8 1 point Number of Radioactive Atoms Present Radioactive Decay of Isotope X 1000 0 3...
Fantastic news! We've Found the answer you've been seeking!
Question:
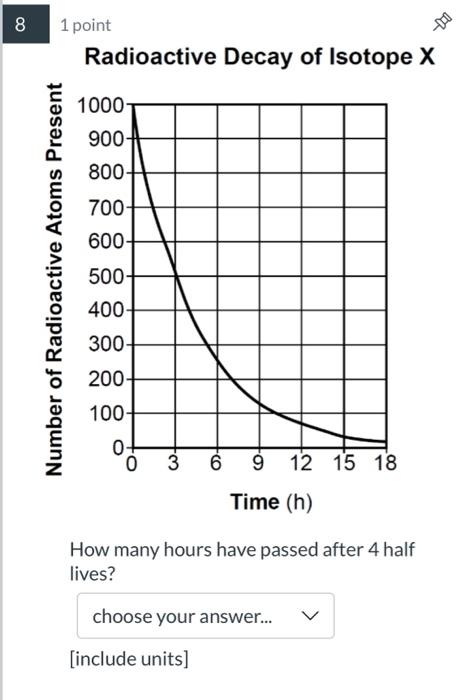
Transcribed Image Text:
8 1 point Number of Radioactive Atoms Present Radioactive Decay of Isotope X 1000 0 3 6 9 12 15 18 Time (h) How many hours have passed after 4 half lives? choose your answer... 900- 800- 700 600- 500- 400- 300- 200 100- 0- [include units] 8 1 point Number of Radioactive Atoms Present Radioactive Decay of Isotope X 1000 0 3 6 9 12 15 18 Time (h) How many hours have passed after 4 half lives? choose your answer... 900- 800- 700 600- 500- 400- 300- 200 100- 0- [include units]
Expert Answer:
Answer rating: 100% (QA)
2 As the at atoms cand at 3 hour radioactive atoons Half lif... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Hair Care is entering the professional hair dryer market. Research shows that their customers wish to pay $301 or less for each dryer. The estimated cost per unit for each dryer is $159 and target...
-
Q5. (6x3 Pts) write the scripts to produce the following matrices: 3 6 9 12 15 18 a) A = equally spaced 6 numbers between 3 and 3 \equally spaced in log scale 6 numbers between 1 and 100/ b) B=(A...
-
How many asymmetric carbon atoms are present in each of the following compounds? (a) (b) (c) H-C-C-C-CI H CI H OH CH CH2OH OH H H OH
-
A company reports the following: Net income ............. $115,000 Preferred dividends .......... $15,000 Shares of common stock outstanding ... 20,000 Market price per share of common stock . $65.00...
-
Bulldog Corporations budgeted balance sheet for the coming year shows total assets of $4,000,000 and total liabilities of $1,900,000. Common stock and retained earnings make up the entire...
-
Outline the main reasons for e-procurement.
-
Do Lab 12 in this chapter's appendix, but for three-stage systems. Operation is at \(293 \mathrm{~K}, 1.0 \mathrm{~atm}, \mathrm{~F}=10 \mathrm{kmol} / \mathrm{h}\) and is \(10 \mathrm{~mol} \%\)...
-
As a financial analyst, you have just been handed the 2012 financial report of Firm A, a large, global pharmaceutical company. Firm A competes in both traditional pharmaceutical products and in...
-
Portsmouth Company makes upholstered furniture. Its only variable cost is direct materials. The demand for the company's products far exceeds its manufacturing capacity. The bottleneck (or...
-
The Fruehauf Corporation (Fruehauf) is engaged in the manufacture of large trucks and industrial vehicles. The Edelman group (Edelman) made a cash tender offer for the shares of Fruehauf for $ 48.50...
-
Number of hours worked in a home care program according to the two clientele of the program and the use of hours worked. Consultation expenses Consultation hours worked PAPA Postop expense category....
-
Colter Steel has $5,250,000 in assets. Temporary current assets Permanent current assets Fixed assets Total assets $ 2,500,000 1,575,000 1,175,000 $ 5,250,000 Short-term rates are 9 percent....
-
19. ABC Limited's management is currently seeking to implement a target costing system. An analysis by the marketing consultant revealed the following: Target selling price $200 Target profit margin...
-
Iron Chemicals acquires a machine that should go through a major overhaul every three years. The total price for the equipment is 1 million. It is estimated that each overhaul will cost 200,000. The...
-
Ducks Cable Inc. sells two internet bundles, Donald and Daisy. The company sells 1 . 5 Donald bundles for every one Daisy bundle. The unit contribution margins for Donald bundles and Daisy bundles...
-
You read in The Wall Street Journal that 30-day T-bills are currently yielding 5.6%. Your brother-in-law, a broker at Safe and Sound Securities, has given you the following estimates of current...
-
Required information The state of stress at a point is x=-2 kpsi, y = 18 kpsi, z= -12 kpsi, Txy= 13 kpsi, Tyz = 2 kpsi, and Tzx= -12 kpsi. NOTE: This is a multi-part question. Once an answer is...
-
31. What is the income that can be received over 15 years from $500,000 earning 6% annually? 32. What is the semiannual payment required to retire $50,000 in debt over 5 years at 8% compounded...
-
Write the formulas for the following ionic compounds: (a) Sodium oxide, (b) Iron sulfide (containing the Fe2+ ion), (c) Cobalt sulfate (containing the Co3+ and SO42- ions), (d) Barium fluoride.
-
Consider the following compounds: Experimentally, phenol is found to be a stronger acid than methanol. Explain this difference in terms of the structures of the conjugate bases. phenol methanol
-
Relative humidity is defined as the ratio (expressed as a percentage) of the partial pressure of water vapor in the air to the equilibrium vapor pressure (see Table 5.3) at a given temperature. On a...
-
Distinguish between a multiple-step and a single-step income statement. Which format results in the higher amount of net income?
-
Refer to the Action feature, Channel Stuffing, on page 19. Suppose that performance bonuses based on revenues were eliminated and this behavior ceases. Would you favor eliminating performance-based...
-
Refer to the Action feature, Palliative Care Unit, on page 5. What type of costs in a hospital might be difficult to split between the PCU and non-PCU areas? What type of costs would be easy to...

Study smarter with the SolutionInn App


