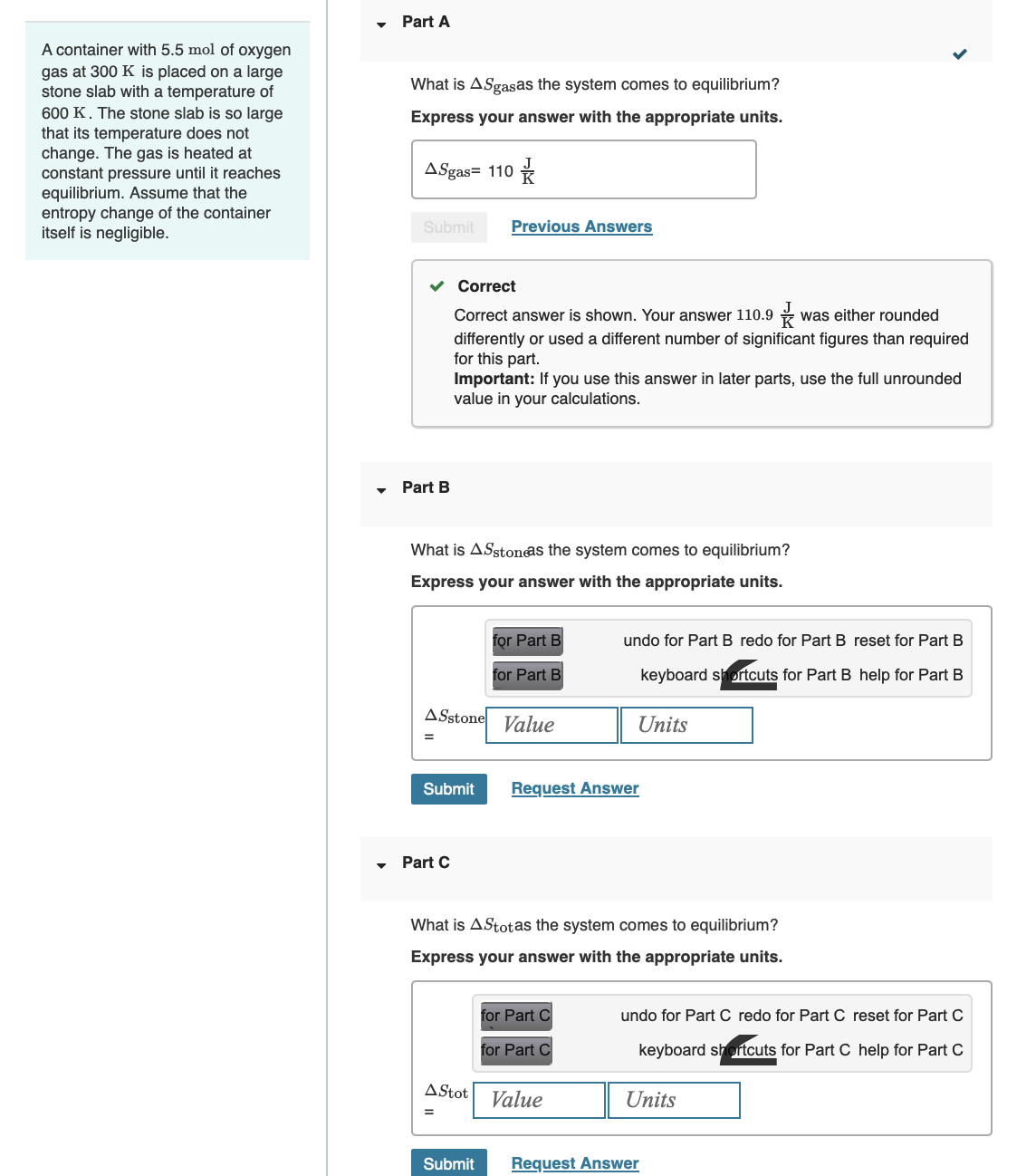
A container with 5.5 mol of oxygen gas at 300 K is placed on a large...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A container with 5.5 mol of oxygen gas at 300 K is placed on a large stone slab with a temperature of 600 K. The stone slab is so large that its temperature does not change. The gas is heated at constant pressure until it reaches equilibrium. Assume that the entropy change of the container itself is negligible. Part A What is ASgasas the system comes to equilibrium? Express your answer with the appropriate units. ASgas= 110 Submit Previous Answers Correct Part B Correct answer is shown. Your answer 110.9 was either rounded differently or used a different number of significant figures than required for this part. Important: If you use this answer in later parts, use the full unrounded value in your calculations. What is AS stone as the system comes to equilibrium? Express your answer with the appropriate units. for Part B for Part B undo for Part B redo for Part B reset for Part B keyboard shortcuts for Part B help for Part B ASstone Value Units Submit Request Answer Part C What is Astotas the system comes to equilibrium? Express your answer with the appropriate units. for Part C for Part C A Stot Value undo for Part C redo for Part C reset for Part C keyboard shortcuts for Part C help for Part C Units Submit Request Answer A container with 5.5 mol of oxygen gas at 300 K is placed on a large stone slab with a temperature of 600 K. The stone slab is so large that its temperature does not change. The gas is heated at constant pressure until it reaches equilibrium. Assume that the entropy change of the container itself is negligible. Part A What is ASgasas the system comes to equilibrium? Express your answer with the appropriate units. ASgas= 110 Submit Previous Answers Correct Part B Correct answer is shown. Your answer 110.9 was either rounded differently or used a different number of significant figures than required for this part. Important: If you use this answer in later parts, use the full unrounded value in your calculations. What is AS stone as the system comes to equilibrium? Express your answer with the appropriate units. for Part B for Part B undo for Part B redo for Part B reset for Part B keyboard shortcuts for Part B help for Part B ASstone Value Units Submit Request Answer Part C What is Astotas the system comes to equilibrium? Express your answer with the appropriate units. for Part C for Part C A Stot Value undo for Part C redo for Part C reset for Part C keyboard shortcuts for Part C help for Part C Units Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
Part B To calculate the change in entropy of the stone slab ASstone as the system come... View the full answer

Posted Date:
Students also viewed these physics questions
-
You are a tax associate with a large tax accounting firm. You have been assigned to prepare a federal income tax return, Form 1120: U.S. Corporation Income Tax Return, and the necessary schedules for...
-
Most law enforcement officers go their whole career without ever having to discharge a firearm. When the situation calls for an officer to use deadly force many issues come up in very rapid...
-
Example of grounded theory in relation to general psychology. Give an example that could be used in the field of psychology and how a grounded theory would fit into that example.
-
Matching Financial Statement Items to Financial Statement Categories According to its annual report, P&Gs more than 250 brands include Pampers, Tide, Ariel, Always, Whisper, Pantene, Bounty,...
-
What are managerial options and why are they important?
-
A stainless steel cylinder (18% Cr, 8% Ni) is heated to a uniform temperature of 200C and then allowed to cool in an environment where the air temperature is maintained constant at 30C. The...
-
What is the probability of drawing at least 1 consonant when drawing four tiles? You are about to draw Scrabble tiles from a bag without replacement; the bag contains the letters A, A, C, E, E, E, L,...
-
The following information is from White Mountain Furniture Showrooms financial records. Collections from customers are normally 70 percent in the month of sale, 20 percent in the month following the...
-
Please just answer the question and put your own words it doesnt have to be very long. 1. Receivables are a significant part of a company's assets. What are included in a company's receivables? 2....
-
Financing: Write the financing information under the FEDERAL TRUTH-IN-LENDING DISCLOSURES part of the contract. 10. Indicate the amount borrowed (balance due) in the Amount Financed box. 11. Assume...
-
To which of the following matters would materiality limits not apply in obtaining written management representations? a. The availability of minutes of stockholders' and directors' meetings. b....
-
This article shows that even major businesses do not necessarily keep appropriate accounting records and report properly for various transactions (such as directors emoluments, taxes, fixed assets...
-
Which of the following is not an audit procedure that the independent auditor would perform concerning litigation, claims, and assessments? a. Obtain assurance from management that it has disclosed...
-
The existence of audit risk is recognized by the statement in the auditor's standard report that the a. Auditor is responsible for expressing an opinion on the financial statements, which are the...
-
To test a hypothesis about a standard deviation using a sample of size 15, we use a chi-square distribution with ___________________ degrees of freedom. In Exercises 5 and 6, fill in each blank with...
-
Elliott and Prescott, CPA's, have recently determined that subsequent to the audit report issuance date, (March 3, 2024) of the audited financial statements of Beltre, Inc., a nonpublic company, for...
-
Give the products of the following reaction, where T is tritium: dioldehydrase Ad- CH CH3C-COH CoIII) coenzyme B12
-
a) Calculate the absolute difference and relative difference from this data. Sales Food Beverage Total Absolute Relative Year 1 Year 2 difference difference % 678,950 787,582 378,450 280,053...
-
What is the cash operating cycle and how is it calculated?
-
What are the key elements of working capital?

Study smarter with the SolutionInn App

