A frictionless and leak-proof piston with a de (in kg) and negligible thickness, is in equilibrium...
Fantastic news! We've Found the answer you've been seeking!
Question:

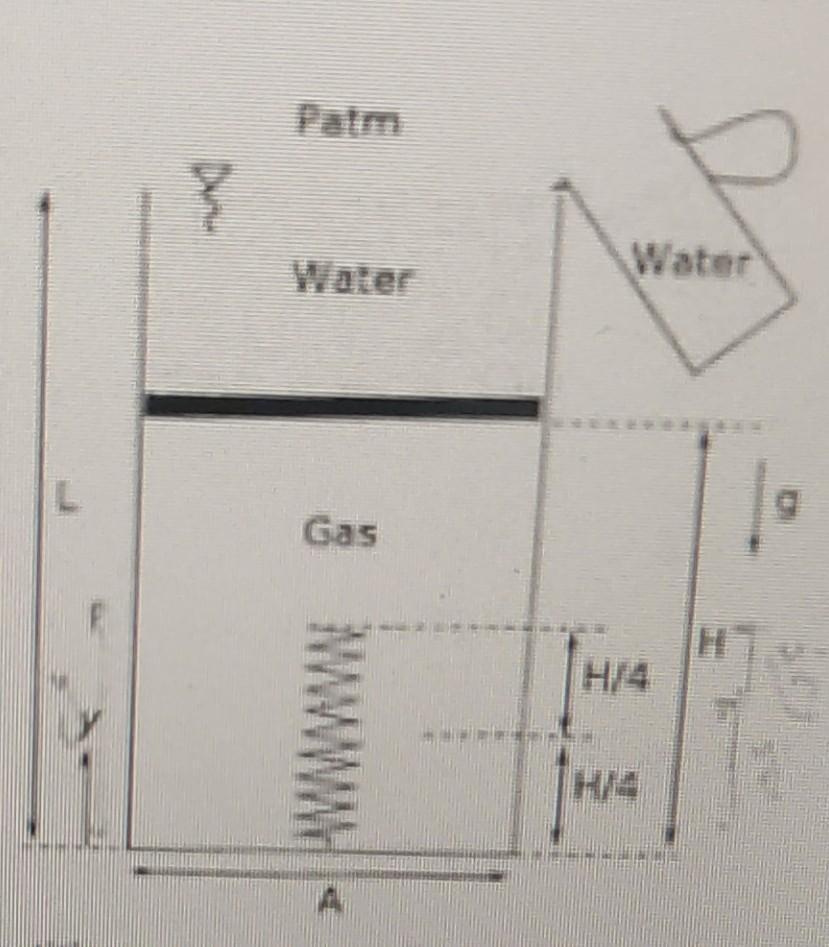
Transcribed Image Text:
A frictionless and leak-proof piston with a de (in kg) and negligible thickness, is in equilibrium as shown in the figure with gas below it and water above it till the top. The cross-sectional area of the cylinder is A (in m) and the total length is L (in m). A linear spring with a stiffness K (in N/m) is placed inside the cylinder, which is initially uncompressed, with a length of H/2 (H in meters). Water is slowly poured such that no water spills from the top of the cylinder and the piston moves down slowly to accommodate the additional water that is being added. The process of adding water is stopped when the spring is compressed by a length of H/4 Neglect the volume of the spring and take the atmospheric pressure as Patm (in Pa). Using the given variables, obtain relations for the following quantity. (a) Initial and final pressures of the gas. wh (b) Expressions for intermediate pressures of the gas bottom, for the ranges H/ 2 L D4 } Patm Water Gas H/4 H/4 A frictionless and leak-proof piston with a de (in kg) and negligible thickness, is in equilibrium as shown in the figure with gas below it and water above it till the top. The cross-sectional area of the cylinder is A (in m) and the total length is L (in m). A linear spring with a stiffness K (in N/m) is placed inside the cylinder, which is initially uncompressed, with a length of H/2 (H in meters). Water is slowly poured such that no water spills from the top of the cylinder and the piston moves down slowly to accommodate the additional water that is being added. The process of adding water is stopped when the spring is compressed by a length of H/4 Neglect the volume of the spring and take the atmospheric pressure as Patm (in Pa). Using the given variables, obtain relations for the following quantity. (a) Initial and final pressures of the gas. wh (b) Expressions for intermediate pressures of the gas bottom, for the ranges H/ 2 L D4 } Patm Water Gas H/4 H/4
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
Low Country Goods has four employees and pays them on an hourly basis. During the week beginning June 2 4 and ending June 3 0 , these employees worked the hours shown below. Information about hourly...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A piston/cylinder with cross sectional area of 0.01 m2 has a piston mass of 100 kg resting on the stops, as shown in Fig. P2.45. with an outside atmospheric pressure of 100 kPa, what should the water...
-
Last year, Lena biked 330 miles. This year, she biked k miles. Using k, write an expression for the total number of miles she biked.
-
Enter the round.html text from Figure 7.2 into a new Web page, then load the page in the browser to verify that it behaves as described. Modify the page so that instead of rounding the number to one...
-
1. What is wrong with how Gold Coast Advertising measures its quality? Explain why Gold Coast should ask its customers about how they define quality. 2. Offer suggestions to George Stein on ways of...
-
Determine the critical buckling load for the column. The column material can be assumed rigid. P 22. 2 k www A
-
Martin made an oral contract with Cresheim Garage to work as its manager for two years. Cresheim wrote Martin a letter stating that the oral contract had been made and setting forth all of its terms....
-
The financial statements for Caterpillar showed the following items for the years ending December 31, 2017, 2016, and 2015. All amounts are in millions of dollars. Income statement items: Cost of...
-
The T. Boom Pickens Corporation has a $1 million capital structure and always maintains this book value amount. Pickens currently earns $250,000 per year before taxes of 50 percent, has an all-equity...
-
Moiz Company has two products. In the past, Moiz has averaged sales of four standard models at a price of $250 and one deluxe model at a price of $750 each day. Variable costs total $75 for the...
-
In each of the following cases, what component of good internal control is being violated (if any)? 1. Tough Ltd pays all its employees minimum wages and does not have pleasant working conditions. 2....
-
Briefly explain what is meant by the statement: 'Under historic cost accounting, depreciation is a process of allocation.' Include in your answer some reference to the key assumptions that support...
-
XYZ commenced operations on 1 June 2016 selling one type of shirt. The company uses FIFO (first in, first out) and perpetual inventory control. The June inventory and sales records for the shirts...
-
Eaglehawk Ltd had the following noncurrent asset on its balance sheet on 30 June 2015. The company adopts a policy of depreciating all relevant items on a straight-line basis at an annual rate of 10...
-
For many companies, their most valuable assets are not their cash and their inventory but their intangible assets. These intangible assets may include brand names, mastheads, patents and items that...
-
Which component in a high-end gaming computer is likely to draw the most power? When you first turn on a computer and you dont hear a spinning drive or fan or see indicator lights, is the problem...
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
Air flowing at 8 psia, 480 R, and Ma1 5 2.0 is forced to undergo a compression turn of 158. Determine the Mach number, pressure, and temperature of air after the compression.
-
Consider a simple ideal Brayton cycle operating between the temperature limits of 300 and 1500 K. Using constant specific heats at room temperature, determine the pressure ratio for which the...
-
Atmospheric contaminants are often measured in parts per million (by volume). What would the partial pressure of refrigerant-134a be in atmospheric air at 100 kPa and 20oC to form a 100-ppm...
-
Using the online code for the example in this chapter as a starting point and the solution from a previous exercise. 1. Implement (or modify your previous) Student: :DropCourse (). When a student...
-
What other examples can you imagine that might easily incorporate the Factory Method pattern?
-
Using the solution from a previous exercise, augment your code as follows: 1. Implement the Factory Method pattern to create various shapes. You will have already created an abstract base class of...

Study smarter with the SolutionInn App


