A 54 kg woman has 8.7 X 10^-3 mol of hemoglobin (molar mass = 65323 g/mol)...
Fantastic news! We've Found the answer you've been seeking!
Question:
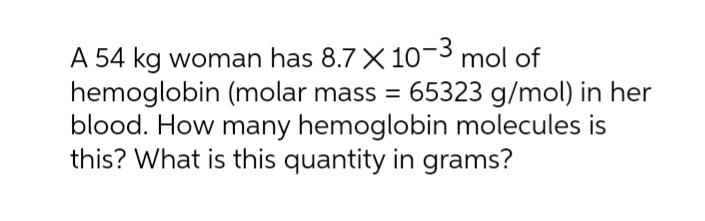
Transcribed Image Text:
A 54 kg woman has 8.7 X 10^-3 mol of hemoglobin (molar mass = 65323 g/mol) in her blood. How many hemoglobin molecules is this? What is this quantity in grams? A 54 kg woman has 8.7 X 10^-3 mol of hemoglobin (molar mass = 65323 g/mol) in her blood. How many hemoglobin molecules is this? What is this quantity in grams?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A woman with mass 50 kg is standing on the rim of a large disk that is rotating at O.50rev/s about an axis through its center. The disk bas mass no kg and radius 4.0 m. calculate the magnitude of the...
-
How many molecules are present in 4.61 102 mol of O2?
-
How many molecules are present in 2.509 mol of H2S?
-
A SQL With respect to database transaction, the ACID concepts should apply! ACID is an acronym for atomicity, consistency, isolation and durability. Please define each of these terms in your own...
-
As plastic bushings are inserted into a 3-in.-diameter cylindrical sheet metal container, the insertion tool exerts the forces shown on the enclosure. Each of the forces is parallel to one of the...
-
Barnes & Noble sells books, magazines, music, and videos through retail stores and online. For a retailer like Barnes & Noble, inventory is a critical element of the business, and it is necessary to...
-
Augusta Oil Corporation, an independent producer, began operations in June 20XA. During the first 2 years of operation, Augusta acquired only two U.S. properties, which were noncontiguous. Costs...
-
The following data were accumulated for use in reconciling the bank account of Commander Co. for March: a. Cash balance according to the companys records at March 31, $13,065. b. Cash balance...
-
Find f. f'(t) = 4 cost + sec t, f(t) = -/2
-
BookWeb, Inc., sells books and software over the Internet. A recent article in a trade journal has caught the attention of management because the company has experienced soaring inventory handling...
-
ences a. Raw materials purchased on account, $210,000 b. Raw materials used in production, $190,000 ($178,000 direct materials and $12,000 indirect materials). c. Accrued direct labor cost of $90,000...
-
The system is initially at rest when it is impacted by a 1 kg mass m2, which is traveling with a speed of 10 m/s just before impact. The coefficient of restitution associated with these two impacting...
-
0 A sample of 200 high school students were asked how many hours per week they spend watching television. The following frequency distribution presents the results. Time Spent Watching Television...
-
Prepare schedules of cost of goods manufactured and cost of goods sold and an income statement. ( Hint: Prepare the income statement and schedule of cost of goods sold first followed by the schedule...
-
A blue bird and a red bird are initially sitting atop two different trees while a hunter (represented by the arrow) is watching them from below. The hunter only has 1 arrow and needs to shoot both...
-
Johnson Controls Corp. , a major U.S. auto parts supplier, has a manufacturing subsidiary in Nuevo Laredo, Mexico, which assembles wiring harnesses for auto electrical systems.Quarterly, Johnson...
-
A company issues 20 years bonds with an 8% coupon paid annually the bonds have a par or face value of $1000 and the current market interest rate for these bonds is 8%. The bonds can be called at the...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
Alveoli are the tiny sacs of air in the lungs whose average diameter is 5.0 10-5 m. Consider an oxygen molecule (5.3 10-26 kg) trapped within a sac. Calculate the uncertainty in the velocity of the...
-
The first line of the Balmer series occurs at a wavelength of 656.3 nm. What is the energy difference between the two energy levels involved in the emission that results in this spectral line?
-
Carbon monoxide (CO) and nitric oxide (NO) are polluting gases contained in automobile exhaust. Under suitable conditions, these gases can be made to react to form nitrogen (N2) and the less harmful...
-
Can any financial product normally make it possible to obtain resources at below market cost?
-
Redo the exercise above, assuming in the first case that the Faurecia share rises to 40 or falls to 25. What is the impact on the value of the option? What basic feature of the option have you...
-
You wish to value a call option on the Faurecia share (which does not pay dividends) after 6 months with a strike price of 35 and a 6-month duration. You do not know what volatility to factor in....

Study smarter with the SolutionInn App



