a) A Cu-Ag alloy consists of 71.9 wt% Ag. What are the compositions of the individual...
Fantastic news! We've Found the answer you've been seeking!
Question:
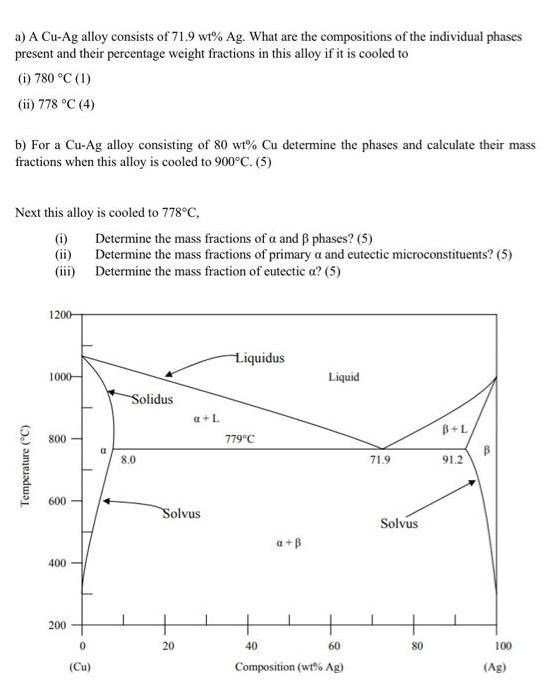
Transcribed Image Text:
a) A Cu-Ag alloy consists of 71.9 wt% Ag. What are the compositions of the individual phases present and their percentage weight fractions in this alloy if it is cooled to (i) 780 °C (1) (ii) 778 °C (4) b) For a Cu-Ag alloy consisting of 80 wt% Cu determine the phases and calculate their mass fractions when this alloy is cooled to 900°C. (5) Next this alloy is cooled to 778°C, Temperature (°C) (i) (ii) (iii) 1200- 1000- 800 600 400 200 0 (Cu) Determine the mass fractions of a and ẞ phases? (5) Determine the mass fractions of primary a and eutectic microconstituents? (5) Determine the mass fraction of eutectic a? (5) Solidus 8.0 a + L Solvus 20 Liquidus 779°C a+B Liquid 40 60 Composition (wt% Ag) 71.9 Solvus 80 B+L 91.2 100 (Ag) a) A Cu-Ag alloy consists of 71.9 wt% Ag. What are the compositions of the individual phases present and their percentage weight fractions in this alloy if it is cooled to (i) 780 °C (1) (ii) 778 °C (4) b) For a Cu-Ag alloy consisting of 80 wt% Cu determine the phases and calculate their mass fractions when this alloy is cooled to 900°C. (5) Next this alloy is cooled to 778°C, Temperature (°C) (i) (ii) (iii) 1200- 1000- 800 600 400 200 0 (Cu) Determine the mass fractions of a and ẞ phases? (5) Determine the mass fractions of primary a and eutectic microconstituents? (5) Determine the mass fraction of eutectic a? (5) Solidus 8.0 a + L Solvus 20 Liquidus 779°C a+B Liquid 40 60 Composition (wt% Ag) 71.9 Solvus 80 B+L 91.2 100 (Ag)
Expert Answer:
Answer rating: 100% (QA)
a i At 780C the CuAg alloy will exist as a single CuAg phase consisting of 100 by weigh... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Haley Corp. issued stock at $27 per share and a dividend of 7% per annum. Dividends are expected to grow at an annual rate of 5% to infinity. What is the stock's required rate of retum? A. 10% B. 11%...
-
A SQL With respect to database transaction, the ACID concepts should apply! ACID is an acronym for atomicity, consistency, isolation and durability. Please define each of these terms in your own...
-
Explain how the management functions of planning, organizing, leading, and controlling apply to a growing entrepreneurial company.
-
If hiring a CEO is not the answer, what should they do to get their business to the next level?
-
A 72-kg woman is walking at \(1.5 \mathrm{~m} / \mathrm{s}\). An \(8-\mathrm{kg}\) dog is running at six times that speed in the same direction. At what speed and in what direction relative to the...
-
1. Treating Cost/Mile as the dependent variable, develop an estimated regression with Family-Sedan and Upscale-Sedan as the independent variables. Discuss your findings. 2. Treating Value Score as...
-
(a) A company has an EPS of Rs. 2.5 for the last year and the DPS of Rs. 1. The earnings is expected to grow at 2% a year in long run. Currently it is trading at 7 times its earnings. If the required...
-
Costa, Inc., recently converted from a 5-day, 40-hour workweek to a 4-day, 40-hour workweek, with overtime continuing to be paid at one and one-half times the regular hourly rate for all hours worked...
-
You now need to calculate the cost of debt for SNA. Go to finra-markets.morningstar.com, enter SNA as the company, and find the yield to maturity for each of SNA's bonds. What is the weighted average...
-
Why the artificial wetland system is characterized?
-
3) How did online banking technology change the cost structure of banks in terms of variable and fixed costs? How did this affect the structure of the banking industry? 4) Suppose that a market is...
-
Why would the probability of an employer offering retiree health insurance increase with the number of employees for large firms over 300 workers. Support your answer with scholarly resources.
-
What laws can be put in place to help protect consumers as well as make them have more trust in the law process? What is an instance where you have seen the law fall short when it comes to...
-
Justin is an unlucky soul who got Herpes Simplex Virus 1 (HSV-1) from his girlfriend in junior high. Now he is a carrier for HSV-1 and anytime he experiences high levels of stress, he develops cold...
-
Salty Co. uses a periodic inventory system. Assume GST at 5% and PST at 8%. (any available cash discount is taken on the sale price before taxes). The following selected transactions occurred: May 5...
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
If hydrogen were used as a fuel, it could be burned according to this reaction: Use average bond energies to calculate H rxn for this reaction and also for the combustion of methane (CH 4 ). Which...
-
Without doing any calculations, determine which of the samples contains the greatest amount of the element in moles. Which contains the greatest mass of the element? a. 55.0 g Cr b. 45.0 g Ti c. 60.0...
-
A particular reaction has an equilibrium constant of K p = 0.50. A reaction mixture is prepared in which all the reactants and products are in their standard states. In which direction does the...
-
Which of the following is a sale of goods and therefore is covered by Article 2 of the Uniform Commercial Code? A. Development, implementation, hosting, and operation of sophisticated computing...
-
When a new health club opens, the owners offer a discount to the first 100 members who enter into a five-year contract. The fee is \($2,400\) annually for the first five years, prepaid, regardless of...
-
Berkley Corp. wanted to buy 1,000 customized umbrellas imprinted with their logo to use for promotional purposes. It planned to use 250 of the umbrellas for an event scheduled for early February 2012...

Study smarter with the SolutionInn App


