6) 2 kg of Oxygen is heated from 30C to 130C. Determine the amount of heat...
Fantastic news! We've Found the answer you've been seeking!
Question:

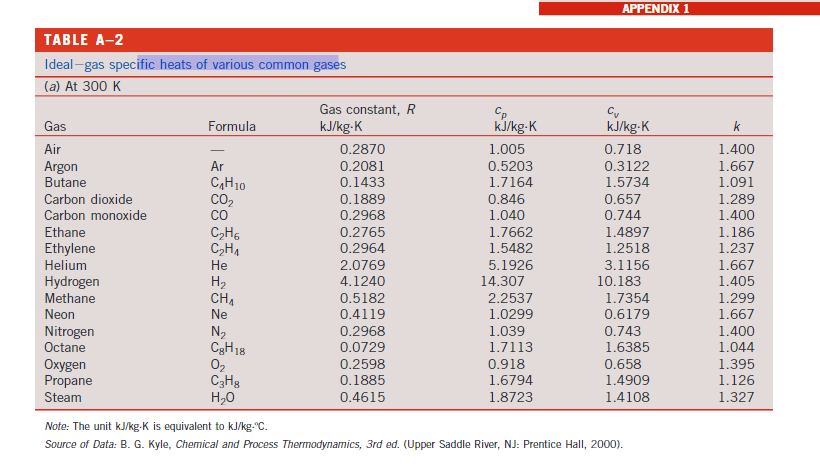
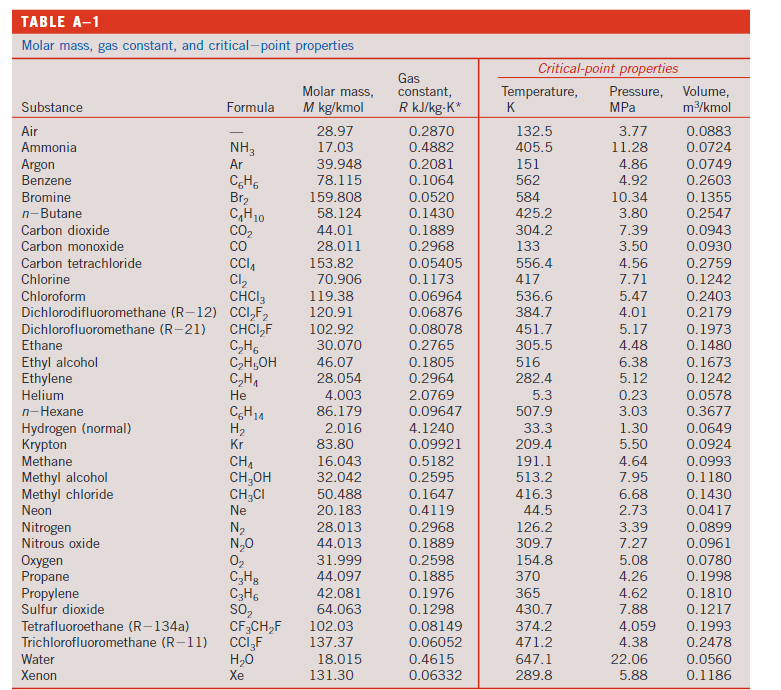
Transcribed Image Text:
6) 2 kg of Oxygen is heated from 30C to 130C. Determine the amount of heat needed if this is done at (a) constant volume and (b) constant pressure. Solve this problem using thermodynamic tables, as well as by using average specific heat, and compare your answers. TABLE A-2 Ideal-gas specific heats of various common gases (a) At 300 K Gas Air Argon Butane Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon Nitrogen Octane Oxygen Propane Steam Formula Ar CH0 CO CH6 CH He H CH Ne N CgH18 02 C3H8 HO Gas constant, R kJ/kg-K 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 0.0729 0.2598 0.1885 0.4615 Cp kJ/kg-K 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 1.7113 0.918 1.6794 1.8723 APPENDIX 1 kJ/kg-K 0.718 0.3122 1.5734 0.657 0.744 1.4897 1.2518 3.1156 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 Note: The unit kJ/kg-K is equivalent to kJ/kg-C. Source of Data: B. G. Kyle, Chemical and Process Thermodynamics, 3rd ed. (Upper Saddle River, NJ: Prentice Hall, 2000). k 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 1.044 1.395 1.126 1.327 TABLE A-1 Molar mass, gas constant, and critical-point properties Substance Air Ammonia Argon Benzene Bromine n-Butane Carbon dioxide Carbon monoxide Carbon tetrachloride Chlorine Chloroform Dichlorodifluoromethane (R-12) Dichlorofluoromethane (R-21) Ethane Ethyl alcohol Ethylene Helium n-Hexane Hydrogen (normal) Krypton Methane Methyl alcohol Methyl chloride Neon Nitrogen Nitrous oxide Oxygen Propane Propylene Sulfur dioxide Tetrafluoroethane (R-134a) Trichlorofluoromethane (R-11) Water Xenon Formula NH3 Ar CH6 Br CH10 CO CO CCIA Cl CHCI 3 CCIF CHCIF CH6 CH5OH CH He C6H1 H Kr CH CHOH CH3CI Ne N N0 0 C3H8 C3H6 SO CF3CHF CCI3F HO Xe Molar mass, M kg/kmol 28.97 17.03 39.948 78.115 159.808 58.124 44.01 28.011 153.82 70.906 119.38 120.91 102.92 30.070 46.07 28.054 4.003 86.179 2.016 83.80 16.043 32.042 50.488 20.183 28.013 44.013 31.999 44.097 42.081 64.063 102.03 137.37 18.015 131.30 Gas constant, R kJ/kg-K* 0.2870 0.4882 0.2081 0.1064 0.0520 0.1430 0.1889 0.2968 0.05405 0.1173 0.06964 0.06876 0.08078 0.2765 0.1805 0.2964 2.0769 0.09647 4.1240 0.09921 0.5182 0.2595 0.1647 0.4119 0.2968 0.1889 0.2598 0.1885 0.1976 0.1298 0.08149 0.06052 0.4615 0.06332 Critical-point properties Temperature, Pressure, MPa K 132.5 405.5 151 562 584 425.2 304.2 133 556.4 417 536.6 384.7 451.7 305.5 516 282.4 5.3 507.9 33.3 209.4 191.1 513.2 416.3 44.5 126.2 309.7 154.8 370 365 430.7 374.2 471.2 647.1 289.8 3.77 11.28 4.86 4.92 10.34 3.80 7.39 3.50 4.56 7.71 5.47 4.01 5.17 4.48 6.38 5.12 0.23 3.03 1.30 5.50 4.64 7.95 6.68 2.73 3.39 7.27 5.08 4.26 4.62 7.88 4.059 4.38 22.06 5.88 Volume, m/kmol 0.0883 0.0724 0.0749 0.2603 0.1355 0.2547 0.0943 0.0930 0.2759 0.1242 0.2403 0.2179 0.1973 0.1480 0.1673 0.1242 0.0578 0.3677 0.0649 0.0924 0.0993 0.1180 0.1430 0.0417 0.0899 0.0961 0.0780 0.1998 0.1810 0.1217 0.1993 0.2478 0.0560 0.1186 6) 2 kg of Oxygen is heated from 30C to 130C. Determine the amount of heat needed if this is done at (a) constant volume and (b) constant pressure. Solve this problem using thermodynamic tables, as well as by using average specific heat, and compare your answers. TABLE A-2 Ideal-gas specific heats of various common gases (a) At 300 K Gas Air Argon Butane Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon Nitrogen Octane Oxygen Propane Steam Formula Ar CH0 CO CH6 CH He H CH Ne N CgH18 02 C3H8 HO Gas constant, R kJ/kg-K 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 0.0729 0.2598 0.1885 0.4615 Cp kJ/kg-K 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 1.7113 0.918 1.6794 1.8723 APPENDIX 1 kJ/kg-K 0.718 0.3122 1.5734 0.657 0.744 1.4897 1.2518 3.1156 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 Note: The unit kJ/kg-K is equivalent to kJ/kg-C. Source of Data: B. G. Kyle, Chemical and Process Thermodynamics, 3rd ed. (Upper Saddle River, NJ: Prentice Hall, 2000). k 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 1.044 1.395 1.126 1.327 TABLE A-1 Molar mass, gas constant, and critical-point properties Substance Air Ammonia Argon Benzene Bromine n-Butane Carbon dioxide Carbon monoxide Carbon tetrachloride Chlorine Chloroform Dichlorodifluoromethane (R-12) Dichlorofluoromethane (R-21) Ethane Ethyl alcohol Ethylene Helium n-Hexane Hydrogen (normal) Krypton Methane Methyl alcohol Methyl chloride Neon Nitrogen Nitrous oxide Oxygen Propane Propylene Sulfur dioxide Tetrafluoroethane (R-134a) Trichlorofluoromethane (R-11) Water Xenon Formula NH3 Ar CH6 Br CH10 CO CO CCIA Cl CHCI 3 CCIF CHCIF CH6 CH5OH CH He C6H1 H Kr CH CHOH CH3CI Ne N N0 0 C3H8 C3H6 SO CF3CHF CCI3F HO Xe Molar mass, M kg/kmol 28.97 17.03 39.948 78.115 159.808 58.124 44.01 28.011 153.82 70.906 119.38 120.91 102.92 30.070 46.07 28.054 4.003 86.179 2.016 83.80 16.043 32.042 50.488 20.183 28.013 44.013 31.999 44.097 42.081 64.063 102.03 137.37 18.015 131.30 Gas constant, R kJ/kg-K* 0.2870 0.4882 0.2081 0.1064 0.0520 0.1430 0.1889 0.2968 0.05405 0.1173 0.06964 0.06876 0.08078 0.2765 0.1805 0.2964 2.0769 0.09647 4.1240 0.09921 0.5182 0.2595 0.1647 0.4119 0.2968 0.1889 0.2598 0.1885 0.1976 0.1298 0.08149 0.06052 0.4615 0.06332 Critical-point properties Temperature, Pressure, MPa K 132.5 405.5 151 562 584 425.2 304.2 133 556.4 417 536.6 384.7 451.7 305.5 516 282.4 5.3 507.9 33.3 209.4 191.1 513.2 416.3 44.5 126.2 309.7 154.8 370 365 430.7 374.2 471.2 647.1 289.8 3.77 11.28 4.86 4.92 10.34 3.80 7.39 3.50 4.56 7.71 5.47 4.01 5.17 4.48 6.38 5.12 0.23 3.03 1.30 5.50 4.64 7.95 6.68 2.73 3.39 7.27 5.08 4.26 4.62 7.88 4.059 4.38 22.06 5.88 Volume, m/kmol 0.0883 0.0724 0.0749 0.2603 0.1355 0.2547 0.0943 0.0930 0.2759 0.1242 0.2403 0.2179 0.1973 0.1480 0.1673 0.1242 0.0578 0.3677 0.0649 0.0924 0.0993 0.1180 0.1430 0.0417 0.0899 0.0961 0.0780 0.1998 0.1810 0.1217 0.1993 0.2478 0.0560 0.1186
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Posted Date:
Students also viewed these mechanical engineering questions
-
Find the 8th term of a GP whose 1st term is 10 and common ratio 3.
-
pleasesolve for last entry (Impairment loss and accumulation depreciationfor dec 31) Cullumber Company has a December 31 fiscal year end and follows ASPE. The following selected transactions are...
-
please answer all parts for parts 3 and 5 for a good rating!! Q3. Differentiate between (a) stand-alone risk and (b) risk in a portfolio context. How are they measured, and are both concepts relevant...
-
A company is implementing Dynamics 365 Supply Chain Management. The company plans to implement the solution in a phased approach across several incremental projects. The project manager needs a...
-
A set of scores measuring aggression is normally distributed with a mean equal to 23 and a standard deviation equal to 2.5. Find the proportion: (a) To the left of x = 19.0 (b) To the right of x =...
-
Green Brands, Inc. (GBI) presents its statement of cash flows using the indirect method. The following accounts and corresponding balances were drawn from GBIs Year 2 and Year 1 year-end balance...
-
Distinguish between turnkey and backbone systems. Which is more flexible?
-
Should a good project plan formulate methods for anticipating problems?
-
Oriole Clothing produces men's athletic socks. The following budgeted and actual amounts are for 2022: Cost Budget at 6,600 Units Actual Amounts at 7,400 Units Direct materials $79,200 $90,590 Direct...
-
Erica and Bob participate in a friendly Hackathon that allows each to solve one question a day out of the three offered. There will be one easy, one medium and one hard question, with points awarded...
-
Divisional income statements with service department charges Instructions Labels and Amount Descriptions Divisional Income Statements X Instructions Grael Technology has two divisions, Consumer and...
-
How can modems improve the reliability of the communication subsystem?
-
When logging input transactions, why is it necessary to distinguish between transactions that have been processed successfully and those that have been processed in error? If this distinction is not...
-
Briefly explain the shadow paging backup and recovery strategy. Give one advantage and one disadvantage of the shadow paging strategy.
-
What factors cause errors in a real memory cell? How are errors often detected?
-
What data must be available in the accounting audit trail so auditors can uniquely identify the process that has been executed on an input data item and the functions performed by that process?
-
Here is a case study with questions I did answer all questions, and Just need someone to read them and tell me if there is any comments or feedback, just need to make sure I am on the right track and...
-
The Ferris wheel in the figure has a radius of 68 feet. The clearance between the wheel and the ground is 14 feet. The rectangular coordinate system shown has its origin on the ground directly below...
-
Air enters a 40-cm-diameter cooling section at 1 atm, 32C, and 70 percent relative humidity at 120 m/min. The air is cooled by passing it over a cooling coil through which cold water flows. The water...
-
Is the effect of turbine and compressor irreversibilities of a turbojet engine to reduce (a) The net work (b) The thrust (c) The fuel consumption rate?
-
A completely reversible air conditioner provides 36,000 Btu/h of cooling for a space maintained at 70F while rejecting heat to the environmental air at 110F. Calculate the rate at which the entropies...
-
The following information is provided for Shaq Co. for the new product it recently introduced. Total unit cost \($30\) Desired ROI per unit \($10\) Target selling price \($40\) What would be Shaq...
-
Sprague Company is considering developing a new product. The company has gathered the following information on this product. Expected total unit cost Estimated investment for new product Desired ROI...
-
Bristle Brush Company operates in a state where corporate taxes and workers' compensation insurance rates have recently doubled. Bristle's president has just assigned you the task of preparing an...

Study smarter with the SolutionInn App


