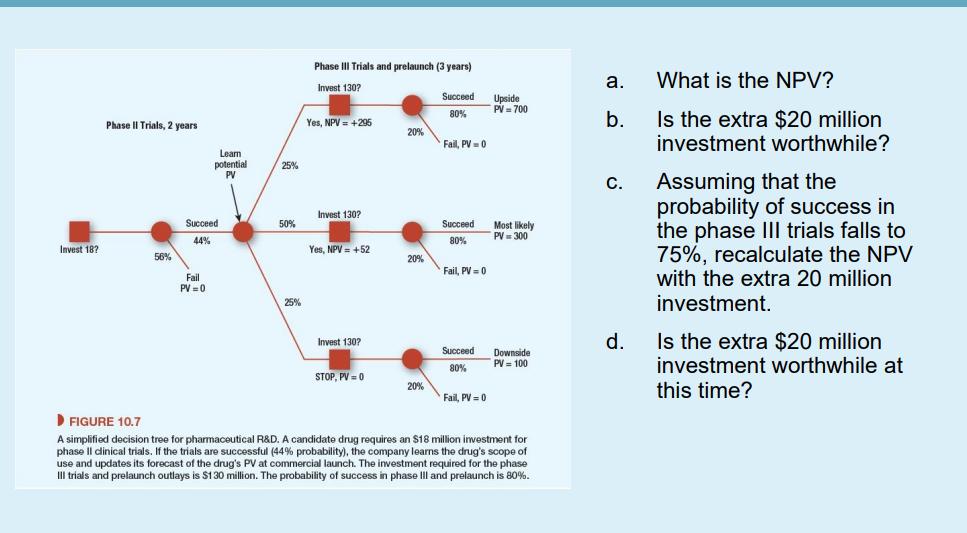
A candidate drug requires an $18 million investment for phase II clinical trials. If the trials...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A candidate drug requires an $18 million investment for phase II clinical trials. If the trials are successful (44% probability), the company will learn the drug's scope of use and update its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. Launch comes three years after the start of phase Ill if the drug is approved by the FDA. The probabilities of the upside, most likely, and downside outcomes are 25%, 50%, and 25%, respectively. If the phase III trials are successful, the manager will learn the commercial potential of the drug, which will depend on how widely it can be used. Suppose that the forecasted PV at launch depends on the scope of use allowed by the FDA. These are an upside outcome of NPV = $700 million if the drug can be widely used, a most likely case with NPV = $300 million, and a downside case of NPV = $100 million if the drug's scope is greatly restricted. We assume a risk-free rate of 4% and market risk premium of 7%. Assume FDA-approved pharmaceutical products have asset betas of 0.8 and the opportunity cost of capital is 9.6%. Accordingly, the NPV works out to $19 million. The R&D team has put forward a proposal to invest an extra $20 million in expanded phase II trials. Phase II takes two years. Th object is to prove that the drug can be administered by a simple inhaler rather than as a liquid. If successful, the scope of use is broadened and the upside PV increases to $1 billion. The probabilities of success are unchanged. Invest 18? Phase II Trials, 2 years 56% Learn potential PV Succeed 44% Fail PV=0 25% 50% 25% Phase Trials and prelaunch Invest 130? Yes, NPV = +295 Invest 130? Yes, NPV +52 Invest 130? STOP, PV = 0 20% 20% 20% years) Succeed 80% Fail, PV = 0 Succeed 80% Fail, PV=0 Succeed 80% Fail, PV = 0 Upside PV = 700 Most likely PV = 300 Downside PV = 100 ▶FIGURE 10.7 A simplified decision tree for pharmaceutical R&D. A candidate drug requires an $18 million investment for phase Il clinical trials. If the trials are successful (44% probability), the company learns the drug's scope of use and updates its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. a. b. C. d. What is the NPV? Is the extra $20 million investment worthwhile? Assuming that the probability of success in the phase III trials falls to 75%, recalculate the NPV with the extra 20 million investment. Is the extra $20 million investment worthwhile at this time? A candidate drug requires an $18 million investment for phase II clinical trials. If the trials are successful (44% probability), the company will learn the drug's scope of use and update its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. Launch comes three years after the start of phase Ill if the drug is approved by the FDA. The probabilities of the upside, most likely, and downside outcomes are 25%, 50%, and 25%, respectively. If the phase III trials are successful, the manager will learn the commercial potential of the drug, which will depend on how widely it can be used. Suppose that the forecasted PV at launch depends on the scope of use allowed by the FDA. These are an upside outcome of NPV = $700 million if the drug can be widely used, a most likely case with NPV = $300 million, and a downside case of NPV = $100 million if the drug's scope is greatly restricted. We assume a risk-free rate of 4% and market risk premium of 7%. Assume FDA-approved pharmaceutical products have asset betas of 0.8 and the opportunity cost of capital is 9.6%. Accordingly, the NPV works out to $19 million. The R&D team has put forward a proposal to invest an extra $20 million in expanded phase II trials. Phase II takes two years. Th object is to prove that the drug can be administered by a simple inhaler rather than as a liquid. If successful, the scope of use is broadened and the upside PV increases to $1 billion. The probabilities of success are unchanged. Invest 18? Phase II Trials, 2 years 56% Learn potential PV Succeed 44% Fail PV=0 25% 50% 25% Phase Trials and prelaunch Invest 130? Yes, NPV = +295 Invest 130? Yes, NPV +52 Invest 130? STOP, PV = 0 20% 20% 20% years) Succeed 80% Fail, PV = 0 Succeed 80% Fail, PV=0 Succeed 80% Fail, PV = 0 Upside PV = 700 Most likely PV = 300 Downside PV = 100 ▶FIGURE 10.7 A simplified decision tree for pharmaceutical R&D. A candidate drug requires an $18 million investment for phase Il clinical trials. If the trials are successful (44% probability), the company learns the drug's scope of use and updates its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. a. b. C. d. What is the NPV? Is the extra $20 million investment worthwhile? Assuming that the probability of success in the phase III trials falls to 75%, recalculate the NPV with the extra 20 million investment. Is the extra $20 million investment worthwhile at this time? A candidate drug requires an $18 million investment for phase II clinical trials. If the trials are successful (44% probability), the company will learn the drug's scope of use and update its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. Launch comes three years after the start of phase Ill if the drug is approved by the FDA. The probabilities of the upside, most likely, and downside outcomes are 25%, 50%, and 25%, respectively. If the phase III trials are successful, the manager will learn the commercial potential of the drug, which will depend on how widely it can be used. Suppose that the forecasted PV at launch depends on the scope of use allowed by the FDA. These are an upside outcome of NPV = $700 million if the drug can be widely used, a most likely case with NPV = $300 million, and a downside case of NPV = $100 million if the drug's scope is greatly restricted. We assume a risk-free rate of 4% and market risk premium of 7%. Assume FDA-approved pharmaceutical products have asset betas of 0.8 and the opportunity cost of capital is 9.6%. Accordingly, the NPV works out to $19 million. The R&D team has put forward a proposal to invest an extra $20 million in expanded phase II trials. Phase II takes two years. Th object is to prove that the drug can be administered by a simple inhaler rather than as a liquid. If successful, the scope of use is broadened and the upside PV increases to $1 billion. The probabilities of success are unchanged. Invest 18? Phase II Trials, 2 years 56% Learn potential PV Succeed 44% Fail PV=0 25% 50% 25% Phase Trials and prelaunch Invest 130? Yes, NPV = +295 Invest 130? Yes, NPV +52 Invest 130? STOP, PV = 0 20% 20% 20% years) Succeed 80% Fail, PV = 0 Succeed 80% Fail, PV=0 Succeed 80% Fail, PV = 0 Upside PV = 700 Most likely PV = 300 Downside PV = 100 ▶FIGURE 10.7 A simplified decision tree for pharmaceutical R&D. A candidate drug requires an $18 million investment for phase Il clinical trials. If the trials are successful (44% probability), the company learns the drug's scope of use and updates its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. a. b. C. d. What is the NPV? Is the extra $20 million investment worthwhile? Assuming that the probability of success in the phase III trials falls to 75%, recalculate the NPV with the extra 20 million investment. Is the extra $20 million investment worthwhile at this time? A candidate drug requires an $18 million investment for phase II clinical trials. If the trials are successful (44% probability), the company will learn the drug's scope of use and update its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. Launch comes three years after the start of phase Ill if the drug is approved by the FDA. The probabilities of the upside, most likely, and downside outcomes are 25%, 50%, and 25%, respectively. If the phase III trials are successful, the manager will learn the commercial potential of the drug, which will depend on how widely it can be used. Suppose that the forecasted PV at launch depends on the scope of use allowed by the FDA. These are an upside outcome of NPV = $700 million if the drug can be widely used, a most likely case with NPV = $300 million, and a downside case of NPV = $100 million if the drug's scope is greatly restricted. We assume a risk-free rate of 4% and market risk premium of 7%. Assume FDA-approved pharmaceutical products have asset betas of 0.8 and the opportunity cost of capital is 9.6%. Accordingly, the NPV works out to $19 million. The R&D team has put forward a proposal to invest an extra $20 million in expanded phase II trials. Phase II takes two years. Th object is to prove that the drug can be administered by a simple inhaler rather than as a liquid. If successful, the scope of use is broadened and the upside PV increases to $1 billion. The probabilities of success are unchanged. Invest 18? Phase II Trials, 2 years 56% Learn potential PV Succeed 44% Fail PV=0 25% 50% 25% Phase Trials and prelaunch Invest 130? Yes, NPV = +295 Invest 130? Yes, NPV +52 Invest 130? STOP, PV = 0 20% 20% 20% years) Succeed 80% Fail, PV = 0 Succeed 80% Fail, PV=0 Succeed 80% Fail, PV = 0 Upside PV = 700 Most likely PV = 300 Downside PV = 100 ▶FIGURE 10.7 A simplified decision tree for pharmaceutical R&D. A candidate drug requires an $18 million investment for phase Il clinical trials. If the trials are successful (44% probability), the company learns the drug's scope of use and updates its forecast of the drug's PV at commercial launch. The investment required for the phase III trials and prelaunch outlays is $130 million. The probability of success in phase III and prelaunch is 80%. a. b. C. d. What is the NPV? Is the extra $20 million investment worthwhile? Assuming that the probability of success in the phase III trials falls to 75%, recalculate the NPV with the extra 20 million investment. Is the extra $20 million investment worthwhile at this time?
Expert Answer:
Answer rating: 100% (QA)
Original case NPV upside 130 0870010963 295 million NPV most likely 130 0830010963 52 million NPV do... View the full answer

Related Book For 

Spreadsheet Modeling And Decision Analysis A Practical Introduction To Business Analytics
ISBN: 9781305947412
8th Edition
Authors: Cliff T. Ragsdale
Posted Date:
Students also viewed these finance questions
-
A 40-year bond pays quarterly coupons and has 12 years left until maturity. If you bought the bond, you would - receive a coupon payment of R98,50 every three months. The bond has an interest rate of...
-
What is the present value of a perpetual stream of R1 350 annual payments discounted at an interest rate of 8% compounded annually, if the first payment occurs in one year's time?
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A standard "35-mm" slide measures 24.0 mm by 36.0 mm. Suppose a slide projector produces a 60.0-cm by 90.0-cm image of the slide on a screen. The focal length of the lens is 12.0 cm. (a) What is the...
-
A single-slit diffraction pattern is formed on a distant screen. Assuming the angles involved are small, by what factor will the width of the central bright spot on the screen change if (a) The...
-
In 2013, the City of Coyote received a $320,000 cash grant from the state to stop air pollution. Assume that although a special revenue fund could have been set up, the money remained in the General...
-
Tristate Recreation Park (TRP) finances operations with both bonds and stock. Suppose TRP issued \(\$ 200,000\) of 10 -year, \(6 \%\) bonds payable under various market conditions. Match each market...
-
Varner Inc. and King Inc. have the following operating data: a. Compute the operating leverage for Varner Inc. and King Inc.b. How much would income from operations increase for each company if the...
-
Question 2, T18-3 (similar to) Part 1 of 4 HW Score: 0%, 0 of 100 points O Points: 0 of 30 Save Jagan Company uses the allowance method to account for uncollectible receivables. On July 2, Jagan...
-
Using Bayes decision rule, consider the decision analysis problem having the following payoff table (in units of thousands of dollars): (a) Which alternative should be chosen? What is the resulting...
-
Shares of a company are valued at 40 per share at the moment, with 8,000,000 shares out- standing. They plan to issue 2,000,000 new shares at a price per share of 30. Balance sheet before capital...
-
Canyon Dental Services is a specialized dental practice whose only service is filling cavities. Canyon has recorded the following for the past nine months: Number of Cavities Month January Filled...
-
2. A 25-kg crate rest atop a platform as shown at right. If the friction force acting on the block sliding is 120 N find: a) The work done by gravity as the block falls to the ground off the incline...
-
Crane Service Center just purchased an automobile hoist for $31,500. The hoist has an 8-year life and an estimated salvage value of $3,800. Installation costs and freight charges were $4,100 and...
-
Find the net electric flux through the spherical closed surface shown in the figure below. The two charges on the right are inside the spherical surface. (Take 91 -2.97 nC.) 91 N m/c Need Help? Read...
-
Coney Island enters into a lease agreement for a new ride. The lease payments have a present value of $2.5 million. Prior to agreement, the company's total assets are $26.5 million and its total...
-
On 31 December 2020, IAG presents the following information related to its Cash Flow (mill. Euros): Operating profit: 3,250. Depreciation: 1,250. Movement in working capital: -790. Interest paid:...
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
A bank has $650,000 in assets to allocate among investments in bonds, home mortgages, car loans, and personal loans. Bonds are expected to produce a return of 10%, mortgages 8.5%, car loans 9.5%, and...
-
Benjamin Franklin once said, In this world nothing is certain but death and taxes. Although that might be true, there is often great uncertainty involved in when one will encounter death and how much...
-
Road Racer Sports, Inc. is a mail-order business dedicated to the running enthusiast. The company sends out full-color catalogs several times a year to several hundred thousand people on its mailing...
-
Linear acceleration method a. Assumes that acceleration varies linearly between \(t_{i}\) and \(t_{i}+\theta \Delta t ; \theta \geq 1\) b. Assumes that acceleration varies linearly between \(t_{i}\)...
-
Fill in the Blanks. In a conditionally stable method, the use of \(\Delta t\) larger than \(\Delta t_{\text {cri }}\) makes the method _____________.
-
The finite difference method requires the use of finite difference approximations in a. governing differential equation only b. boundary conditions only c. governing differential equation as well as...

Study smarter with the SolutionInn App


