The coldest seawater ever recorded (in a liquid state) was in a stream under an Antarctic...
Fantastic news! We've Found the answer you've been seeking!
Question:

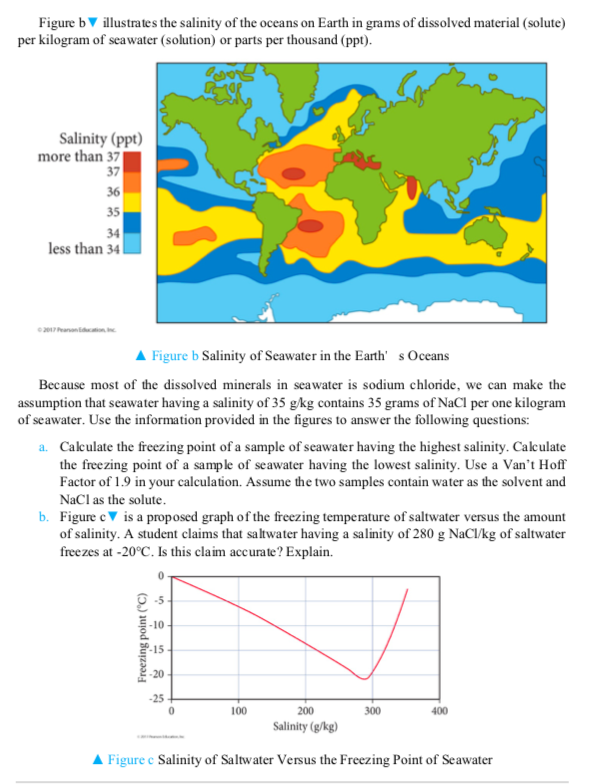

Transcribed Image Text:
The coldest seawater ever recorded (in a liquid state) was in a stream under an Antarctic glacier in 2010; it measured -2.6 °C (27.3 °F). On average, water in the world's oceans has a salinity of about 3.5% (35 g/L, or 599 mm). This means that every kilogram (roughly one liter by volume) of seawater contains approximately 35 grams (1.2 ounces) of dissolved salts (predominantly sodium (Na) and chloride (CI) ions). Figure a provides information on the amounts of various cations and anions in seawater with an average density of 1.025 g/ml. Sea Salts Seawater Sulfate- 7.7% (2.7g) Calcium- 1.2% (0.42g) Potassium- 1.1% (0.39g) Chloride 55% (19.25g) Sodium 30.6% (10.7g) Magnesium 3.7% (1.3g) -Minor constituents 0.7% (0.25g) Water 96.5% (965g) Salt 3.5% (35g) A Figure a Cations and anions in Ocean Water Figure by illustrates the salinity of the oceans on Earth in grams of dissolved material (solute) per kilogram of seawater (solution) or parts per thousand (ppt). Salinity (ppt) more than 371 37 36 35 34 less than 34 © 2017 Pearson Education, Inc. Figure b Salinity of Seawater in the Earth' s Oceans Because most of the dissolved minerals in seawater is sodium chloride, we can make the assumption that seawater having a salinity of 35 g/kg contains 35 grams of NaCl per one kilogram of seawater. Use the information provided in the figures to answer the following questions: a. Calculate the freezing point of a sample of seawater having the highest salinity. Calculate the freezing point of a sample of seawater having the lowest salinity. Use a Van't Hoff Factor of 1.9 in your calculation. Assume the two samples contain water as the solvent and NaCl as the solute. b. Figure c is a proposed graph of the freezing temperature of saltwater versus the amount of salinity. A student claims that saltwater having a salinity of 280 g NaCl/kg of saltwater freezes at -20°C. Is this claim accurate? Explain. Freezing point (°C) -10 POT 198 -15- -20 100 300 400 200 Salinity (g/kg) ▲ Figure e Salinity of Saltwater Versus the Freezing Point of Seawater c. Use the information provided in Figure b and redraw the graph in Figure c using a range of salinity values from six different oceans. The y-axis should be the freezing point of water, and the x-axis should be the salinity of seawater in units of grams of NaCl/kg of seawater. Does Figure e provide an accurate view of the freezing point of the oceans? The coldest seawater ever recorded (in a liquid state) was in a stream under an Antarctic glacier in 2010; it measured -2.6 °C (27.3 °F). On average, water in the world's oceans has a salinity of about 3.5% (35 g/L, or 599 mm). This means that every kilogram (roughly one liter by volume) of seawater contains approximately 35 grams (1.2 ounces) of dissolved salts (predominantly sodium (Na) and chloride (CI) ions). Figure a provides information on the amounts of various cations and anions in seawater with an average density of 1.025 g/ml. Sea Salts Seawater Sulfate- 7.7% (2.7g) Calcium- 1.2% (0.42g) Potassium- 1.1% (0.39g) Chloride 55% (19.25g) Sodium 30.6% (10.7g) Magnesium 3.7% (1.3g) -Minor constituents 0.7% (0.25g) Water 96.5% (965g) Salt 3.5% (35g) A Figure a Cations and anions in Ocean Water Figure by illustrates the salinity of the oceans on Earth in grams of dissolved material (solute) per kilogram of seawater (solution) or parts per thousand (ppt). Salinity (ppt) more than 371 37 36 35 34 less than 34 © 2017 Pearson Education, Inc. Figure b Salinity of Seawater in the Earth' s Oceans Because most of the dissolved minerals in seawater is sodium chloride, we can make the assumption that seawater having a salinity of 35 g/kg contains 35 grams of NaCl per one kilogram of seawater. Use the information provided in the figures to answer the following questions: a. Calculate the freezing point of a sample of seawater having the highest salinity. Calculate the freezing point of a sample of seawater having the lowest salinity. Use a Van't Hoff Factor of 1.9 in your calculation. Assume the two samples contain water as the solvent and NaCl as the solute. b. Figure c is a proposed graph of the freezing temperature of saltwater versus the amount of salinity. A student claims that saltwater having a salinity of 280 g NaCl/kg of saltwater freezes at -20°C. Is this claim accurate? Explain. Freezing point (°C) -10 POT 198 -15- -20 100 300 400 200 Salinity (g/kg) ▲ Figure e Salinity of Saltwater Versus the Freezing Point of Seawater c. Use the information provided in Figure b and redraw the graph in Figure c using a range of salinity values from six different oceans. The y-axis should be the freezing point of water, and the x-axis should be the salinity of seawater in units of grams of NaCl/kg of seawater. Does Figure e provide an accurate view of the freezing point of the oceans?
Expert Answer:
Answer rating: 100% (QA)
A Calculating the freezing point of a sample of seawater with the highest salinity 37 ppt and the lo... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
A and B react to produce C according to the following chemical equation: A+ B C Amounts of A and B are added to an equilibrium reaction mixture of A, B, and C such that when equilibrium is again...
-
A and B disjoint, B and C not disjoint, A and C not disjoint. Draw Venn diagrams with sets A, B, and C satisfying the above requirements.
-
A and B disjoint, B and C disjoint, A and C not disjoint. Draw Venn diagrams with sets A, B, and C satisfying the above requirements.
-
Suppose that you borrow $1000.00 from a friend and promise to pay back $1975.00 in 5 years. What simple interest rate will you pay?
-
A collar is established by buying a share of stock for $50, buying a 6-month put option with exercise price $45, and writing a 6-month call option with exercise price $55. On the basis of the...
-
The marketing concept focuses on the ability of marketing to satisfy customer needs. As a typical college student, how does marketing satisfy your needs? What areas of your life are affected by...
-
How to prepare exhibits for trial?
-
Apply the value chain model to a video game developer, such as Electronic Arts (www.EA.com). What is its competitive strategy? Describe the tasks Electronic Arts must accomplish for each of the...
-
Brisbun and Adlayde are two companies selling similar products in a competitive market. While their profitability is similar, their cost structure is different. The summary results for each company...
-
The Fashion Rack is a retail merchandising business that sells brand-name clothing at discount prices. The firm is owned and managed by Teresa Lojay, who started the business on April 1, 20X1. This...
-
Your role as head of the Municipality, amongst others, is to choose the most appropriate way to ensure the business model of the municipality serves the community optimally. The role of a municipal...
-
What methodologies can be employed to optimize the coherence and conciseness of a comprehensive report, ensuring its efficacy in conveying multifaceted information to diverse stakeholders ?
-
Perry Bell paid $6,438 of state and local property and income tax this year. Assume the taxable year is 2023.\ \ Required:\ Compute the after-tax cost of these payments assuming Perry doesnt itemize...
-
An object is dropped from a tower, 1936 ft above the ground. The object's height above the ground t seconds after it is dropped is s(t)=1936-1612. Determine the velocity and acceleration of the...
-
A 1.1-kg peregrine falcon moving due East at 54 m/s grabs onto a 1.2-kg Mallard duck in mid-air. It only takes 0.025 seconds for the falcon to latch onto the duck, after which the two are moving due...
-
5:17 Curtis Incorporated uses a job order cost system. Manufacturing overhead is applied on the basis of direct labor cost. Total manufacturing overhead was estimated to be $52,920 for the year;...
-
When an object/donor such as a shoe sole or a tire tread comes in contact with another object with force, a(n) may be left in the recipient as evidence.
-
TRUE-FALSE QUESTIONS 1. In terms of preliminary analytical procedures, assume that the company has introduced a new product with a low price point and significant customer demand. The auditor would...
-
Robert Ramos (age 36) is a single taxpayer, living at 8765 Bay Dr., Monterey, CA 93940. His Social Security number is 976-23-5132. Robert's earnings and income tax withholding as the manager of a...
-
During 2012, William purchases the following capital assets for use in his catering business: New passenger automobile (September 30)........................$21,500 Baking equipment (June 30)...
-
Sherry rents her vacation home for 6 months and lives in it for 6 months during the year. Her gross rental income during the year is $4,000. Total real estate taxes for the home are $950, and...
-
a. Identify the three steps involved in quantitatively evaluating sample results. b. Indicate the steps involved in using sample evaluation tables.
-
a. What factors should be considered in qualitatively evaluating sample results? b. What alternative courses of action should be considered when sample results do not support the auditor's planned...
-
a. Why might the auditor select some extra sample items when sampling from a population of documents? b. State what is meant by a nonapplicable item and give an example.

Study smarter with the SolutionInn App


