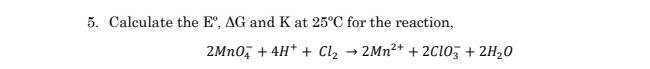5. Calculate the E, AG and K at 25C for the reaction, 2Mno, + 4H* +...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
5. Calculate the E°, AG and K at 25°C for the reaction, 2Mno, + 4H* + Cl2 2MN2+ + 2C10, + 2H20 5. Calculate the E°, AG and K at 25°C for the reaction, 2Mno, + 4H* + Cl2 2MN2+ + 2C10, + 2H20
Expert Answer:
Answer rating: 100% (QA)
Step 1 During electrochemical reaction redox reaction occur in electrolyti... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
Calculate the standard free-energy change at 25C for the following reaction. 3Cu(s) + 2NO3 (aq) + 8H+(aq) 3Cu2+(aq) + 2NO(g) + 4H2O(l)
-
Calculate the standard free-energy change at 25C for the following reaction. 4Al(s) + 3O2(g) + 12H + (aq) 4Al3 + (aq) + 6H2O(l) Use standard electrode potentials.
-
a. Calculate K1 at 25C for phosphoric acid: b. Which thermodynamic factor is the most significant in accounting for the fact that phosphoric acid is a weak acid? Why? H3PO4(aq)--H"(aq) + H,PO4-(aq)...
-
Professional Codes of Ethics Discuss why lawyers are sometimes viewed as being unethical. Is the average lawyer more or less ethical than the average business manager?
-
Process costing: determining equivalent units and allocating costs Bell Corporation, which makes suitcases, completed 30,000 suitcases in August 2011. At the end of August, work in process inventory...
-
Write expressions for the magnitudes and directions of the electric fields measured in Problem 24. Data from Problem 24 A positively charged particle is at rest on the positive \(z\) axis in...
-
During 2013, Jake Gore worked for two different employers. Until May, he worked for Wonderman Construction Company in Kansas City, Kansas, and earned $21,500. The state unemployment rate for...
-
8. A dielectric slab of dielectric constants k is slowly inserted inside the parallel plate capacitor having plate area A and separation between plates d as shown in figure. If dimensions of...
-
Refer to Exercise 29 of Section 4.2. For what values of h will a sensitivity analysis on the effect of a change of h pounds of peat be valid? In exercise A lawn and garden store creates three...
-
A facility is to house 50 female and 50 male employees. Using a 40% allowance for aisles and clearances, how much space should be planned for the restrooms?
-
Is it possible to improve one governance principle in a firm but weaken another at the same time? Use an illustration to explain your answer.
-
After extensive medical and marketing research, Pill plc believes it can penetrate the pain reliever market. It is considering two alternative products. The first is a medication for headache pain....
-
Why would we expect managers of a corporation to pursue the objectives of shareholders? What about bondholders?
-
Discuss the evolution of corporate governance in the United Kingdom. In what way are the Codes linked to each other? Explain.
-
Petracci SpA manufactures fine furniture. The company is deciding whether to introduce a new mahogany dining room table set. The set will sell for 5,600, including a set of eight chairs. The company...
-
Mary is preparing to conduct the survey you designed for her. She needs 500 people to complete the survey but she needs your help to develop a recruitment and distribution plan. 1. Define a sample...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
Ferns that eject spores generally do so in pairs, with two spores flying off in opposite directions. The structure from which the spores are launched is quite lightweight. If it takes a certain...
-
A dog can provide sufficient power to pull a sled with a 60 N force at a steady 2.0 m/s. Suppose the dog is hitched to a different sled that requires 120 N to move at a constant speed. How fast can...
-
Most of the energy you expend in cycling is dissipated by the drag force. If you double your speed, you increase the drag force by a factor of 4. This increases the power to cycle at this greater...

Study smarter with the SolutionInn App