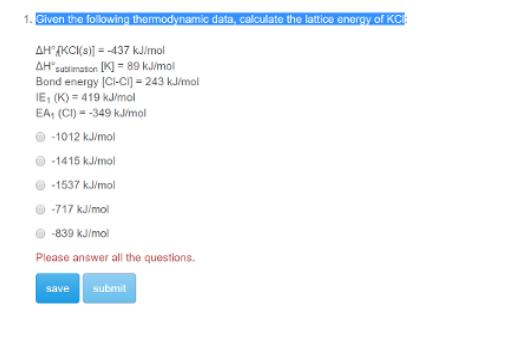
Given the following thermodynamic data, calculate the lattice energy of KC AHKCI(S)]=-437 kJ/mol AHsublimation [K]=89 kJ/mol...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Given the following thermodynamic data, calculate the lattice energy of KC AHKCI(S)]=-437 kJ/mol AH"sublimation [K]=89 kJ/mol Bond energy [CI-CI] = 243 kJ/mol IE, (K) = 419 kJ/mol EA, (CI)-349 kJ/mol -1012 kJ/mol -1415 kJ/mol -1537 kJ/mol -717 kJ/mol -839 kJ/mol Please answer all the questions. save submit Given the following thermodynamic data, calculate the lattice energy of KC AHKCI(S)]=-437 kJ/mol AH"sublimation [K]=89 kJ/mol Bond energy [CI-CI] = 243 kJ/mol IE, (K) = 419 kJ/mol EA, (CI)-349 kJ/mol -1012 kJ/mol -1415 kJ/mol -1537 kJ/mol -717 kJ/mol -839 kJ/mol Please answer all the questions. save submit
Expert Answer:
Answer rating: 100% (QA)
Kes AMsub K g 9 I Allahringute Hall s o lattice kcg llig Jonizati... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Given the following thermodynamic data, calculate the lattice energy of CaBr2(s). Term Value (kJ/mol) ?H?f[CaBr2(s)] -675 ?H?f[Ca(g)] 179 ?H?f[Br(g)] 112 I1(Ca) 590. I2(Ca) 1145 E(Br) -325 Express...
-
please answer all the questions for me B and C (i, ii) (b) You are employed as the Quality Control Officer of Guinness Ghana limited, Kumasi. As part of your responsibilities, you are to ensure that...
-
Practice Problem 1. Use differentiation from first principles to find the derivative of f(x)=x at x = 5.
-
Use transaction analysis charts to analyze the following adjustments: a. Depreciation on equipment, $1,000. b. Rent expired, $600.
-
Jeremy Costa, owner of Costa Cabinets Inc., is preparing a bid on a job that requires $1,800 of direct materials, $1,600 of direct labor, and $800 of overhead. Jeremy normally applies a standard...
-
According to a recent Pew Internet & American Life Project Survey (October 2010), 67% of adults who use the Internet have paid to download music. In a random sample of n = 1,000 adults who use the...
-
A friend of yours tells you that his parents plan to pay for his college education by selling some family jewelry. Write About It Write a paragraph explaining why you might question this plan.
-
Bowles Countys fund structure is as follows: General Fund Three Special Revenue Funds One Capital Projects Fund Two Debt Service Funds Three Internal Service Funds Five Enterprise Funds Assume that...
-
HELP ME FILL THIS FORM PLEASE THE DETAILS ARE GIVEN ABOVE. COLLECTION LETTER: FOREIGN BILLS To: Bank name: Bank Address: We enclose the following item for collection Drawer reference: Date of Draft:...
-
The founder of Frenza asks us to assist her in accounting and analysis of the corporations bonds, which have an annual contract rate of 8%. She wants to know the business and accounting implications...
-
Can you please revise my two summery below I feel like my the wording doesn't make sense? Male-owned businesses are more educated than Women-owned businesses. As you can see in the table below,...
-
Sheffield Industries projects its unit sales of part 998 to be 14,200 units in January, 17,500 units in February, and 19,500 units in March. Sheffield wants its ending finished goods inventory to...
-
Given the equation 5y+ where = 3.410.59 y=3.710.61 and z=1.89 +0.29 What is the absolute uncertainty in f (with the correct number of significant figures)? Select one: O a. 0.3 O b. 1.60 O c. 2 O d....
-
given the following vertex (-4,5) What would be the equation of the axis of symmetry?
-
Jack Ma the co - owner of Alibaba Group is a leading multinational technology conglomerate. With a complex structure that included consolidation, multi - currency transactions, and multiple...
-
A 500-gallon water tank drains at a rate of 50 gallons per minute. The amount of water left in the tank depends on the number of minutes it has been draining. What is the range of this relation?
-
The sorting of codes into categories and then categories into themes would be called Answers: 1) familiarization. 2) organization. 3) coding. 4) reducing.
-
A crop-dusting plane flies over a level field at a height of 25 ft. If the dust leaves the plane through a 30 angle and hits the ground after the plane travels 75 ft, how wide a strip is dusted? See...
-
Nitrogen monoxide, NO, reacts with oxygen to produce nitrogen dioxide. 2NO(g)+ O2(g) 2NO2(g) (overall equation) If the mechanism is what is the predicted rate law? Remember to express this in terms...
-
Consider the reaction Draw a graph illustrating the changes of concentrations of N2O4 and NO2 as equilibrium is approached. Describe how the rates of the forward and reverse reactions change as the...
-
Which of the following is the weakest acid: HClO 4 , HCN, or HC 2 H3O 2 ? See Table 15.1 and Table 16.1. TABLE 15.1 Common Strong Acids and Bases Strong Acids ......................... Strong Bases*...
-
On January 1, 2020, Sharp Company purchased \(\$ 50,000\) of Sox Company \(5 \%\) bonds, at a time when the market rate was \(6 \%\). The bonds mature on December 31, 2024, and pay interest...
-
Assume the same facts as in Brief Exercise 14-16, except that Sharp Company does not intend to trade the bonds or to hold them until maturity. a. Prepare the entry for the purchase of the debt...
-
Tracking Co. holds an AFS bond investment in Fields Corp. The carrying value of the investment is \(\$ 4,500\) at December 31, 2020. Tracking Co. determines the fair value of the investment at the...

Study smarter with the SolutionInn App


