(5) Calculate the volume of CO gas at STP obtained by reacting a 10.00 g sample...
Fantastic news! We've Found the answer you've been seeking!
Question:
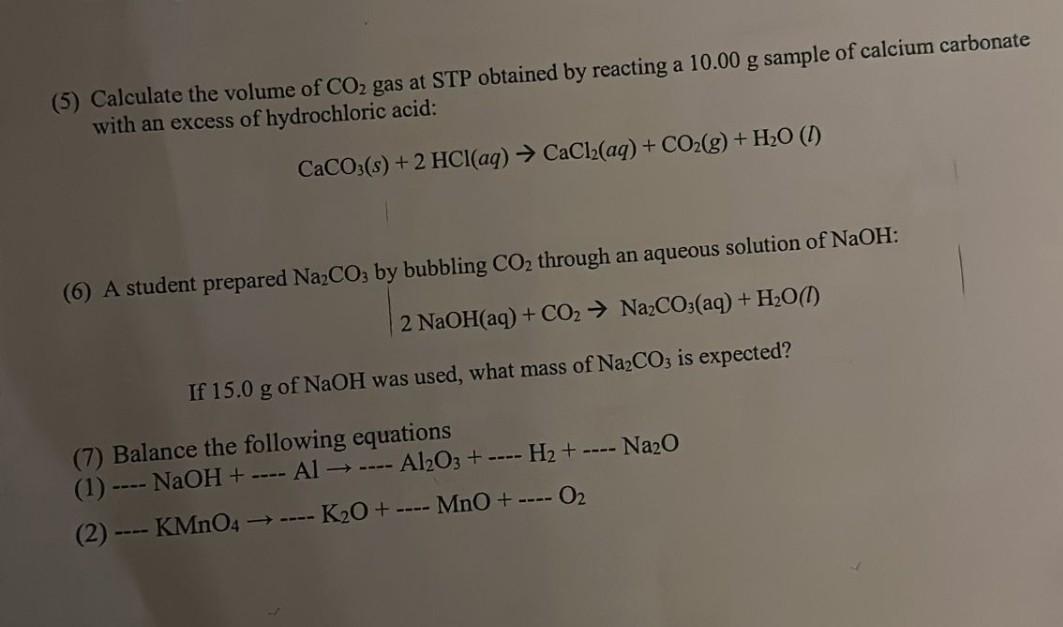
Transcribed Image Text:
(5) Calculate the volume of CO₂ gas at STP obtained by reacting a 10.00 g sample of calcium carbonate with an excess of hydrochloric acid: CaCO3(s) + 2 HCl(aq) → CaCl₂(aq) + CO₂(g) + H₂O (1) (6) A student prepared Na₂CO3 by bubbling CO₂ through an aqueous solution of NaOH: 2 NaOH(aq) + CO₂ → Na₂CO3(aq) + H₂O(1) If 15.0 g of NaOH was used, what mass of Na₂CO3 is expected? (7) Balance the following equations (1) - - NaOH + ---- (2) ---- KMnO4 → ---- ---- Al → ---- Al2O3+ - K₂O + H₂+ ---- MnO + ---- 0₂ Na₂O (5) Calculate the volume of CO₂ gas at STP obtained by reacting a 10.00 g sample of calcium carbonate with an excess of hydrochloric acid: CaCO3(s) + 2 HCl(aq) → CaCl₂(aq) + CO₂(g) + H₂O (1) (6) A student prepared Na₂CO3 by bubbling CO₂ through an aqueous solution of NaOH: 2 NaOH(aq) + CO₂ → Na₂CO3(aq) + H₂O(1) If 15.0 g of NaOH was used, what mass of Na₂CO3 is expected? (7) Balance the following equations (1) - - NaOH + ---- (2) ---- KMnO4 → ---- ---- Al → ---- Al2O3+ - K₂O + H₂+ ---- MnO + ---- 0₂ Na₂O (5) Calculate the volume of CO₂ gas at STP obtained by reacting a 10.00 g sample of calcium carbonate with an excess of hydrochloric acid: CaCO3(s) + 2 HCl(aq) → CaCl₂(aq) + CO₂(g) + H₂O (1) (6) A student prepared Na₂CO3 by bubbling CO₂ through an aqueous solution of NaOH: 2 NaOH(aq) + CO₂ → Na₂CO3(aq) + H₂O(1) If 15.0 g of NaOH was used, what mass of Na₂CO3 is expected? (7) Balance the following equations (1) - - NaOH + ---- (2) ---- KMnO4 → ---- ---- Al → ---- Al2O3+ - K₂O + H₂+ ---- MnO + ---- 0₂ Na₂O
Expert Answer:
Answer rating: 100% (QA)
The question youve presented is question 6 which requires calculating the expected mass of sodium ca... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
You bought an apartment of UF 2,300, taking a loan for 80% of the value of the apartment, making monthly payments of UF 10.59350642 using an effective annual rate of 3.85%. Calculate the nominal...
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
A 15.0-mL sample of an oxalic acid solution requires 25.2mL of 0.149 M NaOH for neutralization. Calculate the volume of a 0.122 M KMnO4 solution needed to react with a second 15.0-mL sample of the...
-
True or False: 1. Economists are not completely in agreement on what constitutes money for all purposes. 2. People use nontransaction accounts primarily because they generally pay higher interest...
-
Suppose Carat, Inc., reported net receivables of $2,584 million and $2,265 million at January 31, 2017, and 2016, respectively, after subtracting allowances of $68 million and $65 million at these...
-
Plasma Ltd, a manufacturer of electrical goods, guarantees them for 12 months from the date of purchase by the customer. If a fault occurs after the guarantee period, but is due to faulty manufacture...
-
5. Sam Kay signed a promissory note for $220,000 that was payable to Investments, S.A., Inc., a company of which he was the principal stockholder. The company then indorsed the note over to its...
-
John Campbell, an employee of Manhattan Construction Company, claims to have injured his back as a result of a fall while repairing the roof at one of the Eastview apartment buildings. He filed a...
-
Statement of Partnership Liquidation Schedule of Cash Payments to support the distribution of cash as it becomes available. ACCTG.112 - Activity Week 10 and week 12 MEA Partnership Post-closing Trial...
-
The first Brillouin zone of an infinite chain of H atoms has an infinite number of crystal orbitals, each with a different value of k. (a) State the limiting values of k within the first Brillouin...
-
Why should a speaker vary using logic and emotion as supporting material based on the audience?
-
Thunder Corporation, an amusement park, is considering a capital investment in a new exhibit. The exhibit would cost $144,123 and have an estimated useful life of 7 years. It can be sold for $68,000...
-
(a) Calculate the absolute pressure at the bottom of a fresh-water lake at a depth of 24.0 m. Assume the density of the water is 1.00 10 3 kg/m 3 and the air above is at a pressure of 101.3 kPa. Pa...
-
Determine the R value and U-factor for the following assemblies.Assume winter conditions. Use the attached Tables. Exterior Wall Component R-Value 3/8\" Plywood 1\" Air Space 3 1/2\" Fiberglass Batt...
-
Match each concept to the corresponding example. Drag each item on the left to its matching item on the right. transnational nongovernmental organizations cultural hybridization transnational...
-
DISCUSS THE CONCEPT OF Change Management.show the barriers of organizational change
-
1. make research using SERVQUAL model and describe and proposed flow diagram illustrating the how service delivery and consumer satisfaction will be measured in the cryptocurrencies? 2. make case...
-
The cost curve for the city water supply is C(Q) = 16 + 1/4 Q2, where Q is the amount of water supplied and C(Q) is the cost of providing Q acre-feet of water. (An acre-foot is the amount of water...
-
At STP, 0.280 L of a gas weighs 0.400 g. Calculate the molar mass of the gas.
-
The alcohol content of hard liquor is normally given in terms of the "proof," which is defined as twice the percentage by volume of ethanol (C2H5OH) present. Calculate the number of grams of alcohol...
-
How many carbon-carbon sigma bonds are present in each of the following molecules? (a) 2-butyne. (b) Anthracene (c) 2,3-dimethylpentane
-
One mile is equal to \(1.60934 \mathrm{~km}\). Convert 200 miles to kilometers. Round off the answer to three decimal places.
-
It is known that 4 quarts equals 3.785 liters. If you have 25 quarts, how many liters do you have? Round off to three decimal places.
-
Rewrite the following as fractions: 1. \(4 \%\) 2. \(50 \%\)

Study smarter with the SolutionInn App


