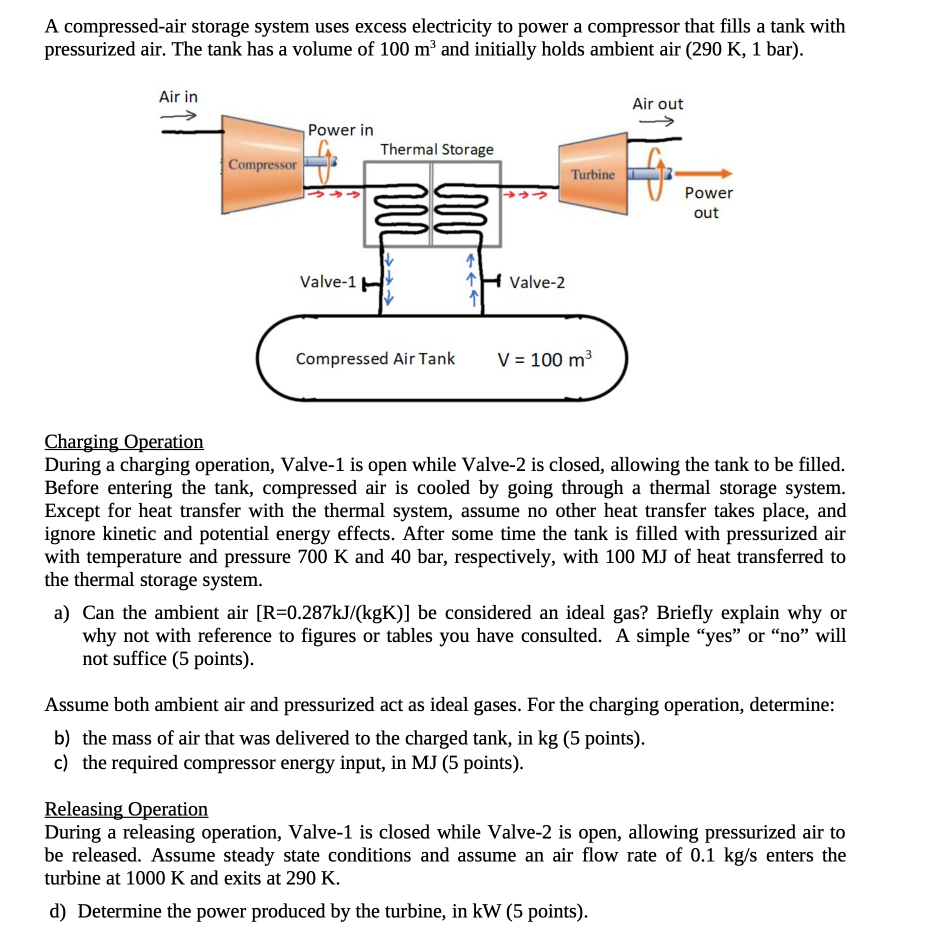
A compressed-air storage system uses excess electricity to power a compressor that fills a tank with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A compressed-air storage system uses excess electricity to power a compressor that fills a tank with pressurized air. The tank has a volume of 100 m and initially holds ambient air (290 K, 1 bar). Air in Compressor Power in Thermal Storage Valve-1 H Compressed Air Tank Valve-2 Turbine V = 100 m Air out Power out Charging Operation During a charging operation, Valve-1 is open while Valve-2 is closed, allowing the tank to be filled. Before entering the tank, compressed air is cooled by going through a thermal storage system. Except for heat transfer with the thermal system, assume no other heat transfer takes place, and ignore kinetic and potential energy effects. After some time the tank is filled with pressurized air with temperature and pressure 700 K and 40 bar, respectively, with 100 MJ of heat transferred to the thermal storage system. a) Can the ambient air [R=0.287kJ/(kgK)] be considered an ideal gas? Briefly explain why or why not with reference to figures or tables you have consulted. A simple "yes" or "no" will not suffice (5 points). Assume both ambient air and pressurized act as ideal gases. For the charging operation, determine: b) the mass of air that was delivered to the charged tank, in kg (5 points). c) the required compressor energy input, in MJ (5 points). Releasing Operation During a releasing operation, Valve-1 is closed while Valve-2 is open, allowing pressurized air to be released. Assume steady state conditions and assume an air flow rate of 0.1 kg/s enters the turbine at 1000 K and exits at 290 K. d) Determine the power produced by the turbine, in kW (5 points). A compressed-air storage system uses excess electricity to power a compressor that fills a tank with pressurized air. The tank has a volume of 100 m and initially holds ambient air (290 K, 1 bar). Air in Compressor Power in Thermal Storage Valve-1 H Compressed Air Tank Valve-2 Turbine V = 100 m Air out Power out Charging Operation During a charging operation, Valve-1 is open while Valve-2 is closed, allowing the tank to be filled. Before entering the tank, compressed air is cooled by going through a thermal storage system. Except for heat transfer with the thermal system, assume no other heat transfer takes place, and ignore kinetic and potential energy effects. After some time the tank is filled with pressurized air with temperature and pressure 700 K and 40 bar, respectively, with 100 MJ of heat transferred to the thermal storage system. a) Can the ambient air [R=0.287kJ/(kgK)] be considered an ideal gas? Briefly explain why or why not with reference to figures or tables you have consulted. A simple "yes" or "no" will not suffice (5 points). Assume both ambient air and pressurized act as ideal gases. For the charging operation, determine: b) the mass of air that was delivered to the charged tank, in kg (5 points). c) the required compressor energy input, in MJ (5 points). Releasing Operation During a releasing operation, Valve-1 is closed while Valve-2 is open, allowing pressurized air to be released. Assume steady state conditions and assume an air flow rate of 0.1 kg/s enters the turbine at 1000 K and exits at 290 K. d) Determine the power produced by the turbine, in kW (5 points).
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
We have titrated natural water in which the primary buffering agent is carbonate species and understood how to predict the titration curve using the equilibrium constants of those weak acids. Plot...
-
Refer to Samsung's balance sheet in Appendix A. How does its cash (titled "Cash and cash equivalents") compare with its other current assets (both in amount and percent) as of December 31, 2012?...
-
Fred Oatly is the loan officer of the National Bank of Dallas. National has a loan of \(\$ 260,000\) outstanding to Regional Delivery Service, a company specializing in delivering products of all...
-
Remember, it is important to draft the motion for summary judgment sufficiently in advance of the hearing date in order to allow time to serve and file the motion. Review CCP 437c, which governs...
-
What is the control requirement of 351? Describe the effect of the following in satisfying this requirement: a. A shareholder renders only services to the corporation for stock. b. A shareholder...
-
GFA has a contract with four local cities to acquire the compost that is recycled by city residents. GFA only pays for transportation from the compost collection facility. Once the compost is...
-
I farm on a small scale. Last year, I started off by planting soybeans and corn. This year, the price of soybeans increased, and the price of corn remained the same. Naturally, I planted more corn...
-
After researching CMA Medical Ethics and Professionalism material. Why did the CMA decide it was necessary to update the Medical Code of Ethics? How will this new code of ethics effect the medical...
-
Assignment 3 Case Study Marketing Management You have been contacted by a European Company with an offer for a joint venture. The company is in to Cotton Garments manufacturing. They have full...
-
Do you think that policy as it relates to the grazing of livestock on federal lands is effective? Why or why not?
-
Car drives on a level circular track. Radius of curve is 2 0 meters. Coefficent of friction between car and ground is 0 . 2 . What is the highest velocity the car can drive and stay on the track...
-
What is the standard deviation of the expected returns on this stock? State of the economy Boom Probability 0.30 Normal 0.70 TIP: E(R) =PiRi Variance ()=Pi(Ri -) E(R) 25% 12%
-
Mod 18.2: Company A is a retailer of high-end pencils and has provided the following inventory information for January 20X1. Day 1: Began the day with 10 pencils with a per unit cost of $7 each. Day...
-
Suppose some wind moves southward from the equator on Jupiter, Which direction will this wind bend? Toward the East Toward the North Toward the South Toward the West
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
The exothermic reaction of stillbene (A) to form the economically important trospophene (B) and methane(C), that is, A B+ C was carried out adiabatically and the following data recorded: The...
-
Suggest combinations of ideal reactors to model the real reactors given in Problem P16-2b(b) for either E(), E(t), F(), F(t), or (1 F()).
-
Terephthalic acid (TPA) finds extensive use in the manufacture of synthetic fibers (e.g., Dacron) and as an intermediate for polyester films (e.g., Mylar). The formation of potassium terephthalate...
-
Derive Maxwell's relations.
-
A spherical balloon of \(1 \mathrm{~m}\) diameter contains a gas at \(120 \mathrm{kPa}\). The gas inside the balloon is heated until the pressure reaches \(360 \mathrm{kPa}\). During heating the...
-
(a) State and prove Clausius inequality theorem. (b) Nitrogen is compressed from an initial state of 1 bar and \(25^{\circ} \mathrm{C}\) to a final state of \(5 \mathrm{bar}\) and \(25^{\circ}...

Study smarter with the SolutionInn App



