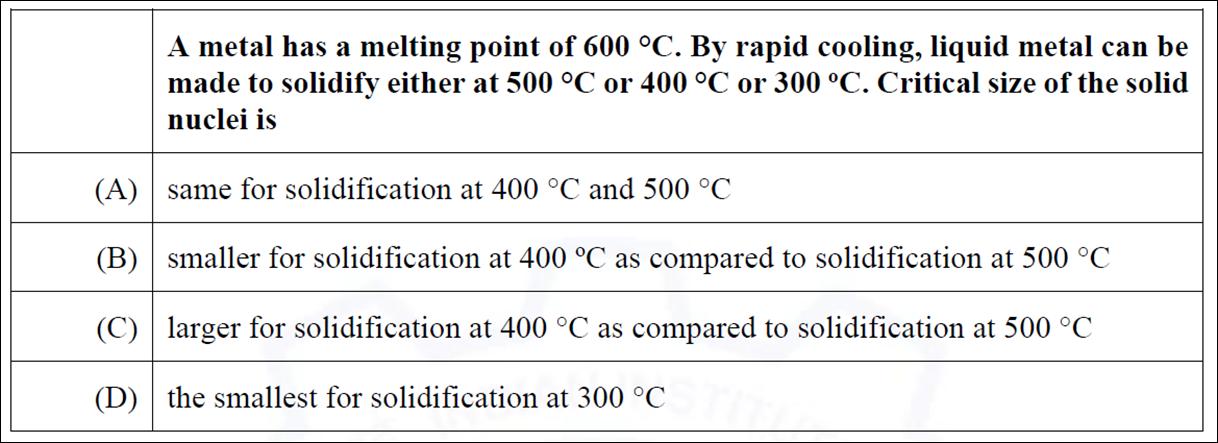
A metal has a melting point of 600 C. By rapid cooling, liquid metal can be...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A metal has a melting point of 600 °C. By rapid cooling, liquid metal can be made to solidify either at 500 °C or 400 °C or 300 °C. Critical size of the solid nuclei is (A) same for solidification at 400 °C and 500 °℃ (B) smaller for solidification at 400 °C as compared to solidification at 500 °C (C) larger for solidification at 400 °C as compared to solidification at 500 °C (D) the smallest for solidification at 300 °C A metal has a melting point of 600 °C. By rapid cooling, liquid metal can be made to solidify either at 500 °C or 400 °C or 300 °C. Critical size of the solid nuclei is (A) same for solidification at 400 °C and 500 °℃ (B) smaller for solidification at 400 °C as compared to solidification at 500 °C (C) larger for solidification at 400 °C as compared to solidification at 500 °C (D) the smallest for solidification at 300 °C
Expert Answer:
Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these chemical engineering questions
-
If copper (which has a melting point of 1085C) homogeneously nucleates at 849C, calculate the critical radius given values of 1.77 109 J/m3 and 0.200 J/m2, respectively, for the latent heat of...
-
If copper (which has a melting point of 1085C) homogeneously nucleates at 849C, calculate the critical radius given values of - 1.77 109 J/m3 and 0.200 J/m2, respectively, for the latent heat of...
-
Tristearin has a melting point of 72 C. Based on this information, would you expect triarachadin to be classified as a fat or as an oil?
-
After examining all the potential projects, you discover that there are many more projects this year with positive NPVs than in a normal year. What two problems might this extra large capital budget...
-
An X-ray photon of wavelength 6 pm that collides with an electron is scattered by an angle of 90o. (a) What is the change in wavelength of the photon? (b) What is the kinetic energy of the scattered...
-
During 2013, Will gives $40,000 cash to Will, Jr. and a remainder interest in a few acres of land to his friend Suzy. The remainder interest is valued at $32,000. Will and his wife, Helen, elect gift...
-
Compare and contrast the positive and negative aspects of both anger and empathy as common emotions that people may display at work.
-
Decide whether each situation is or is not a symptom of revenue fraud. Then explain (1) Why the situation is or is not a symptom and (2) If it is a symptom, how it would be found using computer...
-
Company X enters into a forward contract to hedge against the foreign currency risk associated with a future purchase of equipment costing 100,000. The forward contract specifies a forward rate of...
-
A periodic signal x[n] of fundamental period N can be represented by its Fourier series If you consider this a representation of x[n] (a) Is x 1 [n] = x[n N 0 ] for any value of N 0 periodic? If so...
-
Create new JavaFX project called usernamePart3 in NetBeans. For this part, you need to build an app that stores a collection of Sculptures for a small exhibition (see the attached class). Your app...
-
Your firm is about to begin exporting. In selecting an export intermediary, what characteristics would you look for?
-
What conditions must exist for an intra-corporate transfer to be cost-effective?
-
Describe the five major organizations governing the EU.
-
Why is exporting the most popular initial entry mode?
-
Under what conditions should a firm consider a greenfield strategy for FDI? An acquisition strategy?
-
Look at the attachment. Need for practice problem reference. Part 2: Flynn, Inc. has two temporary differences at the end of 20X2. The first difference stems from installment sales, and the second...
-
Cornell and Roberts are partners who agree to admit Stanley to their partnership. Cornell has a capital balance of $80,000 and Roberts has a capital balance of $120,000. Cornell and Roberts share net...
-
A thin wire with positive charge evenly spread along its length is shaped into a semicircle. What is the direction of the electric field at the center of curvature of the semicircle? Explain.
-
A child swings a rock of mass m in a horizontal circle using a rope of length L. The rock moves at constant speed v. (a) Ignoring gravity, find the tension in the rope. (b) Now include gravity (the...
-
A 0.15-kg baseball traveling in a horizontal direction with a speed of 20 m/s hits a bat and is popped straight up with a speed of 15 m/s. (a) What is the change in momentum (magnitude and direction)...
-
SnoBoard Companys year-end balance in its Allowance for Doubtful Accounts is a credit of $440. By aging accounts receivable, it estimates that $6,142 is uncollectible. Prepare SnoBoards year-end...
-
Calculate the amount recorded as the cost of a new machine given the following payments related to its purchase: gross purchase price, $700,000; sales tax, $49,000; purchase discount taken, $21,000;...
-
Wecker Companys year-end unadjusted trial balance shows accounts receivable of $89,000, allowance for doubtful accounts of $500 (credit), and sales of $270,000. Uncollectibles are estimated to be...

Study smarter with the SolutionInn App



