9. What is the coordination entity formed when excess of aqueous KCN is added to an...
Fantastic news! We've Found the answer you've been seeking!
Question:
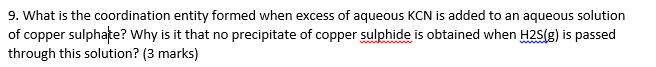
Transcribed Image Text:
9. What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution? (3 marks) 9. What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution? (3 marks)
Expert Answer:
Answer rating: 100% (QA)
Answer P generation F generation RY Ry rY ry RY RRYY Round yellow RRYY Round yellow ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the coordination number of each sphere in (a) A simple cubic cell, (b) A body-centered cubic cell, (c) A face-centered cubic cell? Assume the spheres are all the same?
-
Ammonia and potassium iodide solutions are added to an aqueous solution of Cr(NO 3 ) 3 . A solid is isolated (compound A), and the following data are collected: i. When 0.105 g of compound A was...
-
What is the coordination number of cobalt in the complex [Co(en)(NH 3 )2Cl 2 ]ClO 4 ? a. 2 b. 3 c. 4 d. 5 e. 6
-
Determine if the following strains satisfy the compatibility equations (2.6.2): a. b. c. where A, B and C are constants. Equation 2.6.2 ex=Ay, ey = ez = 0, exy = (Ax+Bz)/2, eyz = Bxz + Cy, ezx = C.x
-
What is the purpose of trading zones? What are some of the most important zones for world trade?
-
Alpha Corporation has outstanding four hundred shares of $100 par value common stock, which has been issued and sold at $105 per share for a total of $42,000. Alpha is incorporated in State X, which...
-
1. Your instructor will divide the class into teams and assign each team the task of investigating the start-up of one of the following businesses: a. Submarine sandwich shop b. Day care service c....
-
Assuming the cost of an associate leaving within 90 days is $3,000, what will be your facility's approximate cost of early turnover for this year? Year-to-Date Turnover Avg. Head- count Total < 90...
-
In the following exercises, use a contour consisting of a long rectangle with one side on the real axis and height 2 for Exercise 6, height for Exercise 7. 7. ex/n dx, n=2, 3,... x ex sinh x dx
-
You have taken out a loan of $19,000 for 4 years with an interest rate of 3% compounded annually. The loan will be repaid by end of year payments. Fill in all the boxes below, and round all entries...
-
What's the number of weights for a network with 6 input nodes, 4hidden nodes (one hidden layer), and 2 output nodes? (PS: Theanswer is not 32. I got this wrong.) 1 answer
-
D Talbingo operates as a landscape gardener and has provided the following account balances (see figure 9.68). For this service business you are required to: write next to each account name the...
-
From the account listing of Dr R Wollondilly, whose service business operates as Riverside Medical Practice (see figure 9.67), you are required to: write next to each account name the...
-
P Indian uses perpetual inventory and during the year carries out physical stocktakes of certain inventory items. During the year ended 30 June 2022, P Indian carries out two partial physical...
-
Global Enterprises Corporation zealously advocates the election of Courtney Smith as the next president of the United States and the defeat of the incumbent, Herbert Dumpty. The corporation produces...
-
I Pacific keeps inventory using the perpetual inventory system and carries out a rotational stock count of a specific portion of the inventory every three months, as well as the full end-of-year...
-
A cylinder of radius r, height h, and mass m floats with its axis vertical in a liquid of density pas shown in the following figure. Archimedes' Principle: An object partially or totally submerged in...
-
Show that if A is any m n matrix, then Im A = A and AIn = A.
-
(a) What is the frequency of light having a wavelength of 456 nm? (b) What is the wavelength (in nanometers) of radiation having a frequency of 2.45 109 Hz? (This is the type of radiation used in...
-
At STP, 0.280 L of a gas weighs 0.400 g. Calculate the molar mass of the gas.
-
Because of its detrimental effect on the environment, the lead compound described in Problem 3.138 has been replaced in recent years by methyl tert -butyl ether (a compound of C, H, and O) to enhance...
-
A probability experiment consists of rolling a single fair die. (a) Identify the outcomes of the probability experiment. (b) Determine the sample space. (c) Define the event E = roll an even number....
-
A pair of fair dice is rolled. Fair die are die where each outcome is equally likely. (a) Compute the probability of rolling a seven. (b) Compute the probability of rolling snake eyes; that is,...
-
Our number system consists of the digits 0, 1, 2, 3, 4, 5, 6, 7, 8, and 9. Because we do not write numbers such as 12 as 012, the first significant digit in any number must be 1, 2, 3, 4, 5, 6, 7, 8,...

Study smarter with the SolutionInn App


