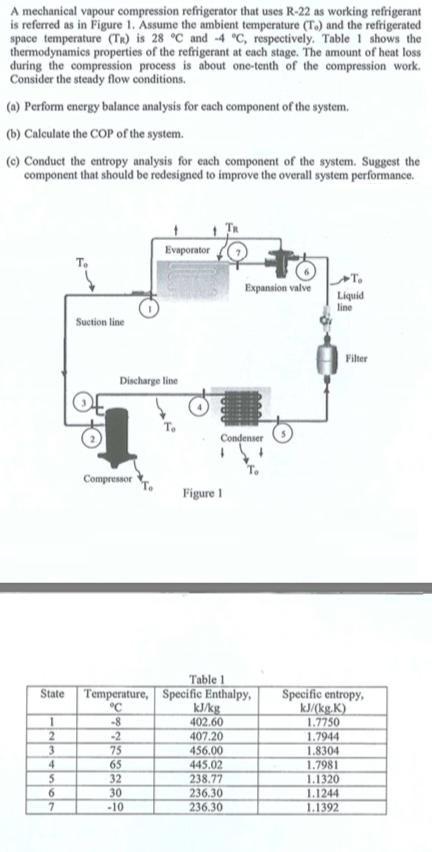
A mechanical vapour compression refrigerator that uses R-22 as working refrigerant is referred as in Figure...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A mechanical vapour compression refrigerator that uses R-22 as working refrigerant is referred as in Figure 1. Assume the ambient temperature (T.) and the refrigerated space temperature (TR) is 28 °C and -4 °C, respectively. Table 1 shows the thermodynamics properties of the refrigerant at each stage. The amount of heat loss during the compression process is about one-tenth of the compression work. Consider the steady flow conditions. (a) Perform energy balance analysis for each component of the system. (b) Calculate the COP of the system. (c) Conduct the entropy analysis for each component of the system. Suggest the component that should be redesigned to improve the overall system performance. State 1 2 3 4 5 6 7 Suction line Discharge line Compressor Evaporator 75 65 32 30 -10 Condenser Figure 1 Expansion valve Table 1 Temperature, Specific Enthalpy, °℃ -8 kJ/kg 402.60 407.20 456.00 445.02 238.77 236.30 236.30 To Liquid line Filter Specific entropy, kJ/(kg.K) 1.7750 1.7944 1.8304 1.7981 1.1320 1.1244 1.1392 A mechanical vapour compression refrigerator that uses R-22 as working refrigerant is referred as in Figure 1. Assume the ambient temperature (T.) and the refrigerated space temperature (TR) is 28 °C and -4 °C, respectively. Table 1 shows the thermodynamics properties of the refrigerant at each stage. The amount of heat loss during the compression process is about one-tenth of the compression work. Consider the steady flow conditions. (a) Perform energy balance analysis for each component of the system. (b) Calculate the COP of the system. (c) Conduct the entropy analysis for each component of the system. Suggest the component that should be redesigned to improve the overall system performance. State 1 2 3 4 5 6 7 Suction line Discharge line Compressor Evaporator 75 65 32 30 -10 Condenser Figure 1 Expansion valve Table 1 Temperature, Specific Enthalpy, °℃ -8 kJ/kg 402.60 407.20 456.00 445.02 238.77 236.30 236.30 To Liquid line Filter Specific entropy, kJ/(kg.K) 1.7750 1.7944 1.8304 1.7981 1.1320 1.1244 1.1392
Expert Answer:

Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these general management questions
-
3 4 5 6 7 8 9 10 11 12 13 A 14 Cost of the Asset 15 Life of the Asset in Years 16 Book Value of the Asset after 5 years 17 Depreciable Basis 18 Yearly depreciation 19 After tax Salvage Value in year...
-
1 2 3 4 5 6 7 cube dimension LxWxH (mm) Surface area (mm^2) volume (mm^3) SA/ V Volume undiffused- white (mm^3) Volume diffused % total volume diffused a) 15x15x15 1350 3375 0.4 0 3375 100% b)...
-
2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 ARASIN22227 17 18 19 20 21 23 24 25 26 1) (2) Sales Price per unit Variable Cost per unit Contribution per unit Contribution Margin Ratio Break Even Point Sales...
-
In a recent year, the total scores for a certain standardized test were normally distributed, with a mean of 500 and a standard deviation of 10.4. Answer parts (a)-(d) below. (a) Find the probability...
-
It has been said that a system without good documentation is worthless. Provide support for this statement. Then comment on how todays advanced tools might alleviate the documentation burden.
-
The radius of a circle is increasing. At a certain instant, the rate of increase of the area of the circle is three times the rate of increase of its circumference. What is the radius of the circle...
-
Do you think it is possible to have win-win negotiations? Why or why not?
-
Bloom Company management predicts that it will incur fixed costs of $160,000 and earn pretax income of $164,000 in the next period. Its expected contribution margin ratio is 25%. Use this information...
-
Mountain Air Limited manufactures a line of room air purifiers. Management is currently evaluating the possible production of an air purifier for automobiles. Based on an annual volume of 1 0 , 0 0 0...
-
A string of length 0.80 m is fixed at both ends. The diagram shows a standing wave formed on the string. P and Q are two particles on the string. 0.8 The variation with time t of the displacement of...
-
1) Graded Question Consider a solution that is made from mixing 0.053 moles of A* and 0.087 moles of 8 in 125.0mL of water, if the AB solid formed has Kip 8.98x10, what is the concentration of...
-
Do you believe that the "ruling class" (decide for yourselves who these people may be) unfairly pass laws favorable to themselves and detrimental to the rest of us? If they do, what can we do about...
-
Which type of preliminary estimate would be the best in each of the following situations, and why did you choose it? a. Decide whether it is feasible to proceed with constructing a new small office...
-
If you were a local elected official and could choose whether to fund a community court, a homeless court, a mental health court, veterans court, or a reentry court, which would you choose? Why?
-
In this zone, we critically evaluate the shadow side of organisations and examine whether the covert, informal aspects of organisational behaviour enable or hinder the completion of day-to-day...
-
Why did arrests for domestic violence increase following the implementation of the Lexington County (South Carolina) Domestic Violence Court?
-
Explain how covid-19 affected the transportation system in South Africa.?
-
General Electric Capital, a division of General Electric, uses long-term debt extensively. In a recent year, GE Capital issued $11 billion in long-term debt to investors, then within days filed legal...
-
A 40 kg aluminum block at 90oC is dropped into an insulated tank that contains 0.5 m3 of liquid water at 20oC. Determine the irreversibility in the resulting process if the surrounding temperature is...
-
A chamber initially contains a gaseous mixture consisting of 4 kmol of CO2, 8 kmol of CO and 2 kmol of H2. Assume an equilibrium mixture formed consists of CO2, CO, H2O, H2 and O2 at 2600 K and 100...
-
Steam enters a turbine with a pressure of 3 MPa, a temperature of 400oC and a velocity of 140 m/s. Steam exits as saturated vapor at 100oC with a velocity of 105 m/s. At steady state, the turbine...
-
A sample contains radioactive atoms of two types, A and B. Initially there are five times as many A atoms as there are B atoms. Two hours later, the numbers of the two atoms are equal. The half-life...
-
About \(12 \%\) of your body mass is carbon; some of this is radioactive \({ }^{14} \mathrm{C}\), a beta-emitter. If you absorb \(100 \%\) of the\(49 \mathrm{keV}\) energy of each \({ }^{14}...
-
You are assisting in an anthropology lab over the summer by carrying out \({ }^{14} \mathrm{C}\) dating. \(\mathrm{A}\) graduate student found a bone he believes to be 20,000 years old. You extract...

Study smarter with the SolutionInn App


