A 999.1 kg (2199 lb) car is moving at 18.3 m/s (41.0 mph). Calculate the magnitude...
Fantastic news! We've Found the answer you've been seeking!
Question:

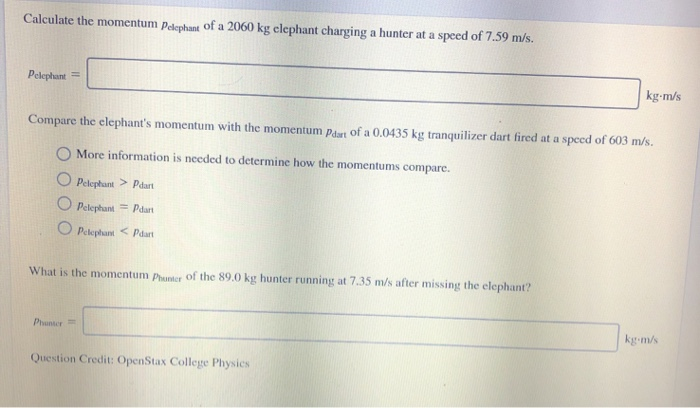
Transcribed Image Text:
A 999.1 kg (2199 lb) car is moving at 18.3 m/s (41.0 mph). Calculate the magnitude of its momentum. magnitude of momentum: kg-m/s Calculate the momentum Pelephant of a 2060 kg elephant charging a hunter at a speed of 7.59 m/s. Pelephant = Compare the elephant's momentum with the momentum Pdart of a 0.0435 kg tranquilizer dart fired at a speed of 603 m/s. O More information is needed to determine how the momentums compare. Pelephant > Pdart O Pelephant =Pdart Pelephant Pdart What is the momentum Phunter of the 89.0 kg hunter running at 7.35 m/s after missing the elephant? Phunter= kg-m/s Question Credit: OpenStax College Physics kg-m/s A 999.1 kg (2199 lb) car is moving at 18.3 m/s (41.0 mph). Calculate the magnitude of its momentum. magnitude of momentum: kg-m/s Calculate the momentum Pelephant of a 2060 kg elephant charging a hunter at a speed of 7.59 m/s. Pelephant = Compare the elephant's momentum with the momentum Pdart of a 0.0435 kg tranquilizer dart fired at a speed of 603 m/s. O More information is needed to determine how the momentums compare. Pelephant > Pdart O Pelephant =Pdart Pelephant Pdart What is the momentum Phunter of the 89.0 kg hunter running at 7.35 m/s after missing the elephant? Phunter= kg-m/s Question Credit: OpenStax College Physics kg-m/s
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
A 1 500-kg car is moving at 20.0 m/s. The driver brakes to a stop. The brakes cool off to the temperature of the surrounding air, which is nearly constant at 20.0C. What is the total entropy change?
-
Calculate the magnitude of the magnetic field at a point 100 cm from a long, thin conductor carrying a current of 1.00 A.
-
Calculate the magnitude of the force between tow 3.60-C point charges 9.3 cm apart.
-
MAX DIVISOR TREE Let a tree exists with the root value N. The property of this tree is that each of its nodes branches out to the nodes with a value equal to one of its divisors (except 1 and the...
-
What internal control procedures would you recommend in each of the following situations? 1. A concession company has one employee who sells towels, coolers, and sunglasses at the beach. Each day,...
-
What are the inputs to an MRP system? What information is provided by each of these inputs?
-
Richmond Sporting Goods uses the LIFO inventory method and values its inven tory using the lower-of-cost-or-market (LCM) rule. Richmond Sporting Goods has the following account balances at May...
-
A mixture of acetone and isopropanol containing 50 mol% acetone is to be distilled continuously to produce an overhead product containing 80 mol% acetone and a bottoms containing 25 mol% acetone. If...
-
For the entries below, Identify the account to be debited and the account to be credited. Indicate which of the accounts is the Income statement account and which is the balance sheet account. Assume...
-
Harrison and Gloria have been negotiating over the sale of bulk raw materials (lumber, iron and steel). Each party has had an army of lawyers acting on both sides trying to get the deal done and it...
-
Complete the table by indicating the present value amount of the annuity Frequency of Annual Number of Beg. Or End of Deferral of Present Value Investment Payments Interest Rate Payment Years Payment...
-
Assume that you have a $22,500 balance on your credit card . You plan to make monthly payments of $450 until the balance is paid off. The interest rate on your credit card is 17.5% p.a., compounded...
-
Write a regular expression that can matching all phonenumbers listedbelow: (You should write a python program to check thematching results) 555.123.4565 +1-(800)-545-2468 2-(800)-545-2468...
-
Question ONE (a) Carboxylic acid CH3CH2COOH reacts with the following reagents to form different products. (i) NaOH (ii) Alcohol in presence of acid catalyst. (iii) LiAlH4. (iv) Cl. Write equation of...
-
Create a resource data filled management plan Resource Histogram (editable template-double click on the chart below) Resource Group Month 1 Month 2 Month 3 Month 4 Month 5 Month 6 Month 7 Month 8...
-
Working with teams in a diverse workplace requires commitment from managers and employees. In this way, a culture can be created where everyone has a sense of belonging. Employers use diversity and...
-
earch Font O Question # 3&4 Draw the NFA, Regular Expression and CFG of the given DFA (10) wird Paragraph DRAWR DFA for the given RE. (a+bb)+(aba+bab) (a+bb)*(a) (b)(a+bb)* 12 13 b a TM 11 a b b b...
-
Why do markets typically lead to an efficient outcome for buyers and sellers?
-
Calculate the pressure differential of ethanol across the surface of a spherical droplet of radius 220 nm at 20e. The surface tension of ethanol at that temperature is 22.39 mN m-1
-
For 14N2 G values for the transitions v = 1 0, 2 0, and 3 0 are, respectively, 2345.15, 4661.40, and 6983.73 cm-1. Calculate v and xe- Assume ye to be zero.
-
The wave number of the fundamental vibrational transition of 79BrB1Bris 323.2 cm-1, Calculate the force constant of the bond (mC9Br) = 78.9183 u, m(8IBr) = 80.9163 u).
-
Refer to Exercise 5. Four of the eight students are from Middle Georgia State University. What is the probability that all three of the interviewed students are from Middle Georgia State University?
-
The law of large numbers states that as a probability experiment is repeated, the proportion of times that a given outcome occurs will approach its probability. In Exercises 912, determine whether...
-
The following table presents numbers of U.S. workers, in thousands, categorized by type of occupation and educational level. a. What is the probability that a randomly selected worker is a college...

Study smarter with the SolutionInn App



