A mixture of two ideal gases X and Y has n, and n, in the ratio...
Fantastic news! We've Found the answer you've been seeking!
Question:
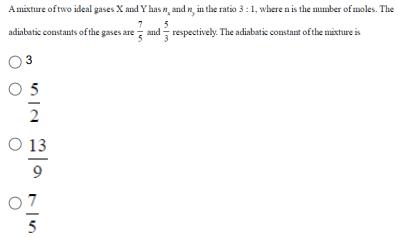
Transcribed Image Text:
A mixture of two ideal gases X and Y has n, and n, in the ratio 3:1, where n is the number of moles. The 7 5 adiabatic constants of the gases are t and respectively. The adiabatic constant of the mixture is 03 546555 O 13 A mixture of two ideal gases X and Y has n, and n, in the ratio 3:1, where n is the number of moles. The 7 5 adiabatic constants of the gases are t and respectively. The adiabatic constant of the mixture is 03 546555 O 13
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
Given the following data, use least-squares regression to derive a trend equation. What is your estimate of the demand in period 7? In period 12? PERIOD DEMAND 1 7 2 9 m 5 4 11 5 10 6 13
-
Two gases X (molecular weight Mx) and Y (molecular weight My; My > Mx) are at the same temperature T in two different containers. Their root mean square velocities are Cx and Cy respectively. If the...
-
A container has a mixture of two gases: n1 mol of gas 1 having molar specific heat C1 and n2 mol of gas 2 of molar specific heat C2. (a) Find the molar specific heat of the mixture. (b) What If? What...
-
A test rocket is launched by accelerating it along a 200.0-m incline at 1.25 m/s2 starting from rest at point A (Figure 3.45.) The incline rises at 35.0o above the horizontal, and at the instant the...
-
An object of mass M 1 = m collides with velocity v 0 i into an object of mass M 2 = 2m with velocity 1/2v 0 j. Following the collision, the mass m 2 has a velocity v 0 /4 i. (a) Determine the...
-
For the situation considered in Problem 12.5, what is the value of a one-year European put option with a strike price of $100? Verify that the European call and European put prices satisfy putcall...
-
Tanisha Greggs, a stockholder in Mentar Corp., has just received a report of the companys ten-year financial performance that includes the following graph. In the report, management states that the...
-
A household appliance dealer buys microwave ovens from a manufacturer and resells them to its customers. a. The manufacturer sets a list or catalogue price of $1,500 for a microwave. The manufacturer...
-
On October 10, the stockholders' equity section of Sherman Systems appears as follows. Common stock-$10 par value, 76,000 shares authorized, issued, and outstanding Paid-in capital in excess of par...
-
Fulcrum Industries manufactures dining chairs and tables. The following information is available: Dining Chairs Tables Total Cost 600 470 2,400 Machine setups Inspections Labor hours Problem 2. 200...
-
the following questions 1) Looking at Apple's supply chain, what differences set it apart from competitors? 2) What are Apple's key advantages in how it manages its supply chain operations (use data...
-
Janes Company providedthe following information on intangible assets: A patent was purchased from the Lou Company for $1,100,000 onJanuary 1, 2016. Janes estimated the remaining useful life of...
-
Pam Corporation holds 70 percent ownership of NorthernEnterprises. On December 31, 20X6, Northern paid Pam $31,000 for atruck that Pam had purchased for $36,000 on January 1, 20X2. Thetruck was...
-
The following information is provided for the first month of operations for Legal Services Inc.: Prepare a T-accounts to reflect the following business transaction: Legal Services, Inc. received the...
-
STU Company expects $2,000,000 of FCF next year. Growth is expected to be 3% per year, and the WACC is 7%. If the company has no debt or preferred stock and 2 million shares of common stock...
-
Sakelaris Corporation makes a product with the following standard costs: Direct materials Standard Quantity or Hours 8.6 kilos 0.4 hours Standard Price or Rate $ 6.00 per kilo $ 11.00 per hour $5.00...
-
3) A beam of square cross section of the dimensions shown below is subjected to a shear force of V = 8 kN. (a) Calculate the average shear stress at the neutral axis (y' = 0), (b) at y' = 0.01 m and...
-
At the beginning of the year, Lam Ltd. had total assets of $800,000 and total liabilities of $500,000. Use this information to answer each of the following independent questions. (a) If Lam's total...
-
From information in the Data section, calculate the standard Gibbs energy and the equilibrium constant at (a) 25C (b) 50C for the reaction CH4(g) + 3 C12(g) ='0CHC13(l) + 3 HCl(g). Assume that the...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
The isothermal compressibility of lead at 293 K is 2.21 X 10-6 atm-1, Calculate the pressure that must be applied in order to increase its density by 0.08 per cent.
-
For each of the following organizational units, indicate whether the unit would most likely be classified as a cost center \((C)\), a revenue center \((R)\), a profit center \((P)\), or an investment...
-
Premiere Produce Inc. summarized the following results for its most recent fiscal year: Based on the sales information, compute the company's sales price variance and sales volume variance. Budgeted...
-
Management at Consulting Inc. has decided to allocate costs of the company's two support departments (administration and information technology) to the two revenue-generating consulting departments...

Study smarter with the SolutionInn App



