 2800 2600 2400 2200 Wavenumbers (cm-1) 2000 1800 1600 P 1400 1200 What peaks were present/should be present in camphor vs. starting material? Give the signal value and tell what bond it represents. Based on IR, was there any starting material? What was the purpose of adding sodium bisulfite at the end of the reaction? Explain the difference between extraction vs. washing. You did both in this experiment. Predict the major organic product for each reaction. a. HO OH CrO H₂SO4 H₂O CrO3 H₂SO4 H₂O C. d. OH OH PCC PCC
Expert Answer:
Answer rating: 100% (QA)
Answer Camphor is an organic compound generally prepared from borneol you have not mentioned startin... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Predict the major organic product for each of the following reactions. a. b. c. d. e. f. ? PCC C-Clz - - NazCrz07 H2SO4. H20
-
Predict the major organic product from each of the following reactions. (a) (b) (c) (d) (1) CH MgBr (2) HO (1) MgBr (2) NH,CI, H20 MgBr (1 equiv.) O (1) o (2) H,O (1) CH,CHLi (excess) (2) NH,CI, H2O O
-
Predict the major organic product from each of the following reaction sequences. (a) (b) (c) (d) (e) (f) (1) MeMgBr (excess) (2) NH,CI, H20 (1) Mg (2) H H (3) HyO (1) PBr3 (2) Mg (3) H,o OH OH (1)...
-
Timco is considering the construction of a new retail outlet. The construction cost will be 400000. Net working capital will increase by 10000. The depreciation is 10 year MACRS. The new location...
-
The CFO of Mega Munchies recently received a report that contains the following information: The WACC is 12 percent if the firm does not have to issue new common equity; if new common equity is...
-
A box rests on the (frictionless) bed of a truck. The truck driver starts the truck and accelerates forward. The box immediately starts to slide toward the rear of the truck bed. Discuss the motion...
-
Refer to the information in Problem 21-1B. Tohono Companys actual income statement for 2017 follows. Required 1. Prepare a flexible budget performance report for 2017. Analysis Component 2. Analyze...
-
On September 1, 2012, Winans Corporation acquired Aumont Enterprises for a cash payment of $700,000. At the time of purchase, Aumonts balance sheet showed assets of $620,000, liabilities of $200,000,...
-
Discuss the breath of responsibilities required of a radiographer. List the five main areas of responsibility in each specialty of radiologic technology. Discuss advantages to working in each of the...
-
Tucker, Inc., produces high-quality suits and sport coats for men. Each suit requires 1.2 hours of cutting time and 0.7 hours of sewing time, uses 6 yards of material, and provides a profit...
-
Kinmi Financial Corporation is the parent company of Kinmi Bank. The company's stock split was announced in the following wire: LOS ANGELES Jan. 20 BUSINESS WIRE-Kinmi Financial Corporation (Nasdaq),...
-
What would be the pro's and con's of having welfare applicants being drug tested before they receive welfare benefits?
-
At the conclusion of a client meeting, your client says that they heard they can contribute mutual funds to a charitable organization in-kind versus in cash and deduct it on their return. Their AGI...
-
It seems that every student who took chemistry in high school remembers that one mole of air occupies 22.4 L. However, many students forget that this conversion is valid only for specific conditions....
-
Calculate the principle paid over the first year on a mortgage of $143,500 with an interest rate of 5.5%. Monthly payments are $815. What is the total principle paid for the year? What is the ending...
-
Find the work W done by the 15-newton force. Use two significant figures in your answer. Express your answer in joules. | ? W= J
-
Say HVN issued a $100 million 30-year bond at par on 31.March, 2000 (lets call it Bond A). Bond A has a yield-to-maturity today of 6.5% (today being the day Min and Kelvin are having their...
-
If the amplifier indicated by the box input impedance of oo, which of the following statements are true ? has an open loop gain as well as Feedback factor (\beta = 1/ R_1\) The feedback is voltage...
-
An optically active fat, when completely hydrolyzed, yields twice as much stearic acid as palmitic acid. Draw the structure of the fat.
-
Design a multistep synthesis to show how each of the following compounds could be prepared from the given starting material: a. b. c. d. H3CH2CH2CH2Br e. BrCH2CH2CH2CH2Br O-O-OR CH-CH2...
-
Upon treatment with ozone followed by work-up with hydrogen peroxide, an alkene forms formic acid and a compound that shows three signals (a singlet, a triplet, and a quartet) in its 1 H NMR...
-
Consider the following cash flow profile, and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...

Study smarter with the SolutionInn App


