(8 marks) (b) Acetadehyde (0.05kg/s) decomposes homogeneously at 550 oC at 1 atm in a PFR...
Fantastic news! We've Found the answer you've been seeking!
Question:
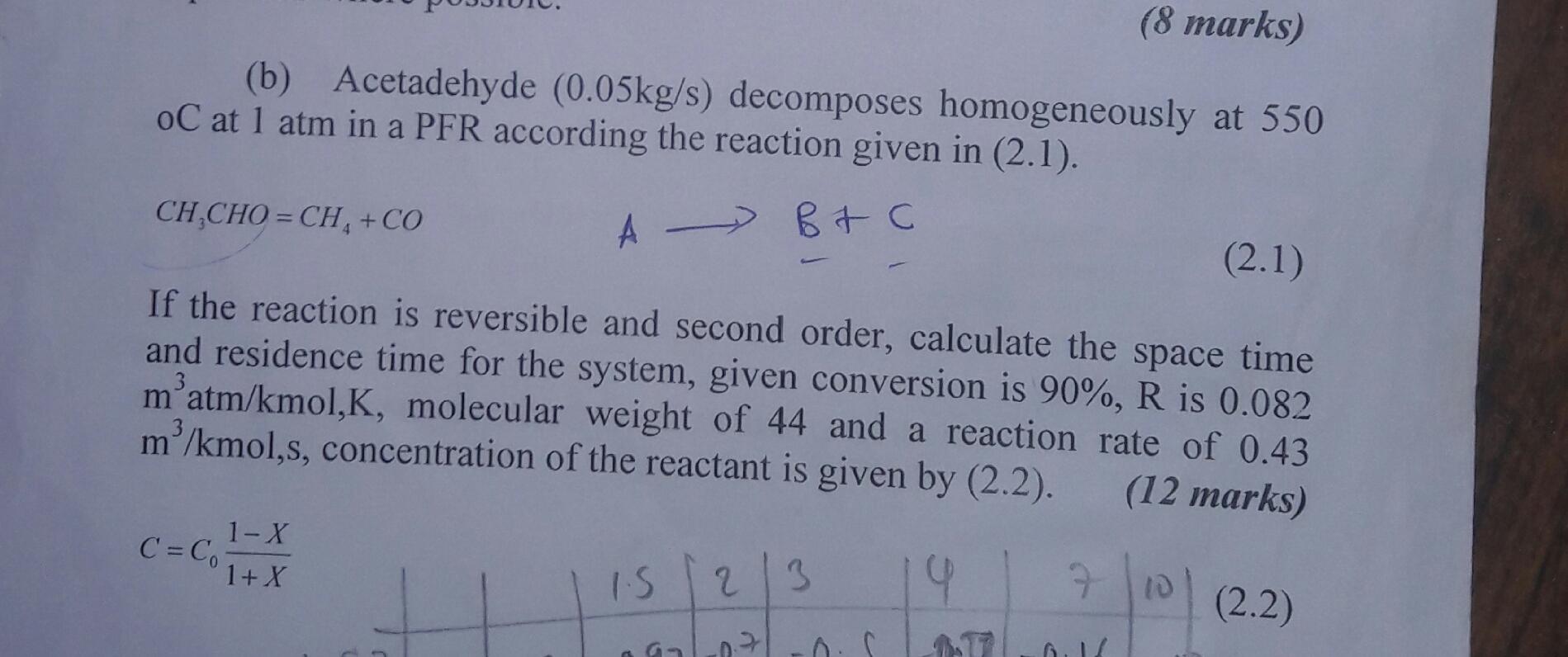
Transcribed Image Text:
(8 marks) (b) Acetadehyde (0.05kg/s) decomposes homogeneously at 550 oC at 1 atm in a PFR according the reaction given in (2.1). CH,CHO = CH, +CO (2.1) If the reaction is reversible and second order, calculate the space time and residence time for the system, given conversion is 90%, R is 0.082 m'atm/kmol,K, molecular weight of 44 and a reaction rate of 0.43 m/kmol,s, concentration of the reactant is given by (2.2). (12 marks) 1-X C = C,- 1+ X 19 710 IS 2. 3 (2.2) (8 marks) (b) Acetadehyde (0.05kg/s) decomposes homogeneously at 550 oC at 1 atm in a PFR according the reaction given in (2.1). CH,CHO = CH, +CO (2.1) If the reaction is reversible and second order, calculate the space time and residence time for the system, given conversion is 90%, R is 0.082 m'atm/kmol,K, molecular weight of 44 and a reaction rate of 0.43 m/kmol,s, concentration of the reactant is given by (2.2). (12 marks) 1-X C = C,- 1+ X 19 710 IS 2. 3 (2.2)
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
A mixture of 1 kmol of CO and 2 kmol of O2 is heated to 2000 K at a pressure of 2 atm. Determine the equilibrium composition of (a) O2, (b) CO2 (in kmols) assuming the mixture consists of CO2, CO and...
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
To load and register a driver, creating a new instance of the Driver class method is better than using the Class.forName() method. (True/False)
-
Arrange substances CCl4, Si, and Ar in order of increasing boiling point?
-
Two identical point charges in free space are connected by a string 7.6 cm long. The tension in the string is 0.21 N. (a) Find the magnitude of the charge on each of the point charges. (b) Using the...
-
The BirthRate variable in the AllCountries dataset gives the birth rate per 1000 people for all the countries in the world. Use technology to create a histogram for this variable, and describe the...
-
A camera of mass 240 g is mounted on a small tripod of mass 200g, assuming that the mass of the camera is uniformly distributed and that the line of action of the weight of the tripod passes through...
-
Single plantwide factory overhead rate Tallulah Inc.'s Machining Department incurred $234,000 of factory overhead cost in producing hoses and valves. The two products consumed a total of 6,000 direct...
-
Antelopes, native to Africa and Asia, range in size from 30 cm to over 180 cm at the shoulder, with most between 90-120 cm. This is related to the humerus length. The data below gives the length and...
-
When Steve Jobs first demonstrated "the pinch"-the two-finger gesture to zoom in and out on photos and Web pages on the iPhone, it just rocked the mobile phone industry-the whole digital world heard...
-
Have you ever been misinterpreted in an email or instant message because the receiver could not see your facial expressions or hear your tone of voice? Describe what happened and how you handled the...
-
How to improve vaccination rates has been a publicized topic over the last few years. There is controversy on the safety and unknown adverse negative externalities. A lack of trust in our government,...
-
What are the potential downsides of authoritarian, democratic, and laissez-faire styles of leadership? When is laissez-faire leadership appropriate? Why? What style of leadership (authoritarian,...
-
What is the maximum capacity utilization rating a company should strive for with no side effects? Explain.
-
If you were the leader of your organization, how would you change the way your organization communicates in order to assure that all employees contribute to the organization's mission, vision, and...
-
The order-to-delivery process for a manufacturing company, Company ABC, can be summarized as follows A quality issue faced by this company is late deliveries to customers. (a) Select any three (3)...
-
An auto-parts manufacturer is considering establishing an engineering computing center. This center will be equipped with three engineering workstations each of which would cost $25,000 and have a...
-
Write a molecular formula for each compound, and indicate the oxidation state of the group 5A element in each formula: (a) Phosphorous acid (b) Pyrophosphoric acid (c) Antimony trichloride (d)...
-
Iridium crystallizes in a face-centered cubic unit cell that has an edge length of 3.833 . (a) Calculate the atomic radius of an iridium atom. (b) Calculate the density of iridium metal?
-
(a) What do ethane and ethanol have in common? (b) How does 1-propanol differ from propane?
-
Using an economic balance sheet, which of the Laws current financial assets is most concerning from an asset allocation perspective? A. Equities B. Real estate C. Fixed income Raye uses a costbenefit...
-
Using the economic balance sheet approach, the Laws economic net worth is closest to: A. $925,000. B. $1,425,000. C. $1,675,000. Raye uses a costbenefit approach to rebalancing and recommends that...
-
Raye believes the previous advisers asset class specifications for equity and derivatives are inappropriate given that, for purposes of asset allocation, asset classes should be: A. diversifying. B....

Study smarter with the SolutionInn App



