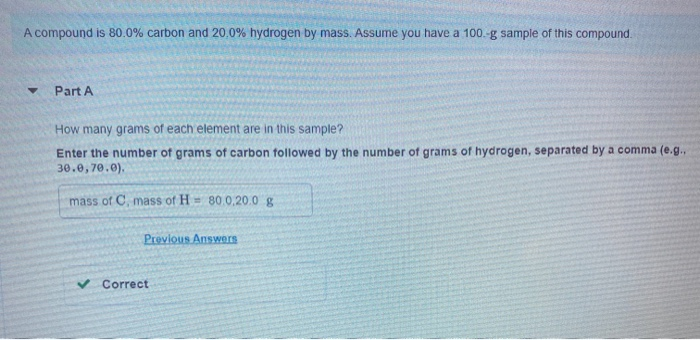
A compound is 80.0% carbon and 20.0% hydrogen by mass. Assume you have a 100.-g sample...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
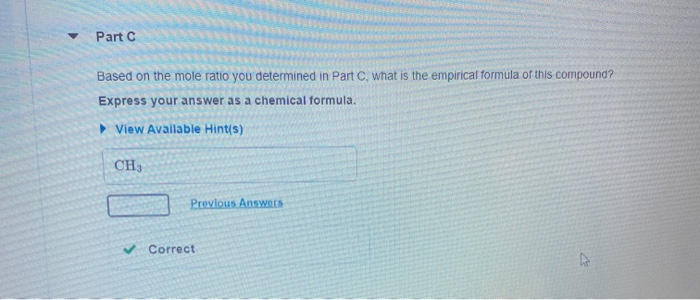
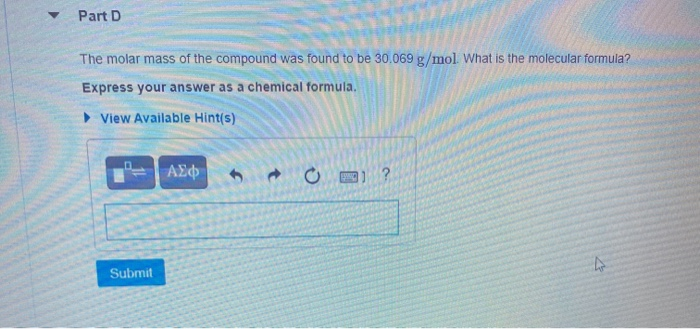
A compound is 80.0% carbon and 20.0% hydrogen by mass. Assume you have a 100.-g sample of this compound. Part A How many grams of each element are in this sample? Enter the number of grams of carbon followed by the number of grams of hydrogen, separated by a comma (e.g.. 30.0,70.0). mass of C, mass of H 80.0.20.0 g Previous Answers ✓ Correct Part B How many moles of each element are in this sample? Enter the number of moles of carbon followed by the number of moles of hydrogen, separated by a comma. View Available Hint(s) moles of C, moles of H= 6.66,19.8 mol Previous Answers ✓ Correct ▼ Part C Based on the mole ratio you determined in Part C, what is the empirical formula of this compound? Express your answer as a chemical formula. View Available Hint(s) CH3 Previous Answers ✓ Correct N Part D The molar mass of the compound was found to be 30.069 g/mol. What is the molecular formula? Express your answer as a chemical formula. View Available Hint(s) Submit ΑΣΦ A compound is 80.0% carbon and 20.0% hydrogen by mass. Assume you have a 100.-g sample of this compound. Part A How many grams of each element are in this sample? Enter the number of grams of carbon followed by the number of grams of hydrogen, separated by a comma (e.g.. 30.0,70.0). mass of C, mass of H 80.0.20.0 g Previous Answers ✓ Correct Part B How many moles of each element are in this sample? Enter the number of moles of carbon followed by the number of moles of hydrogen, separated by a comma. View Available Hint(s) moles of C, moles of H= 6.66,19.8 mol Previous Answers ✓ Correct ▼ Part C Based on the mole ratio you determined in Part C, what is the empirical formula of this compound? Express your answer as a chemical formula. View Available Hint(s) CH3 Previous Answers ✓ Correct N Part D The molar mass of the compound was found to be 30.069 g/mol. What is the molecular formula? Express your answer as a chemical formula. View Available Hint(s) Submit ΑΣΦ
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Based on the mole ratio you determined in Part C, what is the empirical formula of this compound? Carbon: 6.66 Hydrogen: 19.8 Express your answer as a chemical formula. Part C answers are correct...
-
Express your answer as a chemical formula. a-Write a formula for the compound that forms from potassium and acetate. b-Write a formula for the compound that forms from potassium and chromate. c-Write...
-
C 8 H 10 , express your answer as a chemical formula. n
-
Suppose you are given the following set of data with three Boolean input variables a, b, and c, and a single Boolean output variable K. a bc K 1 0 1 1 1 1 1 1 1 1 1 1 1 1 0 01 For parts (a) and (b),...
-
On June 21, 2007, the Livingston Company, a U.S. company, sold merchandise on credit to a Swiss company for 25,000 francs. The company received payment for the merchandise on July 10, 2007. The...
-
What is a reorder point system in inventory control?
-
1. Tariq Ahmad owned Shankman Laboratories. He decided to dispose of some of the lab's hazardous chemicals by shipping them to his home in Pakistan. He sent the chemicals to Castelazo & Associates...
-
Jellyfish Machine Shop is a manufacturer of motorized carts for vacation resorts. Patrick Cullin, the plant manager of Jellyfish, obtains the following information for Job # 10 in August 2014. A...
-
(4 markah) ii. Nilai kini bersin. (6 markah) iii. Kadar pulangan dalaman (IRR) untuk projek B sahaja. (4 markah) iv. Manakah projek yang harus dipilih oleh Syarikat Kenanga? Jelaskan dapatan anda. (2...
-
The Alfredo Fragrance Company produces only one product, a perfume called Hint of Elegance. Hint of Elegance consists of two secret ingredients blended into an exclusive fragrance, which is marketed...
-
Use the residential sales property data below: Sales Price $880,000 . . Sales Date 23 September 2021 Land Area 730m Dwelling floor area 246m (including garage) Dwelling includes a 6m x 6m internal...
-
Sheena Stone 1S an attorney in Los Angeles. Stone uses the direct write-off method to account for uncollectible receivables. At November 30, 2010, Stones accounts receivable totaled \($21,000\)....
-
Write an application letter to apply for this position, outlining how you would be able to meet the needs of the role.
-
z = 0.77, z = 1.54, z = -1.54 In Exercises 41 and 42, the midpoints A, B, and C are marked on the histograms at the left. Match them with the indicated z-scores. Which z-scores, if any, would be...
-
A nnapolis Hospital is trying to reduce costs, yet it aims to improve the patient count and medical services. The hospital pharmacy, which is considered a primary process, uses two types of...
-
Lee, one of Smart Sports clothing suppliers, is considering upgrading his cutting equipment. Currently, he has a manually intensive cutting process, but this could be more efficient with new cutting...
-
Office Max purchased office furniture in the amount of $89,517 and was invoiced with terms of 2/10, n/30. Cash strapped at the time, Office Max decided to make a partial payment of $50,000 within 10...
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
Shown below is the cubic unit cell of an ionic compound. Answer the questions by referring to this structure. Be careful to note that some atoms are hidden by those in front. a. One of the spheres...
-
How many grams of oxygen gas are required to produce 7.60 kJ of heat when hydrogen gas burns at constant pressure to produce gaseous water? 2H2(g) + O2(g) 2H2O(g); H = 484 kJ Liquid water has a heat...
-
You react nitrogen and hydrogen in a container to produce ammonia, NH3(g). The following figure depicts the contents of the container after the reaction is complete. a. Write a balanced chemical...
-
Question: To ensure that its employees did not use illegal drugs in or outside of the workplace, Rain Co. required all employees to take a lie detector test. Moreover, managers began to screen the...
-
Question: Pippa became pregnant the week she started work as an administrator at Awesome University. Her supervisor was so annoyed at her that he would not consider her for promotion to another job...
-
Question: When Theodore Staats went to his company's "Council of Honor Con- vention," he was accompanied by a woman who was not his wife although he told everyone she was. The company fired him....

Study smarter with the SolutionInn App


