A 150 mL sample of oxygen is collected over water at 25 C and 570.0 torr...
Fantastic news! We've Found the answer you've been seeking!
Question:
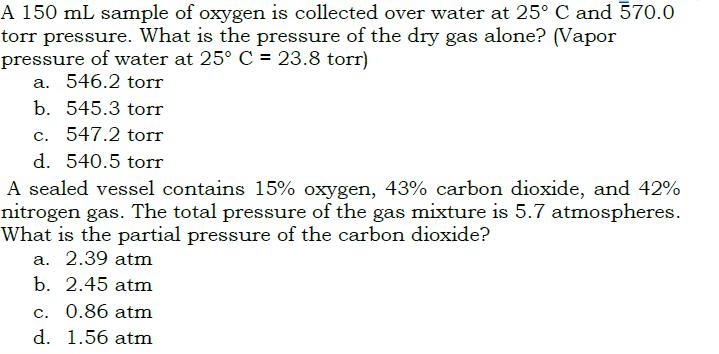
Transcribed Image Text:
A 150 mL sample of oxygen is collected over water at 25° C and 570.0 torr pressure. What is the pressure of the dry gas alone? (Vapor pressure of water at 25° C = 23.8 torr) a. 546.2 torr %3D b. 545.3 torr c. 547.2 torr d. 540.5 torr A sealed vessel contains 15% oxygen, 43% carbon dioxide, and 42% nitrogen gas. The total pressure of the gas mixture is 5.7 atmospheres. What is the partial pressure of the carbon dioxide? a. 2.39 atm b. 2.45 atm c. 0.86 atm d. 1.56 atm A 150 mL sample of oxygen is collected over water at 25° C and 570.0 torr pressure. What is the pressure of the dry gas alone? (Vapor pressure of water at 25° C = 23.8 torr) a. 546.2 torr %3D b. 545.3 torr c. 547.2 torr d. 540.5 torr A sealed vessel contains 15% oxygen, 43% carbon dioxide, and 42% nitrogen gas. The total pressure of the gas mixture is 5.7 atmospheres. What is the partial pressure of the carbon dioxide? a. 2.39 atm b. 2.45 atm c. 0.86 atm d. 1.56 atm
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
1. The following enthalpy changes are known for reactions at 25 "C and one atm. No Reaction AH (kJ/mol) CsHolg) + Hz{g) CHs(g) -123.8 2 CHa(g) + 502(8) 3CO; (8) + 4H20(1) -2220.0 3. Ha(8) + %...
-
A sample of nitrogen gas was collected over water at 20. o C and a total pressure of 1.00 atm. A total volume of 2.50 10 2 mL was collected. What mass of nitrogen was collected? (At 20. o C the...
-
A sample of NO2 is collected over water in a 775 mL container at 25C. If the total pressure is 0.990 atm, how many moles of NO2 are collected?
-
John Hasty opened his bakery on March 1, 1999. The following transactions took place in early March: 1. Deposited $10,000 into a checking account in the name of the Hasty Bakery. 2. Leased a small...
-
When buying health care, most of us dont behave like regular consumers: Seven out of the eight dollars we spend is somebody elses money, and we dont have very good information about doctors or...
-
The time to replace vehicle wiper blades at a service center was monitored using a mean and a range chart. Six samples of n 20 observations were obtained and the sample means and ranges computed: a....
-
How does make determine whether a target needs to be rebuilt?
-
You are creating a customer database for the Modesto Nuts minor league baseball team. Draw a project network given the information below. Complete the forward and backward pass, compute activity...
-
4-21. A boat moves diagonally across a river at a heading of 60 south of east at a speed of V = 8 mph. The river flows due west at a speed of V = 3 mph. The resulting vector diagram for resultant...
-
On January 1, 2017, Hunter Ltd. entered into an agreement to lease a truck from Situ Ltd. Both Hunter and Situ use IFRS 16. The details of the agreement are as follows: Additional information: 1....
-
It takes the Blacksburg shipyard twenty six weeks with 500 workers to build a small tanker Material costs are $32 million and overhead costs are $2 million Workers cost $40 per hour including...
-
Charlene's husband passed away. She is due insurance of $120,000. The insurance company offered that instead of giving her the money right away that she invest the money with them. They will give her...
-
Loser has the following schedule of business gains and losses in current years 1 through 4 : - Year 1 Gains: $ 8 0 , 0 0 0 Losses: $ 1 0 0 , 0 0 0 - Year 2 Gains: $ 1 0 0 , 0 0 0 Losses: $ 1 0 0 ,...
-
A company paid a $2 per share dividend at the end of the previous fiscal year (December 31st). Today is the first day of the new fiscal year (January 1st) and you are deciding whether to purchase one...
-
What is a good question an auditor should always ask in when deciding on a ratio to use? Is management aware of the increases in X liability account over the last five years? What should this...
-
Janet owns 50% of Company X. In year 1, ordinary income in Company X amounted to 8,000,000. Janet is the general manager of Company X, and for this work she received a special remuneration of...
-
Which of these is an example of preventative facework, in interpersonal communication? a. Apologizing for being late to a meeting because of traffic. b. Blaming a coworker for a mistake that was...
-
Complete problem P10-21 using ASPE. Data from P10-21 Original cost ................................................................. $7,000,000 Accumulated depreciation...
-
Consider the following changes: a. N2(g) N2(l) b. CO(g) + H2O(g) H2(g) + CO2(g) c. Ca3P2(s) + 6H2O(l) 3Ca(OH)2(s) + 2PH3(g) d. 2CH3OH(l) + 3O2(g) 2CO2(g) + 4H2O(l) e. I2(s) I2(g) At constant...
-
The element silver (Ag) has two naturally occurring isotopes: 109Ag and 107Ag with a mass of 106.905 amu. Silver consists of 51.82% 107Ag and has an average atomic mass of 107.868 amu. Calculate the...
-
Calculate the wavelength of light emitted when each of the following transitions occur in the hydrogen atom. What type of electromagnetic radiation is emitted in each transition? a. n = 4 n = 3 b. n...
-
Use the Internet to access Home Depot's most recent annual report. a. What evidence did you find that Home Depot continues to rely on present worth analysis in its corporate decision making? b. Does...
-
Use an Internet search engine to identify three companies or agencies that use present worth analysis to make business decisions as evidenced in their annual reports.
-
Use the Internet to access ConocoPhillips's most recent annual report. a. What evidence do you find that ConocoPhillips continues to rely on present worth analysis in its corporate decision making?...

Study smarter with the SolutionInn App


