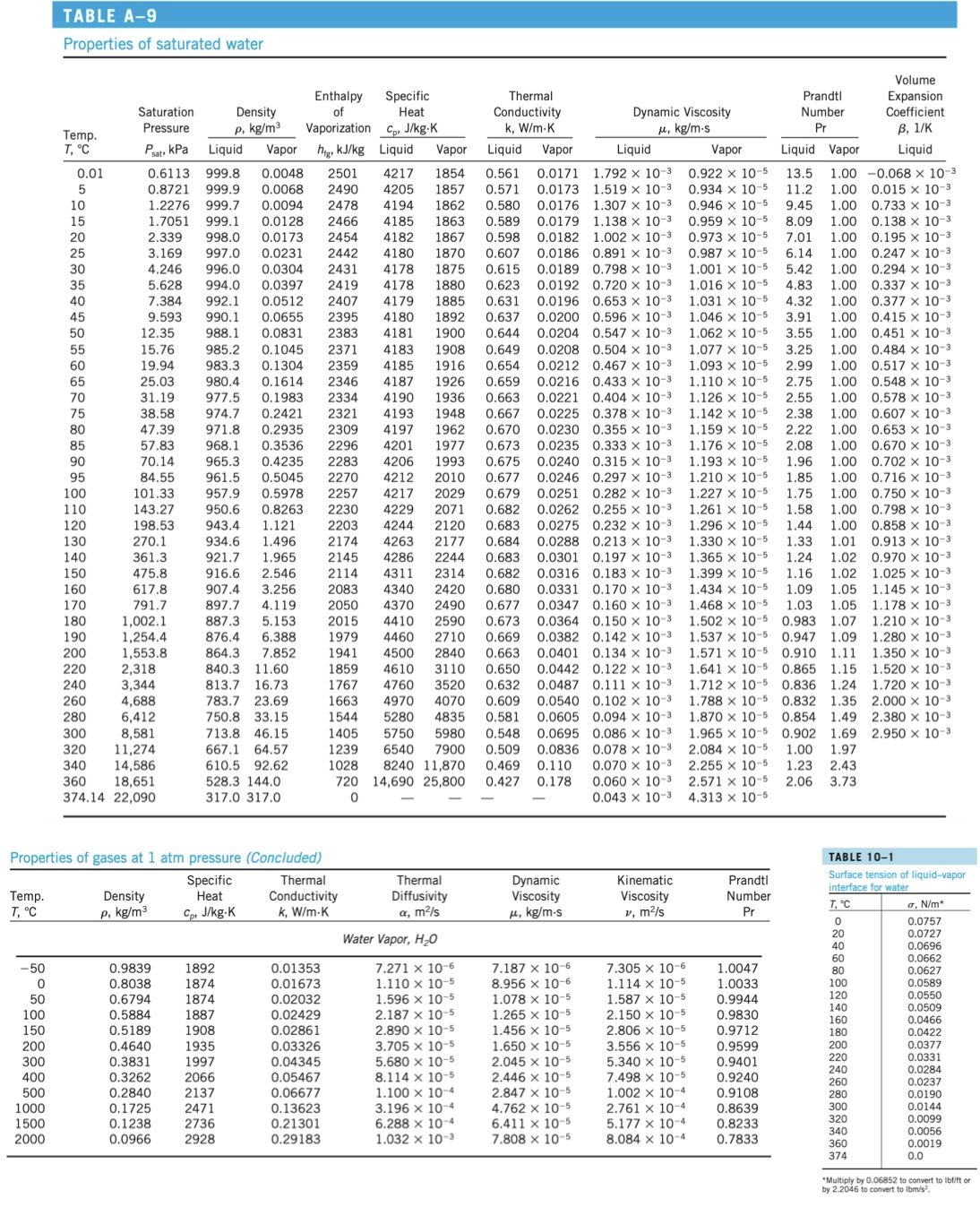
A 0.1-meter diameter steel sphere is heated to a temperature of 700C, then immersed in water...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
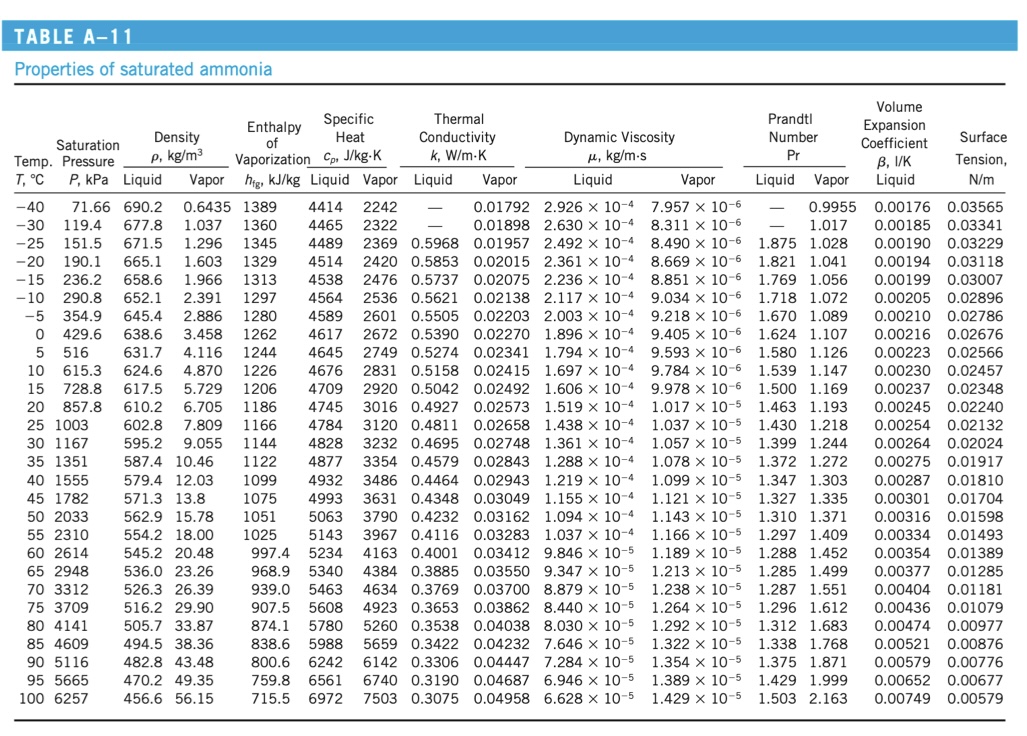
A 0.1-meter diameter steel sphere is heated to a temperature of 700C, then immersed in water at atmospheric pressure. Find the heat flux at the instant the sphere is dropped into the water. Emissivity is .3. Temp. T, C -50 0 50 100 150 200 300 400 500 TABLE A-9 Properties of saturated water 1000 1500 2000 Temp. T, C 0.01 5 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 110 120 130 140 150 160 170 180 190 200 38.58 47.39 57.83 70.14 84.55 101.33 143.27 198.53 270.1 361.3 475.8 617.8 791.7 1,002.1 1,254.4 1,553.8 2,318 3,344 4,688 6,412 8,581 11,274 14,586 18,651 374.14 22,090 Enthalpy of Vaporization C, J/kg-K 12.35 15.76 19.94 983.3 980.4 0.1614 977.5 0.1983 974.7 0.2421 2321 4193 1948 4197 1962 971.8 0.2935 2309 968.1 0.3536 2296 965.3 0.4235 2283 961.5 0.5045 2270 4201 1977 4206 1993 0.675 Liquid Vapor higkJ/kg Liquid Vapor Liquid Vapor Liquid 0.6113 999.8 0.0048 2501 4217 1854 0.561 0.0171 1.792 x 10-3 0.8721 999.9 0.0068 2490 4205 1857 0.571 0.0173 1.519 x 10-3 1.2276 999.7 0.0094 2478 4194 1862 0.580 0.0176 1.307 x 10-3 1.7051 999.1 0.0128 2466 4185 1863 0.589 0.0179 1.138 x 10-3 2.339 998.0 0.0173 2454 4182 1867 0.598 0.0182 1.002 x 10- 3.169 997.0 0.0231 2442 4180 1870 0.607 0.0186 0.891 x 10-3 4.246 996.0 0.0304 2431 4178 1875 0.615 0.0189 0.798 x 10-3 5.628 994.0 0.0397 2419 4178 1880 0.623 0.0192 0.720 x 10- 7.384 992.1 0.0512 2407 4179 1885 0.631 0.0196 0.653 x 10-3 9.593 990.1 0.0655 2395 4180 1892 0.637 0.0200 0.596 x 10-3 988.1 0.0831 2383 4181 1900 0.644 0.0204 0.547 x 10-3 985.2 0.1045 2371 4183 1908 0.649 0.0208 0.504 x 10-3 0.1304 2359 4185 1916 0.654 0.0212 0.467 x 10- 2346 4187 1926 0.659 0.0216 0.433 x 10-3 2334 4190 1936 0.663 0.0221 0.404 x 10-3 0.667 0.0225 0.378 x 10-3 0.670 0.0230 0.355 x 10-3 0.673 0.0235 0.333 x 10- 0.0240 0.315 x 10-3 0.677 0.0246 0.297 x 10-3 2029 0.679 0.0251 0.282 x 10- 2071 0.682 0.0262 0.255 x 10-3 2120 0.683 0.0275 0.232 x 10- 2177 0.684 0.0288 0.213 x 10-3 2244 0.683 0.0301 0.197 x 10-3 4311 2314 0.682 0.0316 0.183 x 10-3 0.680 0.0331 0.170 x 10-3 4370 2490 0.677 0.0347 0.160 x 10-3 4410 2590 0.673 0.0364 0.150 x 10-3 4460 2710 0.669 0.0382 0.142 x 10-3 4500 2840 0.663 0.0401 0.134 x 10-3. 4610 3110 0.650 0.0442 0.122 x 10-3 4760 3520 0.632 0.0487 0.111 x 10-3 4970 4070 0.609 0.0540 0.102 x 10-3 5280 4835 0.581 0.0605 0.094 x 10-3 5750 5980 0.548 0.0695 0.086 x 10-3 6540 7900 0.509 0.0836 0.078 x 10-3 8240 11,870 0.469 0.110 0.070 x 10-3 14,690 25,800 0.427 0.178 0.060 x 10-3 0.043 x 10-3 4212 2010 957.9 0.5978 2257 4217 950.6 0.8263 2230 4229 943.4 1.121 4244 934.6 1.496 4263 921.7 1.965 4286 916.6 2.546 907.4 3.256 4340 2420 897.7 4.119 887.3 5.153 876.4 6.388 864.3 7.852 840.3 11.60 220 240 260 280 300 320 340 360 Saturation Pressure Psat, kPa 25.03 31.19 Properties of gases at 1 atm pressure (Concluded) Specific Heat Cp J/kg-K Density p. kg/m 0.9839 0.8038 0.6794 0.5884 0.5189 0.4640 0.3831 0.3262 0.2840 0.1725 0.1238 0.0966 813.7 16.73 783.7 23.69 750.8 33.15 Density p, kg/m 713.8 46.15 667.1 64.57 610.5 92.62 528.3 144.0 317.0 317.0 1892 1874 1874 1887 1908 1935 1997 2066 2137 2471 2736 2928 2203 2174 2145 2114 2083 2050 2015 1979 1941 1859 1767 1663 1544 1405 1239 1028 720 0 Thermal Conductivity k, W/m-K 0.01353 0.01673 0.02032 0.02429 0.02861 0.03326 0.04345 0.05467 0.06677 0.13623 0.21301 0.29183 Specific Heat Thermal Diffusivity , m/s Water Vapor, HO 7.271 x 10-6 1.110 x 10-5 1.596 x 10-5 2.187 x 10-5 2.890 x 10-5 3.705 x 10-5 5.680 x 10-5 8.114 x 10-5 1.100 x 10-4 3.196 x 10-4 6.288 x 10-4 1.032 x 10-3 Thermal Conductivity k, W/m-K Dynamic Viscosity , kg/m-s 7.187 x 10-6 8.956 x 10-6 1.078 x 10-5 1.265 x 10-5 1.456 x 10-5 1.650 x 10-5 2.045 x 10-5 2.446 x 10-5 2.847 x 10-5 4.762 x 10-5 6.411 x 10-5 7.808 x 10-5 Dynamic Viscosity , kg/m-s Kinematic Viscosity v, m/s 7.305 x 10-6 1.114 x 10-5 1.587 x 10-5 2.150 x 10-5 2.806 x 10-5 3.556 x 10-5 5.340 x 10-5 7.498 x 10-5 1.002 x 10-4 2.761 x 10-4 5.177 x 10-4 8.084 x 10-4 Vapor 0.922 x 10-5 0.934 x 10-5 0.946 x 10-5 0.959 x 10-5 0.973 x 10-5 0.987 x 10-5 1.001 x 10-5 1.016 x 10-5 1.031 x 10-5 1.046 x 10-5 1.062 x 10-5 1.077 x 10-5 1.093 x 10-5 1.110 x 10-5 1.126 x 10-5 1.142 x 10-5 1.159 x 10-5 1.176 x 10-5 1.193 x 10-5 1.210 x 10-5 1.227 x 10-5 1.261 x 10-5 1.296 x 10-5 1.330 x 10-5 1.365 x 10-5 1.399 x 10-5 1.434 x 10-5 1.468 x 10-5 1.502 x 10-5 1.537 x 10-5 1.571 x 10-5 1.641 x 10-5 1.712 x 10-5 1.788 x 10- 1.870 X 10-5 1.965 x 10-5 2.084 x 10-5 2.255 x 10-5 2.571 x 10-5 4.313 x 10-5 Prandtl Number Pr 1.0047 1.0033 0.9944 0.9830 0.9712 0.9599 0.9401 0.9240 0.9108 0.8639 0.8233 0.7833 Volume Expansion. Coefficient B, 1/K Liquid Liquid Vapor 13.5 1.00 -0.068 x 10-3 11.2 1.00 0.015 x 10- 9.45 1.00 0.733 x 10-3 8.09 1.00 7.01 1.00 1.00 0.138 x 10-3 0.195 x 10-3 0.247 x 10-3 1.00 0.294 x 10-3 6.14 5.42 4.83 4.32 3.91 1.00 0.337 x 10-3 1.00 0.377 x 10-3 1.00 3.55 3.25 2.99 1.00 1.00 1.00 2.75 1.00 2.55 1.00 1.00 Prandtl Number Pr 2.22 2.08 1.96 1.00 1.85 1.00 1.75 1.00 1.58 1.00 1.44 1.00 1.33 1.01 1.24 1.02 1.02 1.16 1.09 1.05 1.03 1.05 0.983 1.07 0.947 1.09 0.910 1.11 0.865 1.15 0.836 1.24 0.832 1.35 0.854 1.49 1.69 1.97 2.43 3.73 1.00 1.00 0.902 1.00 1.23 2.06 0.415 x 10-3 0.451 x 10-3 0.484 x 10-3 0.517 x 10-3 0.548 x 10-3 0.578 x 10-3 200 220 240 260 280 300 320 340 360 374 0.607 x 10-3 0.653 x 10-3 0.670 x 10-3 0.702 x 10-3 0.716 x 10-3 0.750 x 10-3 0.798 x 10-3 0.858 x 10-3 0.913 x 10-3 0.970 x 10-3 1.025 x 10-3 1.145 x 10-3 1.178 x 10-3 1.210 x 10-3 1.280 x 10-3 1.350 x 10-3 1.520 x 10-3 1.720 x 10-3 2.000 x 10-3 2.380 x 10-3 2.950 x 10-3 TABLE 10-1 Surface tension of liquid-vapor interface for water T, C 0 20 40 60 80 100 120 140 160 180 a, N/m* 0.0757 0.0727 0.0696 0.0662 0.0627 0.0589 0.0550 0.0509 0.0466 0.0422 0.0377 0.0331 0.0284 0.0237 0.0190 0.0144 0.0099 0.0056 0.0019 0.0 *Multiply by 0.06852 to convert to lbf/ft or by 2.2046 to convert to lbm/s. TABLE A-11 Properties of saturated ammonia Saturation Temp. Pressure T, C Enthalpy of Vaporization Cp, J/kg-K hig, kJ/kg Liquid Vapor Liquid Vapor -40 , kg/m-s Liquid 2.926 x 10-4 2.630 x 10-4 2.492 x 10-4 2.361 x 10-4 2.236 x 10-4 2.117 x 10-4 2.003 x 10-4 1.896 x 10-4 1.794 x 10-4 1.697 x 10-4 1.606 x 10-4 1.519 x 10-4 1.438 x 10- 1.361 x 10-4 1.288 x 10-4 1.219 x 10-4 1.155 x 10-4 0 5 71.66 690.2 0.6435 1389 4414 2242 0.01792 -30 119.4 677.8 1.037 1360 4465 2322 0.01898 -25 151.5 671.5 1.296 1345 4489 2369 0.5968 0.01957 -20 190.1 665.1 1.603 1329 4514 2420 0.5853 0.02015 -15 236.2 658.6 1.966 1313 4538 2476 0.5737 0.02075 -10 290.8 652.1 2.391 1297 4564 2536 0.5621 0.02138 -5 354.9 645.4 2.886 1280 4589 2601 0.5505 0.02203 429.6 638.6 3.458 1262 4617 2672 0.5390 0.02270 631.7 4.116 1244 4645 2749 0.5274 0.02341 10 615.3 624.6 4.870 1226 4676 2831 0.5158 0.02415 15 728.8 617.5 5.729 1206 4709 2920 0.5042 0.02492 20 857.8 610.2 6.705 1186 4745 3016 0.4927 0.02573 25 1003 602.8 7.809 1166 4784 3120 0.4811 0.02658 30 1167 595.2 9.055 1144 4828 3232 0.4695 0.02748 35 1351 587.4 10.46 1122 4877 3354 0.4579 0.02843 40 1555 4932 45 1782 50 2033 55 2310 60 2614 65 2948 70 3312 75 3709 80 4141 85 4609 90 5116 95 5665 100 6257 516 1099 3486 0.4464 0.02943 1075 4993 3631 0.4348 0.03049 5063 3790 0.4232 0.03162 1051 1025 5143 997.4 5234 968.9 5340 4384 0.3885 0.03550 939.0 5463 4634 0.3769 0.03700 907.5 5608 4923 0.3653 0.03862 874.1 5780 5260 0.3538 0.04038 838.6 5988 5659 0.3422 0.04232 800.6 6242 759.8 6561 715.5 6972 7503 0.3075 1.094 x 10-4 3967 0.4116 0.03283 1.037 x 10-4 4163 0.4001 0.03412 9.846 x 10-5 9.347 x 10-5 8.879 x 10-5 8.440 x 10-5 8.030 x 10-5 7.646 x 10-5 7.284 x 10-5 6142 0.3306 0.04447 6740 0.3190 0.04687 6.946 x 10-5 0.04958 6.628 x 10-5 Density p, kg/m P, kPa Liquid Vapor Specific Heat 579.4 12.03 571.3 13.8 562.9 15.78 554.2 18.00 545.2 20.48 536.0 23.26 526.3 26.39 516.2 29.90 505.7 33.87 494.5 38.36 482.8 43.48 470.2 49.35 456.6 56.15 Thermal Conductivity k, W/m-K Dynamic Viscosity Vapor 7.957 x 10-6 8.311 x 10-6 8.490 x 10-6 8.669 x 10-6 8.851 x 10-6 9.034 x 10-6 9.218 x 10-6 9.405 x 10-6 9.593 x 10-6 9.784 x 10-6 9.978 x 10-6 1.017 x 10-5 1.037 x 10-5 1.057 x 10-5 1.078 x 10-5 1.099 x 10-5 1.121 x 10-5 1.143 x 10-5 1.166 x 10-5 1.189 x 10-5 1.213 x 10-5 1.238 x 10-5 1.264 x 10-5 1.292 x 10-5 1.322 x 10-5 1.354 x 10-5 1.389 x 10-5 1.429 x 10-5 Prandtl Number Pr Liquid Vapor 0.9955 1.017 1.875 1.028 1.821 1.041 1.769 1.056 1.718 1.072 1.670 1.089 1.624 1.107 1.580 1.126 1.539 1.147 1.500 1.169 1.463 1.193 1.430 1.218 1.399 1.244 1.372 1.272 1.347 1.303 1.327 1.335 1.310 1.371 1.297 1.409 1.288 1.452 1.285 1.499 1.287 1.551 1.296 1.612 1.312 1.683 1.338 1.768 1.375 1.871 1.429 1.999 1.503 2.163 Volume Expansion Coefficient B, I/K Liquid Surface Tension, N/m 0.00176 0.03565 0.00185 0.03341 0.00190 0.03229 0.00194 0.03118 0.00199 0.03007 0.00205 0.02896 0.00210 0.02786 0.00216 0.02676 0.00223 0.02566 0.00230 0.02457 0.00237 0.02348 0.00245 0.02240 0.00254 0.02132 0.00264 0.02024 0.00275 0.01917 0.00287 0.01810 0.00301 0.01704 0.00316 0.01598 0.00334 0.01493 0.00354 0.01389 0.00377 0.01285 0.00404 0.01181 0.00436 0.01079 0.00474 0.00977 0.00521 0.00876 0.00579 0.00776 0.00652 0.00677 0.00749 0.00579 A 0.1-meter diameter steel sphere is heated to a temperature of 700C, then immersed in water at atmospheric pressure. Find the heat flux at the instant the sphere is dropped into the water. Emissivity is .3. Temp. T, C -50 0 50 100 150 200 300 400 500 TABLE A-9 Properties of saturated water 1000 1500 2000 Temp. T, C 0.01 5 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100 110 120 130 140 150 160 170 180 190 200 38.58 47.39 57.83 70.14 84.55 101.33 143.27 198.53 270.1 361.3 475.8 617.8 791.7 1,002.1 1,254.4 1,553.8 2,318 3,344 4,688 6,412 8,581 11,274 14,586 18,651 374.14 22,090 Enthalpy of Vaporization C, J/kg-K 12.35 15.76 19.94 983.3 980.4 0.1614 977.5 0.1983 974.7 0.2421 2321 4193 1948 4197 1962 971.8 0.2935 2309 968.1 0.3536 2296 965.3 0.4235 2283 961.5 0.5045 2270 4201 1977 4206 1993 0.675 Liquid Vapor higkJ/kg Liquid Vapor Liquid Vapor Liquid 0.6113 999.8 0.0048 2501 4217 1854 0.561 0.0171 1.792 x 10-3 0.8721 999.9 0.0068 2490 4205 1857 0.571 0.0173 1.519 x 10-3 1.2276 999.7 0.0094 2478 4194 1862 0.580 0.0176 1.307 x 10-3 1.7051 999.1 0.0128 2466 4185 1863 0.589 0.0179 1.138 x 10-3 2.339 998.0 0.0173 2454 4182 1867 0.598 0.0182 1.002 x 10- 3.169 997.0 0.0231 2442 4180 1870 0.607 0.0186 0.891 x 10-3 4.246 996.0 0.0304 2431 4178 1875 0.615 0.0189 0.798 x 10-3 5.628 994.0 0.0397 2419 4178 1880 0.623 0.0192 0.720 x 10- 7.384 992.1 0.0512 2407 4179 1885 0.631 0.0196 0.653 x 10-3 9.593 990.1 0.0655 2395 4180 1892 0.637 0.0200 0.596 x 10-3 988.1 0.0831 2383 4181 1900 0.644 0.0204 0.547 x 10-3 985.2 0.1045 2371 4183 1908 0.649 0.0208 0.504 x 10-3 0.1304 2359 4185 1916 0.654 0.0212 0.467 x 10- 2346 4187 1926 0.659 0.0216 0.433 x 10-3 2334 4190 1936 0.663 0.0221 0.404 x 10-3 0.667 0.0225 0.378 x 10-3 0.670 0.0230 0.355 x 10-3 0.673 0.0235 0.333 x 10- 0.0240 0.315 x 10-3 0.677 0.0246 0.297 x 10-3 2029 0.679 0.0251 0.282 x 10- 2071 0.682 0.0262 0.255 x 10-3 2120 0.683 0.0275 0.232 x 10- 2177 0.684 0.0288 0.213 x 10-3 2244 0.683 0.0301 0.197 x 10-3 4311 2314 0.682 0.0316 0.183 x 10-3 0.680 0.0331 0.170 x 10-3 4370 2490 0.677 0.0347 0.160 x 10-3 4410 2590 0.673 0.0364 0.150 x 10-3 4460 2710 0.669 0.0382 0.142 x 10-3 4500 2840 0.663 0.0401 0.134 x 10-3. 4610 3110 0.650 0.0442 0.122 x 10-3 4760 3520 0.632 0.0487 0.111 x 10-3 4970 4070 0.609 0.0540 0.102 x 10-3 5280 4835 0.581 0.0605 0.094 x 10-3 5750 5980 0.548 0.0695 0.086 x 10-3 6540 7900 0.509 0.0836 0.078 x 10-3 8240 11,870 0.469 0.110 0.070 x 10-3 14,690 25,800 0.427 0.178 0.060 x 10-3 0.043 x 10-3 4212 2010 957.9 0.5978 2257 4217 950.6 0.8263 2230 4229 943.4 1.121 4244 934.6 1.496 4263 921.7 1.965 4286 916.6 2.546 907.4 3.256 4340 2420 897.7 4.119 887.3 5.153 876.4 6.388 864.3 7.852 840.3 11.60 220 240 260 280 300 320 340 360 Saturation Pressure Psat, kPa 25.03 31.19 Properties of gases at 1 atm pressure (Concluded) Specific Heat Cp J/kg-K Density p. kg/m 0.9839 0.8038 0.6794 0.5884 0.5189 0.4640 0.3831 0.3262 0.2840 0.1725 0.1238 0.0966 813.7 16.73 783.7 23.69 750.8 33.15 Density p, kg/m 713.8 46.15 667.1 64.57 610.5 92.62 528.3 144.0 317.0 317.0 1892 1874 1874 1887 1908 1935 1997 2066 2137 2471 2736 2928 2203 2174 2145 2114 2083 2050 2015 1979 1941 1859 1767 1663 1544 1405 1239 1028 720 0 Thermal Conductivity k, W/m-K 0.01353 0.01673 0.02032 0.02429 0.02861 0.03326 0.04345 0.05467 0.06677 0.13623 0.21301 0.29183 Specific Heat Thermal Diffusivity , m/s Water Vapor, HO 7.271 x 10-6 1.110 x 10-5 1.596 x 10-5 2.187 x 10-5 2.890 x 10-5 3.705 x 10-5 5.680 x 10-5 8.114 x 10-5 1.100 x 10-4 3.196 x 10-4 6.288 x 10-4 1.032 x 10-3 Thermal Conductivity k, W/m-K Dynamic Viscosity , kg/m-s 7.187 x 10-6 8.956 x 10-6 1.078 x 10-5 1.265 x 10-5 1.456 x 10-5 1.650 x 10-5 2.045 x 10-5 2.446 x 10-5 2.847 x 10-5 4.762 x 10-5 6.411 x 10-5 7.808 x 10-5 Dynamic Viscosity , kg/m-s Kinematic Viscosity v, m/s 7.305 x 10-6 1.114 x 10-5 1.587 x 10-5 2.150 x 10-5 2.806 x 10-5 3.556 x 10-5 5.340 x 10-5 7.498 x 10-5 1.002 x 10-4 2.761 x 10-4 5.177 x 10-4 8.084 x 10-4 Vapor 0.922 x 10-5 0.934 x 10-5 0.946 x 10-5 0.959 x 10-5 0.973 x 10-5 0.987 x 10-5 1.001 x 10-5 1.016 x 10-5 1.031 x 10-5 1.046 x 10-5 1.062 x 10-5 1.077 x 10-5 1.093 x 10-5 1.110 x 10-5 1.126 x 10-5 1.142 x 10-5 1.159 x 10-5 1.176 x 10-5 1.193 x 10-5 1.210 x 10-5 1.227 x 10-5 1.261 x 10-5 1.296 x 10-5 1.330 x 10-5 1.365 x 10-5 1.399 x 10-5 1.434 x 10-5 1.468 x 10-5 1.502 x 10-5 1.537 x 10-5 1.571 x 10-5 1.641 x 10-5 1.712 x 10-5 1.788 x 10- 1.870 X 10-5 1.965 x 10-5 2.084 x 10-5 2.255 x 10-5 2.571 x 10-5 4.313 x 10-5 Prandtl Number Pr 1.0047 1.0033 0.9944 0.9830 0.9712 0.9599 0.9401 0.9240 0.9108 0.8639 0.8233 0.7833 Volume Expansion. Coefficient B, 1/K Liquid Liquid Vapor 13.5 1.00 -0.068 x 10-3 11.2 1.00 0.015 x 10- 9.45 1.00 0.733 x 10-3 8.09 1.00 7.01 1.00 1.00 0.138 x 10-3 0.195 x 10-3 0.247 x 10-3 1.00 0.294 x 10-3 6.14 5.42 4.83 4.32 3.91 1.00 0.337 x 10-3 1.00 0.377 x 10-3 1.00 3.55 3.25 2.99 1.00 1.00 1.00 2.75 1.00 2.55 1.00 1.00 Prandtl Number Pr 2.22 2.08 1.96 1.00 1.85 1.00 1.75 1.00 1.58 1.00 1.44 1.00 1.33 1.01 1.24 1.02 1.02 1.16 1.09 1.05 1.03 1.05 0.983 1.07 0.947 1.09 0.910 1.11 0.865 1.15 0.836 1.24 0.832 1.35 0.854 1.49 1.69 1.97 2.43 3.73 1.00 1.00 0.902 1.00 1.23 2.06 0.415 x 10-3 0.451 x 10-3 0.484 x 10-3 0.517 x 10-3 0.548 x 10-3 0.578 x 10-3 200 220 240 260 280 300 320 340 360 374 0.607 x 10-3 0.653 x 10-3 0.670 x 10-3 0.702 x 10-3 0.716 x 10-3 0.750 x 10-3 0.798 x 10-3 0.858 x 10-3 0.913 x 10-3 0.970 x 10-3 1.025 x 10-3 1.145 x 10-3 1.178 x 10-3 1.210 x 10-3 1.280 x 10-3 1.350 x 10-3 1.520 x 10-3 1.720 x 10-3 2.000 x 10-3 2.380 x 10-3 2.950 x 10-3 TABLE 10-1 Surface tension of liquid-vapor interface for water T, C 0 20 40 60 80 100 120 140 160 180 a, N/m* 0.0757 0.0727 0.0696 0.0662 0.0627 0.0589 0.0550 0.0509 0.0466 0.0422 0.0377 0.0331 0.0284 0.0237 0.0190 0.0144 0.0099 0.0056 0.0019 0.0 *Multiply by 0.06852 to convert to lbf/ft or by 2.2046 to convert to lbm/s. TABLE A-11 Properties of saturated ammonia Saturation Temp. Pressure T, C Enthalpy of Vaporization Cp, J/kg-K hig, kJ/kg Liquid Vapor Liquid Vapor -40 , kg/m-s Liquid 2.926 x 10-4 2.630 x 10-4 2.492 x 10-4 2.361 x 10-4 2.236 x 10-4 2.117 x 10-4 2.003 x 10-4 1.896 x 10-4 1.794 x 10-4 1.697 x 10-4 1.606 x 10-4 1.519 x 10-4 1.438 x 10- 1.361 x 10-4 1.288 x 10-4 1.219 x 10-4 1.155 x 10-4 0 5 71.66 690.2 0.6435 1389 4414 2242 0.01792 -30 119.4 677.8 1.037 1360 4465 2322 0.01898 -25 151.5 671.5 1.296 1345 4489 2369 0.5968 0.01957 -20 190.1 665.1 1.603 1329 4514 2420 0.5853 0.02015 -15 236.2 658.6 1.966 1313 4538 2476 0.5737 0.02075 -10 290.8 652.1 2.391 1297 4564 2536 0.5621 0.02138 -5 354.9 645.4 2.886 1280 4589 2601 0.5505 0.02203 429.6 638.6 3.458 1262 4617 2672 0.5390 0.02270 631.7 4.116 1244 4645 2749 0.5274 0.02341 10 615.3 624.6 4.870 1226 4676 2831 0.5158 0.02415 15 728.8 617.5 5.729 1206 4709 2920 0.5042 0.02492 20 857.8 610.2 6.705 1186 4745 3016 0.4927 0.02573 25 1003 602.8 7.809 1166 4784 3120 0.4811 0.02658 30 1167 595.2 9.055 1144 4828 3232 0.4695 0.02748 35 1351 587.4 10.46 1122 4877 3354 0.4579 0.02843 40 1555 4932 45 1782 50 2033 55 2310 60 2614 65 2948 70 3312 75 3709 80 4141 85 4609 90 5116 95 5665 100 6257 516 1099 3486 0.4464 0.02943 1075 4993 3631 0.4348 0.03049 5063 3790 0.4232 0.03162 1051 1025 5143 997.4 5234 968.9 5340 4384 0.3885 0.03550 939.0 5463 4634 0.3769 0.03700 907.5 5608 4923 0.3653 0.03862 874.1 5780 5260 0.3538 0.04038 838.6 5988 5659 0.3422 0.04232 800.6 6242 759.8 6561 715.5 6972 7503 0.3075 1.094 x 10-4 3967 0.4116 0.03283 1.037 x 10-4 4163 0.4001 0.03412 9.846 x 10-5 9.347 x 10-5 8.879 x 10-5 8.440 x 10-5 8.030 x 10-5 7.646 x 10-5 7.284 x 10-5 6142 0.3306 0.04447 6740 0.3190 0.04687 6.946 x 10-5 0.04958 6.628 x 10-5 Density p, kg/m P, kPa Liquid Vapor Specific Heat 579.4 12.03 571.3 13.8 562.9 15.78 554.2 18.00 545.2 20.48 536.0 23.26 526.3 26.39 516.2 29.90 505.7 33.87 494.5 38.36 482.8 43.48 470.2 49.35 456.6 56.15 Thermal Conductivity k, W/m-K Dynamic Viscosity Vapor 7.957 x 10-6 8.311 x 10-6 8.490 x 10-6 8.669 x 10-6 8.851 x 10-6 9.034 x 10-6 9.218 x 10-6 9.405 x 10-6 9.593 x 10-6 9.784 x 10-6 9.978 x 10-6 1.017 x 10-5 1.037 x 10-5 1.057 x 10-5 1.078 x 10-5 1.099 x 10-5 1.121 x 10-5 1.143 x 10-5 1.166 x 10-5 1.189 x 10-5 1.213 x 10-5 1.238 x 10-5 1.264 x 10-5 1.292 x 10-5 1.322 x 10-5 1.354 x 10-5 1.389 x 10-5 1.429 x 10-5 Prandtl Number Pr Liquid Vapor 0.9955 1.017 1.875 1.028 1.821 1.041 1.769 1.056 1.718 1.072 1.670 1.089 1.624 1.107 1.580 1.126 1.539 1.147 1.500 1.169 1.463 1.193 1.430 1.218 1.399 1.244 1.372 1.272 1.347 1.303 1.327 1.335 1.310 1.371 1.297 1.409 1.288 1.452 1.285 1.499 1.287 1.551 1.296 1.612 1.312 1.683 1.338 1.768 1.375 1.871 1.429 1.999 1.503 2.163 Volume Expansion Coefficient B, I/K Liquid Surface Tension, N/m 0.00176 0.03565 0.00185 0.03341 0.00190 0.03229 0.00194 0.03118 0.00199 0.03007 0.00205 0.02896 0.00210 0.02786 0.00216 0.02676 0.00223 0.02566 0.00230 0.02457 0.00237 0.02348 0.00245 0.02240 0.00254 0.02132 0.00264 0.02024 0.00275 0.01917 0.00287 0.01810 0.00301 0.01704 0.00316 0.01598 0.00334 0.01493 0.00354 0.01389 0.00377 0.01285 0.00404 0.01181 0.00436 0.01079 0.00474 0.00977 0.00521 0.00876 0.00579 0.00776 0.00652 0.00677 0.00749 0.00579
Expert Answer:

Related Book For 

Fundamentals of Heat and Mass Transfer
ISBN: 978-0471457282
6th Edition
Authors: Incropera, Dewitt, Bergman, Lavine
Posted Date:
Students also viewed these mechanical engineering questions
-
The graphical and symbolic representations off and g are shown. (a) Use the graph to solve f(x) = g(x). (b) Solve f(x) = g(x) symbolically. g(x)=7.5 5 3 f(x)=e -3 -1 0 1 3
-
What is wrong with the following calculation? 3 (-4) --- 1 --4 dx = = -1 X 1 3 3
-
In Exercises 118, simplify the given expression or perform the indicated operation (and simplify, if possible), whichever is appropriate. 2x + 3 x 2 - 7x + 12 2 x - 3
-
How to respond to this response The effective utilization of data is integral to enhancing efficiency and making informed decisions?
-
Using the unit normal table, find the proportion under the standard normal curve that lies to the left of each of the following: (a) z = 0.50 (b) z = 1.32 (c) z = 0 (d) z = 1.96 (e) z = 0.10
-
In the population of people in the United States, about 10% are left handed. After bumping elbows at lunch with several left-handed students, Simon wondered if more than 10% of students at his school...
-
In a transactional data, what is the effect of aggregating similar items into a broader product category? a. Item granularity does not matter as one can simply adjust the minimum support threshold....
-
Gary Bauer opens a computer consulting business called Technology Consultants and completes the following transactions in April. April 1 Bauer invested $100,000 cash along with $24,000 in office...
-
What is the study of abnormal psychology? What are your overall thoughts on the study of abnormal psychology? 2. Explain 1-2 concepts, terms, or statistics that resonated. 3. How can you use the...
-
The director says, "How do you think we could find areas to improve so that we might increase sales at existing stores?" What do you think the best first step would be to determine areas to improve...
-
Find the equation of the line with slope = -2 and passing through (3,-2). Write your equation in point- slope form y-y = m(x-x1) AND slope-intercept forms. point-slope form: slope-intercept form:...
-
Suppose there is a reduced supply of canned tomatoes from China to Australia during COVID-19. Also, consumers are increasingly feeling concerned about supply shortages. What will be the impact of...
-
You are welcome to use Excel to do your calculations and copy and paste your answers here. 1. [6 points] You have been given the following information for NAUFIN Company. They have asked you to do...
-
Let's suppose you (USA dealer) imported 10 BMW (7 series) from a German dealer on March 1, 2018 at 60,000 each, payable in 30 days. The exchange rate on March 1, 2018 was 1.15 US$/. Then you sold...
-
HTBM Ltd. has an outstanding issue of bond with a par value of $1,000, paying 8 percent coupon rate semi-annually. And, the company just paid a dividend of $2.70 per share. The dividends are expected...
-
The three courses below represent a polygon. They are consecutive azimuths measured clockwise from North in DMS. Course A to B: 55-35-21 Course B to C: 175-48-19 Course C to A: 293-22-28 Choose the...
-
In Problems 718, write the augmented matrix of the given system of equations. f0.01x0.03y = 0.06 [0.13x + 0.10y = 0.20
-
A thin-walled container with a hot process fluid at 50C is placed in a quiescent, cold water bath at 100C. Heat transfer at the inner and outer surfaces of the container may be approximated by free...
-
Consider the blister packaging material of Example 14.3. (a) Under the same conditions as in the example, determine the solubility of the polymer material (k mol/m 3 bar) if the temperature is 295 K...
-
Consider a surface-mount type transistor on a circuit board whose temperature is maintained at 35C. Air at 20C flows over the upper surface of dimensions 4 mm by 8 mm with a convection coefficient of...
-
A political pollster approaches people on the street and asks them to describe their political affiliation. Twenty-eight people describe themselves as Democrats, 25 as Republicans, 8 people provide a...
-
Listed below are a number of hypothetical research hypotheses. For each hypothesis, identify the independent and dependent variable. a. Male drivers are more likely to exhibit road rage behaviors...
-
Listed below are a number of research questions and hypotheses from actual published articles. For each hypothesis, identify the independent and dependent variable. a. The use of color in a Yellow...

Study smarter with the SolutionInn App


