a) A stream contains a mixture of 40% normal heptane (by mol) and 60% toluene. Calculate...
Fantastic news! We've Found the answer you've been seeking!
Question:
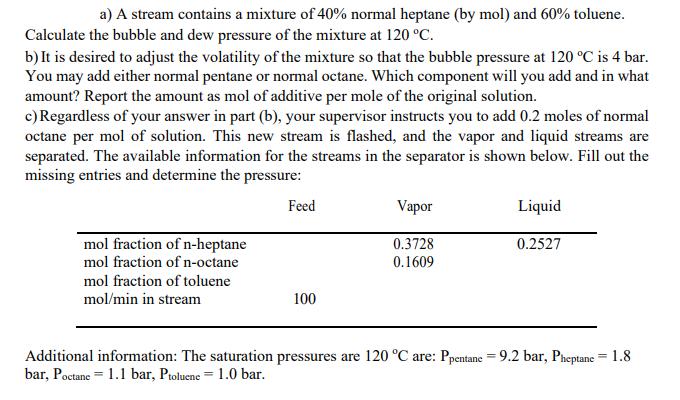
Transcribed Image Text:
a) A stream contains a mixture of 40% normal heptane (by mol) and 60% toluene. Calculate the bubble and dew pressure of the mixture at 120 °C. b) It is desired to adjust the volatility of the mixture so that the bubble pressure at 120 °C is 4 bar. You may add either normal pentane or normal octane. Which component will you add and in what amount? Report the amount as mol of additive per mole of the original solution. c) Regardless of your answer in part (b), your supervisor instructs you to add 0.2 moles of normal octane per mol of solution. This new stream is flashed, and the vapor and liquid streams are separated. The available information for the streams in the separator is shown below. Fill out the missing entries and determine the pressure: Feed Vapor Liquid mol fraction of n-heptane 0.3728 0.2527 mol fraction of n-octane 0.1609 mol fraction of toluene mol/min in stream 100 Additional information: The saturation pressures are 120 °C are: Ppentane = 9.2 bar, Pheptane = 1.8 bar, Poctane 1.1 bar, Ptoluene 1.0 bar. = a) A stream contains a mixture of 40% normal heptane (by mol) and 60% toluene. Calculate the bubble and dew pressure of the mixture at 120 °C. b) It is desired to adjust the volatility of the mixture so that the bubble pressure at 120 °C is 4 bar. You may add either normal pentane or normal octane. Which component will you add and in what amount? Report the amount as mol of additive per mole of the original solution. c) Regardless of your answer in part (b), your supervisor instructs you to add 0.2 moles of normal octane per mol of solution. This new stream is flashed, and the vapor and liquid streams are separated. The available information for the streams in the separator is shown below. Fill out the missing entries and determine the pressure: Feed Vapor Liquid mol fraction of n-heptane 0.3728 0.2527 mol fraction of n-octane 0.1609 mol fraction of toluene mol/min in stream 100 Additional information: The saturation pressures are 120 °C are: Ppentane = 9.2 bar, Pheptane = 1.8 bar, Poctane 1.1 bar, Ptoluene 1.0 bar. =
Expert Answer:
Answer rating: 100% (QA)
a To calculate the bubble and dew pressures of the mixture at 120C we need to use Raoults Law which ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
References: Health Information Management Case Studies Second Edition AHIMA by Dianna M. Foley Health Information Management Technology an Applied Approah Sixth Edition AHIMA by Nanette Sayles and...
-
Sunita inherits $45,000 from a distant relative. She decides to invest her inheritance in a diversified, dividend-paying mutual fund. At the time of her initial investment, she purchases 1,500 shares...
-
List the ethical responsibilities of group members and leaders.
-
Explain how lasers are used to read a compact disc.
-
An investigator is considering two blocking schemes for a $2^{4}$ design with 4 blocks. The two schemes are listed below. Scheme 1: $B_{1}=134, B_{2}=234$ Scheme 2: $B_{1}=12, B_{2}=13, B_{3}=1234$...
-
Kara owns 35% of the KLM Partnership and 45% of the KTV Partnership. Lynn owns 20% of KLM and 3% of KTV. Maura, Karas daughter, owns 15% of KTV. No other partners own an interest in both partnerships...
-
Print Company acquired 80% of the stock of Sand Corp. on January 1, 2023. The stockholder's equity section of Sand's balance sheet at that date is as follows: Stockholder's Equity Section Common...
-
Use PSpice to find V1, V2, and V3 in the network of Fig. 10.128. 8 V. j1012 V, jlOS2 4/0 A
-
Liis Compeny lost 70% of its inventory in a fire on March 25, 2015. The accounting records showed the following gross profe data for February and March. March (to February 1/25) $250,000 Net sales...
-
Repeat Problem 55 using your own age. Data from Problem 55 In 2012 the maximum Social Security deposit by an individual was \(\$ 8,386.75\). Suppose you are 25 and make a deposit of this amount into...
-
Becoming an accountant Referring to the membership areas of CPA Australia and CAANZ, answer the following questions. Required (a) Compare the entry requirements to become a full member of CPA...
-
Two jets flying at the same altitude pass over your head simultancously, one traveling at Mach 1. 5 and the other at Mach 2. 5. Which plane's sonic boom do you hear first?
-
You want to hang a \(10-\mathrm{kg}\) sign that advertises your new business. To do this, you use a pivot to attach the base of a \(5.0-\mathrm{kg}\) beam to a wall (Figure P12.36). You then attach a...
-
A circuit you're building needs an ammeter that goes from \(0 \mathrm{~mA}\) to a full-scale reading of \(50.0 \mathrm{~mA}\). Unfortunately, the only ammeter in the storeroom goes from \(0 \mu...
-
The Upton Company reported a beginning balance of $1,600 and an ending balance of $2,200 in its Unearned Revenue account for Year 2. During the year, $8,000 of revenue was recognized. Based on this...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
The reaction A + B 2C takes place in an unsteady CSTR. The feed is only A and B in equimolar proportions. Which of the following sets of equations gives the correct set of mole balances on A, B, and...
-
Go to the LearnChemE screencast link for Chapter 15 (http://www.learncheme.com/screencasts/kinetics-reactor-design). View the two screencasts: (1) Diffusion and Reaction in a Cylindrical Porous...
-
Example 11-2: Heat of Reaction. (1) What would the heat of reaction be if 50% inerts (e.g., helium) were added to the system? (2) What would be the % error if the CP term were neglected? Example 11-2...
-
To address his concern regarding the previous advisers asset allocation approach, Raye should assess the Laws portfolio using: A. a homogeneous and mutually exclusive asset classbased risk analysis....
-
Raye believes the previous advisers specification for debt is incorrect given that, for purposes of asset allocation, asset classes should be: A. diversifying. B. mutually exclusive. C. relatively...
-
Walker Patel is a portfolio manager at an investment management firm. After successfully implementing meanvariance optimization (MVO), he wants to apply reverse optimization to his portfolio. For...

Study smarter with the SolutionInn App


