?? A medical technician is working with the four samples of radionuclides listed in the table below.
Fantastic news! We've Found the answer you've been seeking!
Question:

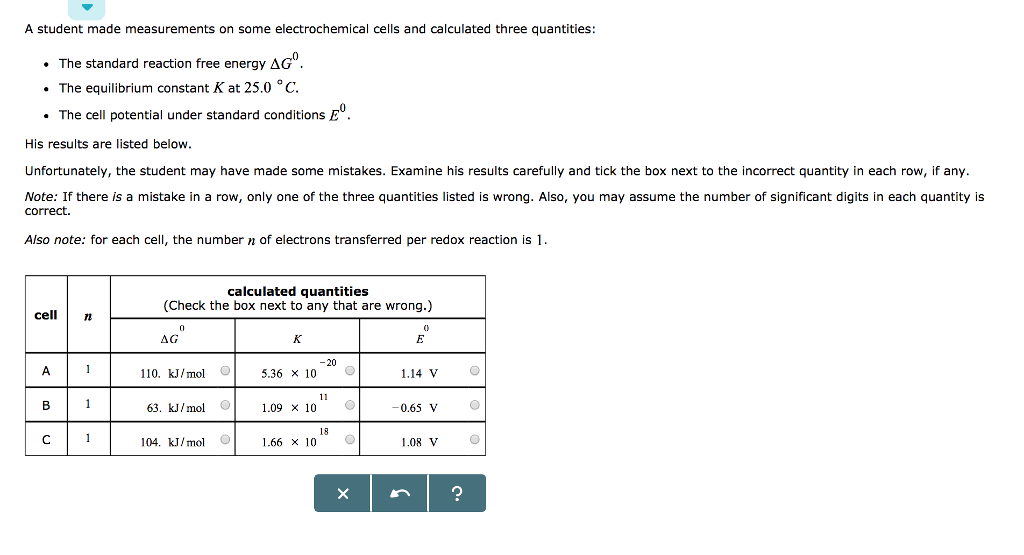
Transcribed Image Text:
A medical technician is working with the four samples of radionuclides listed in the table below. Initially, each sample contains 5.00 μmol of the radionuclide. First, order the samples by decreasing initial radioactivity. Then calculate how long will take for the amount of radionuclide in each sample to decrease to 1/32 of the initial amount. sample A B с D symbol 152 63 Eu 99 42 Mo 82 37 Rb radionuclide 192 77 Ir half-life 13 years. 66.0 hours 1 minute 74.0 days initial radioactivity (choose one) (choose one) + (choose one) # (choose one) time for amount of radionuclide to decrease to 1/32 of Initial amount X years hours minutes days ? A student made measurements on some electrochemical cells and calculated three quantities: • The standard reaction free energy AG⁰. The equilibrium constant K at 25.0 °C. • The cell potential under standard conditions º His results are listed below. Unfortunately, the student may have made some mistakes. Examine his results carefully and tick the box next to the incorrect quantity in each row, if any. Note: If there is a mistake in a row, only one of the three quantities listed is wrong. Also, you may assume the number of significant digits in each quantity is correct. Also note: for each cell, the number of electrons transferred per redox reaction is 1. cell 72 A B с 1 1 1 calculated quantities (Check the box next to any that are wrong.) AG 0 110. kJ/mol O 63. kJ/mol 104. kJ/mol K -20 5.36 x 10 C 1.09 x 10 11 18 1.66 x 10. O O X E 1.14 V -0.65 V 1.08 V ? O O O A medical technician is working with the four samples of radionuclides listed in the table below. Initially, each sample contains 5.00 μmol of the radionuclide. First, order the samples by decreasing initial radioactivity. Then calculate how long will take for the amount of radionuclide in each sample to decrease to 1/32 of the initial amount. sample A B с D symbol 152 63 Eu 99 42 Mo 82 37 Rb radionuclide 192 77 Ir half-life 13 years. 66.0 hours 1 minute 74.0 days initial radioactivity (choose one) (choose one) + (choose one) # (choose one) time for amount of radionuclide to decrease to 1/32 of Initial amount X years hours minutes days ? A student made measurements on some electrochemical cells and calculated three quantities: • The standard reaction free energy AG⁰. The equilibrium constant K at 25.0 °C. • The cell potential under standard conditions º His results are listed below. Unfortunately, the student may have made some mistakes. Examine his results carefully and tick the box next to the incorrect quantity in each row, if any. Note: If there is a mistake in a row, only one of the three quantities listed is wrong. Also, you may assume the number of significant digits in each quantity is correct. Also note: for each cell, the number of electrons transferred per redox reaction is 1. cell 72 A B с 1 1 1 calculated quantities (Check the box next to any that are wrong.) AG 0 110. kJ/mol O 63. kJ/mol 104. kJ/mol K -20 5.36 x 10 C 1.09 x 10 11 18 1.66 x 10. O O X E 1.14 V -0.65 V 1.08 V ? O O O
Expert Answer:
Answer rating: 100% (QA)
Lower the half life higher will be initial radioactivity so initial radio activity Samp... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
The carbon steels listed in the table below were soaked at 1000 C for 1 hour to form austenite and were cooled slowly, under equilibrium conditions to room temperature. Refer to the Fe-Fe3C phase...
-
Lets figure out how long it will take for the average Indian to be as wealthy as the average Western European is today. All numbers are adjusting for inflation, so were measuring output in piles of...
-
Mia Salto wishes to determine how long it will take to repay a loan with initial proceeds of $14,000 where annual end-of-year installment payments of $2,450 are required. a. If Mia can borrow at a...
-
Write the C++ code for a function that receives an integer, a double number, and the address of a double variable from the calling statement. The function should multiply the integer by the double...
-
What is a waiver related to debt covenants, and how does it affect the auditors work?
-
A 50,000-kg barge is pulled along a canal at a constant speed of 3 km/h by a heavy tractor. The towrope makes an angle of 18 with the velocity vector of the barge. The tension in the towrope is 1200...
-
On April 1, Topeka Brake Mfg. purchased new production scheduling software for \(\$ 480,000\). On May 15 , a representative of a computerized manufacturing technology company demonstrated new...
-
Sally Stanford is buying an automobile that costs $12,000. She will pay $2000 immediately and the remaining $10,000 in four annual end-of-year principal payments of $2500 each. In addition to the...
-
33 For the given data of Investments (in Rs. Lakhs) and Profits (in Rs. Lakhs): a) Draw scatter diagram 2023/12/23-2023 b) Find the coefficient of correlation between investments and profits and...
-
Coca Cola (KO) price is $61/share. The company is expected to pay dividend of $1.7/share next year. (In reality, dividends are paid quarterly. In this question we will assume for simplicity that all...
-
Balance Sheet 2023 2024 2023 2024 Assets Liabilities and equity Cash 58$ 31$ Current liabilities 56$ 106$ Other current assets 222 259 Long-term debt 102 112 Net fixed assets 236 198 Stockholder's...
-
As it relates to productive resource deployment within a society, define the difference between a socialistic and a capitalistic method of making these determinations.
-
Simplify 1 14 1 4x 2x 1 5 3y 6y
-
Solve for x. log 3(2x-6)=1 Select the correct choice below and, if necessary, fill in the answer box to complete your choice. OA. The solution is x = (Type an exact answer in simplified form. Use a...
-
Several health care advocacy groups are pushing to mandate higher staffing levels in assisted living facilities. What would be the impact on the labor market for care givers in assisted living...
-
A paragraph with the following topical sentence: "Aquinas's discussion of commercial fraud is concerned with sin not efficiency." St. Thomas Aquinas, "On Fraud Committed in Buying and Selling,"...
-
Discuss the appearance, fresh and quality indicators and combination of ingredients for the following sandwiches. First, one has been done for you. San Appearance - Freshness dwi colour, contrast,...
-
Figure displays a 12.0 V battery 3 four uncharged capacitors of capacitances C1 = 4.00F, C2 = 6.00F, and C3 = 3.00F. The switch is thrown to the left side until capacitor 1 is fully charged. Then the...
-
The Rock Creek Company has an ending inventory valuation using FIFO of \($350,000\). Had the company used LIFO the inventory would have been valued at \($320,000\). The company has determined the...
-
Walgreens Boots Alliance, Inc. (Walgreens) is a global pharmacy, health and well-being company. It operates over 13,000 stores in 11 countries, generating net sales of \($103.4\) billion. The...
-
The comparative financial statements for CyberOptic Corporation are in the Working Papers. The financial statements have been completed up to the trend analysis section. The following information is...

Study smarter with the SolutionInn App

