5. QIN Bove m -3- HP L.P. to COND- QOUT wp AN IDEAL REGEAT RANKINE CYCLE...
Fantastic news! We've Found the answer you've been seeking!
Question:
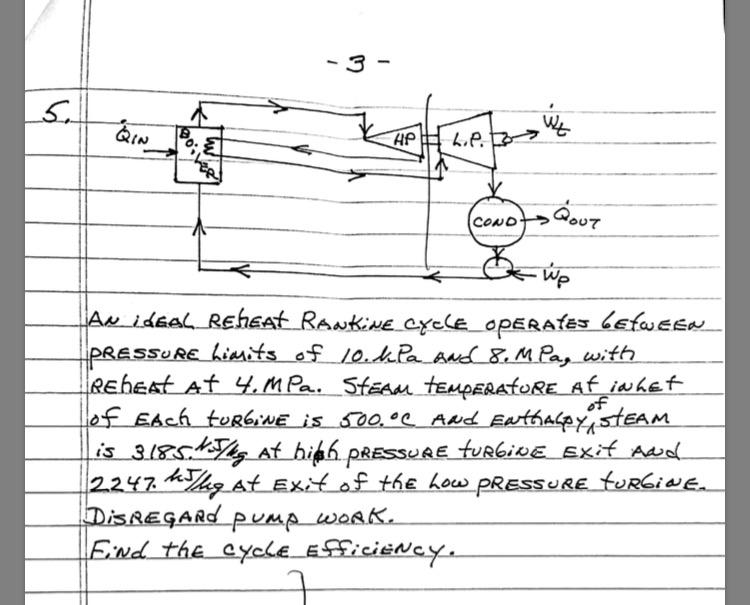
Transcribed Image Text:
5. QIN Bove m -3- HP L.P. to COND- QOUT wp AN IDEAL REGEAT RANKINE CYCLE OPERATES BETWEEN -PRESSURE Limits of 10. kPa And 8. M Pag with REGEAT At 4. MPa. StEAM TEMPERATURE At in het of EACH FURGiNE is 500,°C AND ENthalpy STEAM is 3185. NJ/kg At high PRESSURE TURGiNE Exit And 2247. Jhg At Exit of the Low PRESSURE tuRGINE. MJ/kg DISREGARD PUMP WORK. Find the CYCLE EFFICIENCY. 5. QIN Bove m -3- HP L.P. to COND- QOUT wp AN IDEAL REGEAT RANKINE CYCLE OPERATES BETWEEN -PRESSURE Limits of 10. kPa And 8. M Pag with REGEAT At 4. MPa. StEAM TEMPERATURE At in het of EACH FURGiNE is 500,°C AND ENthalpy STEAM is 3185. NJ/kg At high PRESSURE TURGiNE Exit And 2247. Jhg At Exit of the Low PRESSURE tuRGINE. MJ/kg DISREGARD PUMP WORK. Find the CYCLE EFFICIENCY.
Expert Answer:
Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these mechanical engineering questions
-
An ideal Rankine steam cycle modified with two closed feedwater heaters and one open feedwater heater is shown below. The power cycle receives 100 kg/s of steam at the high pressure inlet to the...
-
An ideal Rankine steam cycle modified with two closed feedwater heaters is shown below. The power cycle receives 75 kg/s of steam at the high pressure inlet to the turbine. The feedwater heater exit...
-
Steam at 500 C and 4 MPa enters a turbine with an isentropic efficiency of 86%. The exit pressure is 100 kPa. The exiting temperature of the steam is nearest: (A) 95 C (B) 107 C (C) 118 C (D) 126 C
-
Calculate the following ratios for both 2021 and 2020 years: current ratio, quick ratio, debt to equity ratio, debt ratio, gross profit ratio, net profit ratio, return on equity, return on total...
-
How successful have companies in the United States been in adapting new trends in job design that mostly have originated in Japan?
-
When Patey Pontoons issued 6% bonds on January 1, 2021, with a face amount of $600,000, the market yield for bonds of similar risk and maturity was 7%. The bonds mature December 31, 2024 (4 years)....
-
Limo.com had the following balances on December 31, 2010, before the year- end adjustments: Accounts Receivable 104,000 Allowance for Uncollectible Accounts 1,300 The aging of receivables yields...
-
A bakery buys flour in 25- pound bags. The bakery uses 1,215 bags a year. Ordering cost is $ 10 per order. Annual carrying cost is $ 75 per bag. a. Determine the economic order quantity. b. What is...
-
See photo attached for question Homework: MT217 M5 Competency Assessment Score: 0 of 5 pts Problem 7.LO4.16 15 of 20 (17 complete) Save HW Score: 80%, 80 of 100 pts Question Help AU.S. Government...
-
A model for the surface area of some solid object is given by S = 0.171uw0.683 h0.439, where w is the weight (in pounds), h is the height (in inches), and S is measured in square feet. If the errors...
-
Assume you were hired to design a managed care organization targeted to baby boomers in San Antonio Tx a city with large Hispanic population. How would you make service offering to respond to the...
-
Explain put-call parity.
-
Explain how you can use put options to create portfolio insurance. How can you create portfolio insurance using call options?
-
How does an understanding of the accounting equation facilitate the preparation of financial reports?
-
John bought 1,000 shares of Intel stock on October 18, 2015, for $30 per share plus a $750 commission he paid to his broker. On December 12, 2019, he sells the shares for $42.50 per share. He also...
-
Explain how debt can be viewed as an option portfolio.
-
Which of following reasons of first trough in the development history of Artificial Intelligence is incorrect? A . . Many people think AI is just a "toy" B . . Appearance of sensor based on reasoning...
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
One mole of hydrogen and dry air enter a combustion chamber at 25 8C and 1 atm and leave at 800 K. Calculate the heat transfer from the chamber for stoichiometric air. (A) 200 MJ (B) 150 MJ (C) 100...
-
Show that saturated water vapor at 20 kPa can be treated as an ideal gas (compare v = RT/P with vg from Table C-2). Could saturated water vapor at 100 kPa be treated as an ideal gas?
-
The pressure and temperature are measured in a tank containing air to be 60 kPa and 2 80 8 C, respectively. Determine the specific volume using the: i) Ideal-gas law ii) Van der Waals equation iii) Z...
-
The value of \(\Delta W=\int_{1}^{2} P d V\) of an ideal gas in a reversible isothermal process is (a) 0 (b) \(\frac{P_{1} V_{1}-P_{2} V_{2}}{\gamma-1}\) (c) \(P_{1} V_{1} \ln \frac{V_{2}}{V_{1}}\)...
-
On a Mollier chart, the slope of the curve representing a reversible isobaric process is equal to (a) \(T-\beta\) (b) \(T+\frac{1}{\beta}\) (c) \(T\) (d) \(\beta T-1\)
-
The work done in reversible polytropic steady flow processes is given by (a) \(\frac{P_{1} V_{1}-P_{2} V_{2}}{n-1}\) (b) \(\frac{n\left(P_{1} V_{1}-P_{2} V_{2} ight)}{n-1}\) (c) \(n\left(P_{1}...

Study smarter with the SolutionInn App



