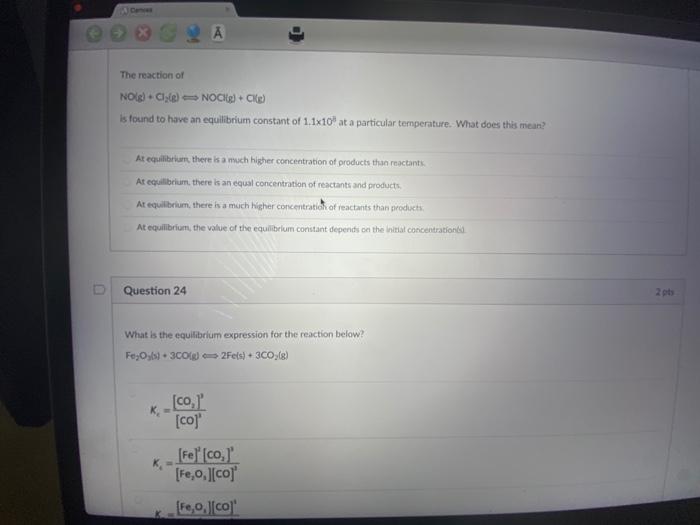
A Conver The reaction of NO(g) + Cl(g) NOCK(g) + CKg) is found to have an...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
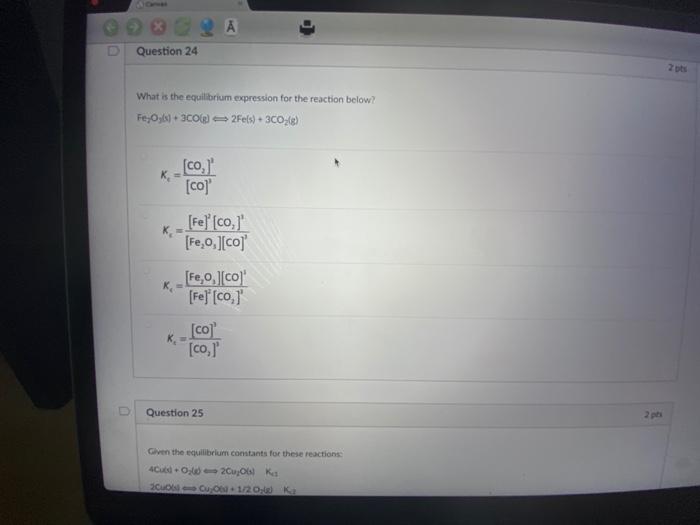
A Conver The reaction of NO(g) + Cl(g) NOCK(g) + CKg) is found to have an equilibrium constant of 1.1x10 at a particular temperature. What does this mean? 2 A At equilibrium, there is a much higher concentration of products than reactants At equilibrium, there is an equal concentration of reactants and products. At equilibrium, there is a much higher concentration of reactants than products. At equilibrium, the value of the equilibrium constant depends on the initial concentrationé D Question 24 What is the equilibrium expression for the reaction below? Fe₂Oys) + 3C0(g) 2Fe(s) + 3CO; (g) K₂ K [co, [co]' [Fe] [co, [Fe,o,][co] [Fe,0,][co] 2 pts Question 24 What is the equilibrium expression for the reaction below? Fe,Oys) + 3C0(g) 2Fe(s) + 3CO₂(g) _[co,]' [co]' [Fe] [co,]' [Fe₂O,][co] K [Fe,o,][co]' [Fe] [co,]' [co] [co,]' Question 25 Given the equilibrium constants for these reactions: 2Cu₂06) Kes 4Cubd-O 20u06) Cu 06-1/20g) Ka 2 pts 2 pts A Conver The reaction of NO(g) + Cl(g) NOCK(g) + CKg) is found to have an equilibrium constant of 1.1x10 at a particular temperature. What does this mean? 2 A At equilibrium, there is a much higher concentration of products than reactants At equilibrium, there is an equal concentration of reactants and products. At equilibrium, there is a much higher concentration of reactants than products. At equilibrium, the value of the equilibrium constant depends on the initial concentrationé D Question 24 What is the equilibrium expression for the reaction below? Fe₂Oys) + 3C0(g) 2Fe(s) + 3CO; (g) K₂ K [co, [co]' [Fe] [co, [Fe,o,][co] [Fe,0,][co] 2 pts Question 24 What is the equilibrium expression for the reaction below? Fe,Oys) + 3C0(g) 2Fe(s) + 3CO₂(g) _[co,]' [co]' [Fe] [co,]' [Fe₂O,][co] K [Fe,o,][co]' [Fe] [co,]' [co] [co,]' Question 25 Given the equilibrium constants for these reactions: 2Cu₂06) Kes 4Cubd-O 20u06) Cu 06-1/20g) Ka 2 pts 2 pts
Expert Answer:
Answer rating: 100% (QA)
Question23 Solution The given chemical reaction is as follows NO g Cl 2 g NOCl g Cl g The value of t... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A diatomic molecule is found to have an activation energy of 1.4eV. When the molecule is disassociated, 1.6eV of energy is released. Draw a potential energy curve for this molecule.
-
A hypothetical element X is found to have an atomic mass of 37.45 amu. Element X has only two isotopes, X-37 and X-38. The X-37 isotope has a fractional abundance of 0.7721 and an isotopic mass of...
-
A diatomic molecule is found to have an activation energy of 1.3 eV. When the molecule is disassociated, 1.6 eV of energy is released. Draw a potential energy curve for this molecule.
-
Figure 2 shows a 250 kg beam BC at the time instant when a 100 N horizontal force is applied to its end B for 10 s. a. Determine the tension in rods AB and CD at an arbitrary instant during the time...
-
Who issues commercial paper and for what purpose?
-
An investment broker is instructed by her client to invest up to $20,000, some in a junk bond yielding 9% per annum and some in Treasury bills yielding 7% per annum. The client wants to invest at...
-
What advantages are there to using social media sites for informal discovery? How may the information you find help in settling a civil lawsuit?
-
Theresa Thayer, a friend from college, asks you to form a partnership to import fragrances. Since graduating, Thayer has worked for the Spanish Embassy, developing important contacts among government...
-
5. What if one of the purchasing agents agreed to work half time for $14,000? a. How many purchase orders could be processed by four and a half purchasing agents? purchase orders b. What would unused...
-
Chandra was the sole shareholder of Pet Emporium, which was originally formed as an S corporation. When Pet Emporium terminated its S election on August 31, 2019, Chandra had a stock basis and an...
-
1. The deformation gradient, denoted F, is computed from a finite element analysis of a complex forging process. At a critical location, it is found to have the components (with respect to a RHON...
-
How do social movements mobilize collective action and challenge dominant power structures to effect social change and promote social justice ?
-
Thalassines Kataskeves, S.A., of Greece makes marine equipment. The company has been experiencing losses on its bilge pump product line for several years. The most recent quarterly contribution...
-
What are the implications of postcolonial theory for understanding the legacies of colonialism and ongoing processes of decolonization in globalized societies ?
-
2. For the given structure and boundary conditions, find the allowable load Pallow given the material strengths are: Tf=210 MPa for pins C and D in shear; and of=400 MPa for bars BC and DE in...
-
Discuss an outline of your SWOT analysis identifying the organizational competencies and strategic advantages. strengths include a strong popularity in healthcare, a strong group of employees, strong...
-
Discuss the role of intrinsic & extrinsic cues in the perceived quality of you product/service(Unilever company; Lipton tea as an example). Include it in the about section of assignment
-
Conduct a VRIO analysis by ranking Husson University (in Maine) business school in terms of the following six dimensions relative to the top three rival schools. If you were the dean with a limited...
-
What is the total charge of all the electrons in a 25-kg bar of aluminum? (Aluminum has 13 electrons per atom and an atomic mass of 27 u.)
-
Which of the following statements is true? (a) A larger-diameter lens can better resolve two distant points. (b) Red light can better resolve two distant points than blue light can. (c) It is easier...
-
Estimate the peak wavelength of light emitted from the pupil of the human eye (which approximates a blackbody) assuming normal body temperature.
-
There are several advantages to incorporating your business, but can you list some of the commonly perceived disadvantages?
-
Define the three terms: asset; liability; and equity.
-
Reflect on the supplementary questions raised in Illustration 1.1. Do you think it is straightforward to ascertain accurate and reliable answers? Try to write in jargon-free terms an example of the...

Study smarter with the SolutionInn App


