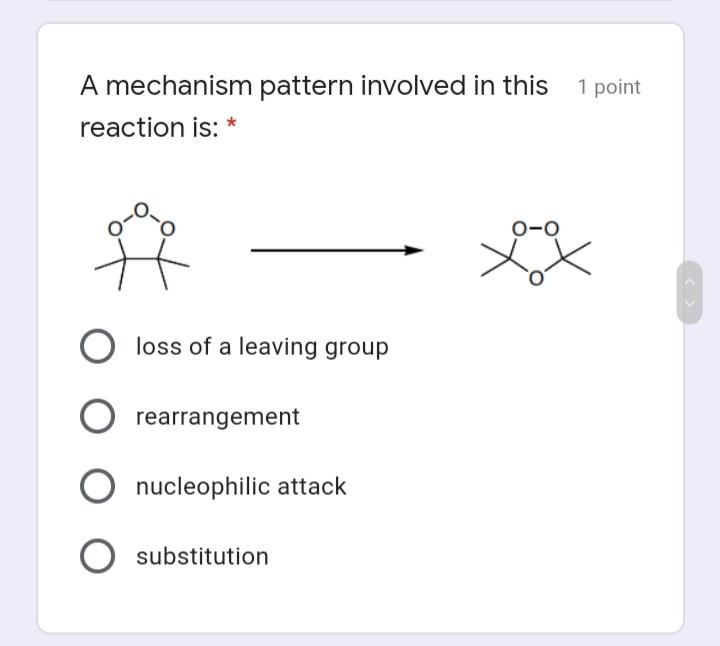
A mechanism pattern involved in this 1 point reaction is: * loss of a leaving group...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
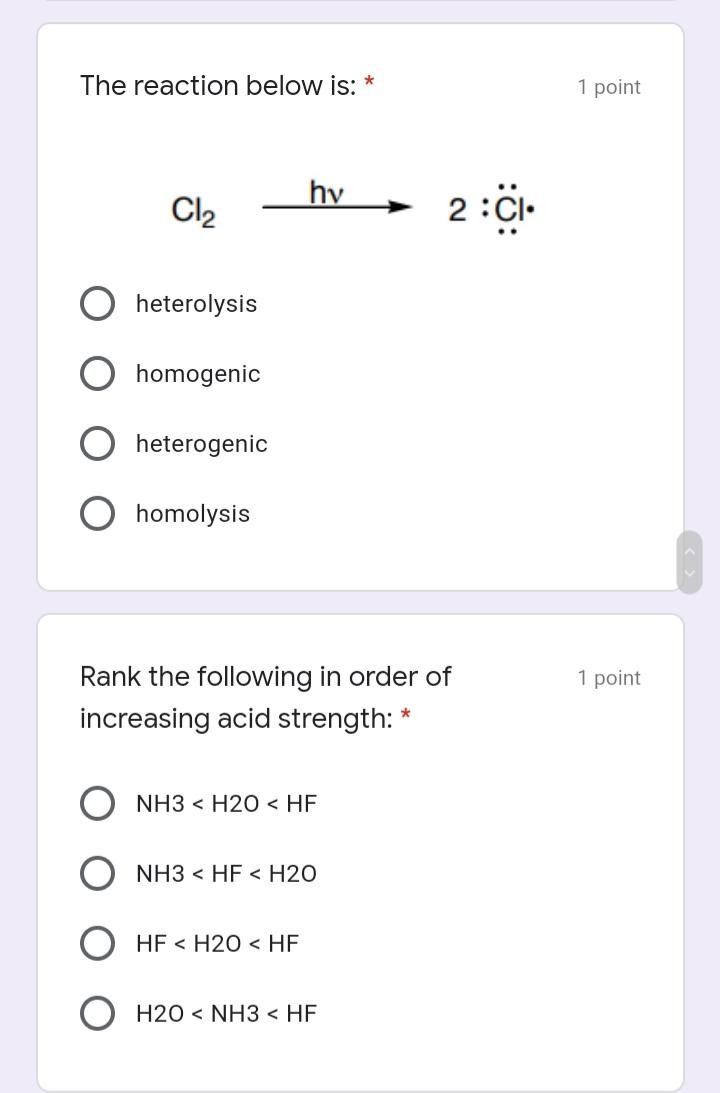
A mechanism pattern involved in this 1 point reaction is: * loss of a leaving group O rearrangement O nucleophilic attack substitution The reaction below is: 1 point hv Cl2 2 :CI- heterolysis homogenic heterogenic homolysis Rank the following in order of 1 point increasing acid strength: * NH3 < H20 < HF NH3 < HF < H2O HF < H20 < HF H20 < NH3 < HF A mechanism pattern involved in this 1 point reaction is: * loss of a leaving group O rearrangement O nucleophilic attack substitution The reaction below is: 1 point hv Cl2 2 :CI- heterolysis homogenic heterogenic homolysis Rank the following in order of 1 point increasing acid strength: * NH3 < H20 < HF NH3 < HF < H2O HF < H20 < HF H20 < NH3 < HF
Expert Answer:
Answer rating: 100% (QA)
1 Rearrangement It is formed in Ozonolysis of alkeneconversion of molozonid... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Rank each group of radicals in order of increasing stability: (a) (b)
-
Rank each group of compounds in order of increasing heat of hydrogenation. (a) hexa-1, 2-diene; hexa-1, 3, 5-triene; hexa-1, 3-diene; hexa-1, 4-diene; hexa-1, 5-diene; hexa-2, 4-diene. (b)
-
Rank the following five compounds in order of increasing acid strength: 2-chloroethanol, p-chlorophenol, p-methylphenol, ethanol, and phenol.
-
Find the critical points of the function. Then use the Second Derivative Test to determine whether they are local minima, local maxima, or saddle points (or state that the test fails). f(x, y) = x -...
-
For each of the following independent cases, calculate the cash flow from operations: Case I Case I Case III $380,000 $575,000 $936,000 210,000 330,000 620,000 65,000 95,000 105,000 18,000 28.000...
-
Access and refer to the December 31, 2013, annual report for Samsung at www.samsung.com/us/aboutsamsung/investor_relations/financial_information/financial_statement.html. Required 1. Identify its...
-
The second law of thermodynamics states that (a) The energy change of a system undergoing any reversible process is zero (b) It is not possible to transfer heat from a lower temperature to a higher...
-
Listed below is the number of movie tickets sold at the Library Cinema-Complex, in thousands, for the period from 2001 to 2013. Compute a five-year weighted moving average using weights of .1, .1,...
-
What are the the differences between a Micro-manager and an Investor. Provide examples of each.
-
The following scenarios might constitute a violation of the AICPA's Code of Professional Conduct. Carrie Jones, CPA, performs various management services for The Perry Corporation including...
-
What methodologies does the researcher employ to discern the multifaceted ramifications of climate change on indigenous communities, and how do these findings inform broader environmental policy...
-
Based only on the material found in our textbook, please answer the following questions: (1) Describe and discuss the performance evaluation process. (2) What is standard deviation, the Sharpe ratio,...
-
Select one specific financial institution or financial market and relate the history in that area to the development of current regulations. Cover the major legislations that resulted and how...
-
How much must you invest today at 10% interest in order to see your investment grow to $8,000 in 3 years? To save for a new car, Samuel will invest $18,000 at the end of each year for the next 5...
-
Consider the CAPM. The expected return market is 20%. The expected return on a stock with a beta of 1.2 is 22%. What is the risk free rate?
-
F is an all-equity firm with assets worth $1Mns. Expected returns on assets equal to 9%. F buys new assets at market value for $0.5Mns (same risk as existing assets). The acquisition is financed by...
-
assuming ppp ratio is .40 provide a balance sheet for an expat from USA to India assuming the salary in USA is $3500
-
What are the four types of poultry production systems? Explain each type.
-
Table 18.14 lists some common nitrogen compounds having oxidation states ranging from 23 to 15. Rationalize this spread in oxidation states. Table 18.14 Oxidation State of Nitrogen Compound Formula...
-
Use the diagram of the unit cell for the hexagonal closest packed structure in Fig. 16.14 to determine the net number of atoms in the hcp unit cell. Figure 16.14. Side view Top view Unit cell Atom in...
-
Arrange the atoms in Exercise 97 in order of increasing first ionization energy. Atoms in Exercise 97 a. Te, S, Se d. Rb, Na, Be b. K, Br, Ni e. Sr, Se, Ne c. Ba, Si, F f. Fe, P, O
-
The reciprocating mass, crank radius, and connecting-rod length of each of the cylinders in a two-cylinder in-line engine are given by \(m, r\), and \(l\), respectively. The crank angles of the two...
-
The force transmitted by an internal combustion engine of mass \(500 \mathrm{~kg}\), when placed directly on a rigid floor, is given by \[F_{t}(t)=(18,000 \cos 300 t+3600 \cos 600 t) \mathrm{N}\]...
-
A shaft, having a stiffness of \(3.75 \mathrm{MN} / \mathrm{m}\), rotates at \(3600 \mathrm{rpm}\). A rotor, having a mass of \(60 \mathrm{~kg}\) and an eccentricity of 2000 microns, is mounted on...

Study smarter with the SolutionInn App


