Written assignment 4-Due Wednesday Aug 3rd in class You are allowed to use your notes, but...
Fantastic news! We've Found the answer you've been seeking!
Question:

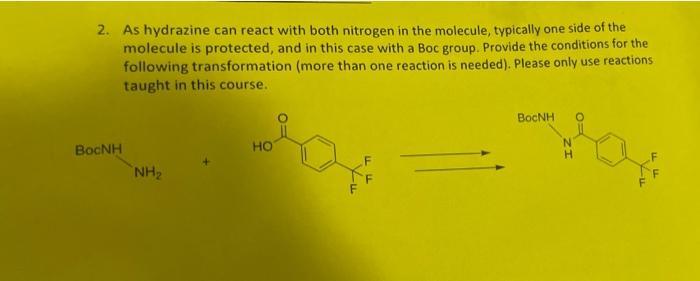
Transcribed Image Text:
Written assignment 4-Due Wednesday Aug 3rd in class You are allowed to use your notes, but your first attempt should be noteless to see how much material you remember. Returning to Tecovirimat (Figure 1), you should now be able to synthesize the bulk of the molecule. Figure 1: Tecovirimat One additional reaction needed is the McLoughlin-Thrower reaction (Figure 2). For this exercise, you can assume this reaction condition will not cause any side reaction with other functional groups in the molecule. F HO -F DMF Cu Figure 2: McLoughlin-Thrower reaction. 1. Please design a synthesis for para-trifluoromethylbenzoic acid (Figure 3) from benzene. FF FF Figure 3: Right fragment of Tecovirimat, para-trifluoromethylbenzoic acid 2. As hydrazine can react with both nitrogen in the molecule, typically one side of the molecule is protected, and in this case with a Boc group. Provide the conditions for the following transformation (more than one reaction is needed). Please only use reactions taught in this course. Bạnh NH₂ HO ما FF BOCNH N H TI-A LLF Written assignment 4-Due Wednesday Aug 3rd in class You are allowed to use your notes, but your first attempt should be noteless to see how much material you remember. Returning to Tecovirimat (Figure 1), you should now be able to synthesize the bulk of the molecule. Figure 1: Tecovirimat One additional reaction needed is the McLoughlin-Thrower reaction (Figure 2). For this exercise, you can assume this reaction condition will not cause any side reaction with other functional groups in the molecule. F HO -F DMF Cu Figure 2: McLoughlin-Thrower reaction. 1. Please design a synthesis for para-trifluoromethylbenzoic acid (Figure 3) from benzene. FF FF Figure 3: Right fragment of Tecovirimat, para-trifluoromethylbenzoic acid 2. As hydrazine can react with both nitrogen in the molecule, typically one side of the molecule is protected, and in this case with a Boc group. Provide the conditions for the following transformation (more than one reaction is needed). Please only use reactions taught in this course. Bạnh NH₂ HO ما FF BOCNH N H TI-A LLF
Expert Answer:
Related Book For 

Process Dynamics and Control
ISBN: 978-1119385561
4th edition
Authors: Dale E. Seborg, Thomas F. Edgar, Duncan A. Mellichamp, Francis J. Doyle
Posted Date:
Students also viewed these chemical engineering questions
-
A steam-heated evaporator used to concentrate a feed stream by evaporating water is shown in Fig. E8.14. The mass fraction of solute in the exit stream x is measured and controlled by adjusting the...
-
A solution contains 0.00750 M calcium ion. A concentrated sodium fluoride solution is added to precipitate calcium fluoride (assume no volume change). a. At what concentration of F does precipitate...
-
A solution contains 0.0150 M lead(II) ion. A concentrated sodium iodide solution is added to precipitate lead iodide (assume no volume change). a. At what concentration of I does precipitate start to...
-
In April 2016, Vanessa bought 100 shares in Entagon plc at a cost of 5 per share. The company went into liquidation and Vanessa received a first distribution of 40p per share in July 2020. The shares...
-
Which of the following statements best explains why a closed balloon filled with helium gas rises in air? (a) Helium is a monatomic gas, whereas nearly all the molecules that make up air, such as...
-
A compound microscope has an objective lens with a focal length of 2.2 cm and an eyepiece with a focal length of 5.4 cm. If the image produced by the objective is 12 cm from the objective, what...
-
\(X\) is the number of bits in error in the next four bits transmitted. What is the expected value of the square of the number of bits in error? Now, \(h(X)=X^{2}\). Therefore, \[ \begin{aligned}...
-
As of December 31, 2015, its first year in business, Kukui Company had taxable temporary differences totaling $110,000. Of this total, $45,000 relates to current items. Kukui also had deductible...
-
Below is a 21x4 register file - it has 2, 4-bit registers. The value of all inputs overlay the diagram and the current state of the registers overlay the registers (i.e., Q = 0x?). All current input...
-
A tennis ball is mounted on a spring that is suspended from a ceiling in John's dorm room. For entertainment, stress relief, boredom or just for a pure conversation piece, the tennis ball can be...
-
-> Let G and H be groups. A function : G H is called a (group) homomorphism if it satisfies (9192) = (91) * (92) for all 91, 92 G. (Note that the product 91*92 uses the group law in the group G,...
-
Brangelina Adoption Agency's general ledger shows a cash balance of $11,701. The balance of cash in the March-end bank statement is $7,349. A review of the bank statement reveals the following...
-
Define and dicuss the bureaucratic leadership model ?
-
On June 10, Sheridan Company purchased $7,850 of merchandise on account from Pronghorn Company, FOB shipping point, terms 2/10, n/30. Sheridan Company pays the freight costs of $490 on June 11. Goods...
-
In its income statement for the year ended December 31, 2025, Oriole Company reported the following condensed data. Salaries and wages expenses $409,200 Loss on disposal of plant assets $57,200 Cost...
-
Exact Photo Service purchased a new color printer at the beginning of Year 1 for $39,400. The printer is expected to have a four-year useful life and a $3,940 salvage value. The expected print...
-
How did technology affect patterns of urban life in late nineteenth-century America? Under what conditions did the urban poor live? Why did technology fail to help these people?
-
What is the mode?
-
A liquid storage facility can be modeled by where y is the liquid level (m) and u is an inlet flow rate (m 3 /s). Both are defined as deviations from the nominal steady-state values. Thus, y = u = 0...
-
Find the Laplace transforms of the following functions, using the information in Table 3.1. (However, some of the individual terms in these functions may not have Laplace transforms.) (a) f(t) = 5 +...
-
Show that the liquid-level system consisting of two interacting tanks (Fig. 6.11) exhibits over damped dynamics; that is, show that the damping coefficient in Eq. 6-57 is larger than one. di h2 hi R2...
-
From the following trial balance of G. Foot after his first year's trading, you are required to draw up a statement of profit or loss for the year ending 30 June 2016. A statement of financial...
-
At the beginning of the financial year on 1 April 2017, a company had a balance on plant account of 372,000 and on provision for depreciation of plant account of 205,400. The company's policy is to...
-
(a) Distinguish between capital and revenue expenditure. (b) Drake Ltd took delivery of a computer network on 1 July 2016, the beginning of its financial year. The list price of the equipment was...

Study smarter with the SolutionInn App



