-9 A charge of q = 1.60 x 10 C is spread evenly on a thin...
Fantastic news! We've Found the answer you've been seeking!
Question:
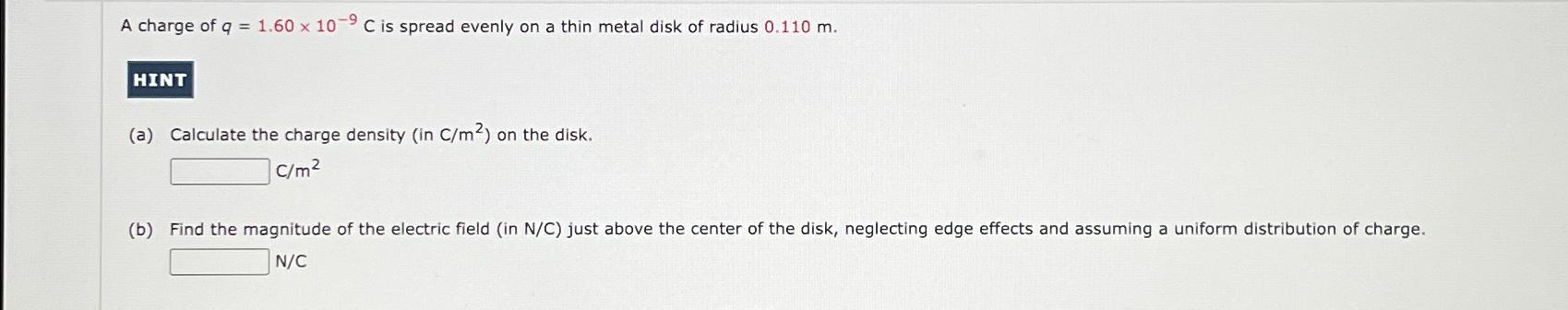
Transcribed Image Text:
-9 A charge of q = 1.60 x 10⁹ C is spread evenly on a thin metal disk of radius 0.110 m. HINT (a) Calculate the charge density (in C/m²) on the disk. C/m² (b) Find the magnitude of the electric field (in N/C) just above the center of the disk, neglecting edge effects and assuming a uniform distribution of charge. N/C -9 A charge of q = 1.60 x 10⁹ C is spread evenly on a thin metal disk of radius 0.110 m. HINT (a) Calculate the charge density (in C/m²) on the disk. C/m² (b) Find the magnitude of the electric field (in N/C) just above the center of the disk, neglecting edge effects and assuming a uniform distribution of charge. N/C
Expert Answer:
Answer rating: 100% (QA)
In the image youve provided youre asked to solve two problems related to the distribution of charge ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
A thin disk with a circular hole at its center, called an annulus, has inner radius R 1 and outer radius R2 (Fig. 21.51). The disk has a uniform positive surface charge density σ on its...
-
On 1 November 2016, Ahmad and Ganeswaran entered into a joint venture for a clothing business. Ganeswaran contributed $7,000 to fund the business, while Ahmad's contribution was $5,000. The...
-
During a worldwide recession in 1983, the oil cartel began to lower prices. Why would a recession make the cartel more vulnerable to price cutting? How would the reduced demand be shared between the...
-
A computer virus destroyed all of your client's files concerning depreciation expense and fixed assets. All other tax records for this manufacturing client remain intact. How should you report...
-
Pick any three customer fulfillment models from Figure 4.9 and: a. Explain how the network operates; b. Identify its advantages; c. Identify its disadvantages; d. Identify the type of market in which...
-
Does Fairmont have any contactor personnel whose have terminated but are being paid through payroll after termination (e.g., ghost employees)?
-
Diane Buswell is preparing the 2013 budget for one of Current Designs rotomolded kayaks. Extensive meetings with members of the sales department and executive team have resulted in the following unit...
-
The unemployment rate among workers under 25 in a populous state went from 8.9% to 6.7% in one year. Assume an average of 1 comma 340 comma 500 workers and estimate the decrease in the number...
-
Skylar and Walter Black have been married for 25 years. They live at 883 Scrub Brush Street, Apt. 52B, Las Vegas, NV 89125. Skylar is a stay-at-home parent and Walt is a high school teacher. His W-2...
-
Mr. Sheridan, bachelor and resident citizen, died leaving the following property: House and lot in San Francisco del Monte Quezon City (Family Home) P3,000,000 Honda Civic car 650,000 Farm in...
-
How does an arbitrator determine that a company had just cause for taking a disciplinary action? What remedy might an arbitrator choose if a company did not have just cause? Will the process be...
-
Meltos Co.is one of the world's largest bakery businesses with a significant market share in many of the world's biggest bakery markets, including many emerging markets. It has a long and proud...
-
How can i describe a business model for my entrepreneurial venture. I am a swim coach and fitness trainer at a fitness center. I am looking to rent a studio a couple hours a week to have my own...
-
Explain the "Separate Procedure" designation as it pertains to certain CPT codes performed during the course of an operation.
-
Discuss the difference and similarity between situational leadership and behavioral leadership Explain in detail
-
SPA Inc are a US based company who have operations throughout Europe and North America. SPA design and install office equipment, all manufacturing is outsourced. The company strategic objective is to...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
A positive point charge (q = +7.2 10-8 C) is surrounded by an equipotential surface A, which has a radius of rA = 1.8 m. A positive test charge (q0 = +4.5 10-11 C) moves from surface A to another...
-
A speeder is pulling directly away and increasing his distance from a police car that is moving at 25 m/s with respect to the ground. The radar gun in the police car emits an electromagnetic wave...
-
A 440.0-Hz tuning fork is sounded together with an out-of-tune guitar string, and a beat frequency of 3 Hz is heard. When the string is tightened, the frequency at which it vibrates increases, and...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App


