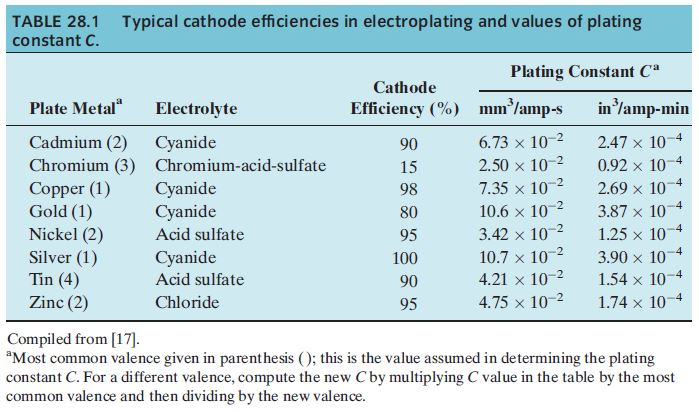
TABLE 28.1 Typical cathode efficiencies in electroplating and values of plating constant C. Plate Metal Cadmium...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
TABLE 28.1 Typical cathode efficiencies in electroplating and values of plating constant C. Plate Metal" Cadmium (2) Chromium (3) Copper (1) Gold (1) Nickel (2) Silver (1) Tin (4) Zinc (2) Electrolyte Cyanide Chromium-acid-sulfate Cyanide Cyanide Acid sulfate Cyanide Acid sulfate Chloride Cathode Efficiency (%) 90 15 98 80 95 100 90 95 Plating Constant Ca mm²/amp-s -2 6.73 x 10-2 2.50 x 10-² 7.35 x 10-2 10.6 x 10-2 3.42 x 10-² 10.7 x 10-2 4.21 x 10-² 4.75 x 10-² in ³/amp-min -4 2.47 x 10- 0.92 x 101 2.69 x 101 3.87 x 107 1.25 x 107 3.90 x 107 1.54 x 107 1.74 x 107 4 4 Compiled from [17]. "Most common valence given in parenthesis (); this is the value assumed in determining the plating constant C. For a different valence, compute the new Cby multiplying C value in the table by the most common valence and then dividing by the new valence. TABLE 28.1 Typical cathode efficiencies in electroplating and values of plating constant C. Plate Metal" Cadmium (2) Chromium (3) Copper (1) Gold (1) Nickel (2) Silver (1) Tin (4) Zinc (2) Electrolyte Cyanide Chromium-acid-sulfate Cyanide Cyanide Acid sulfate Cyanide Acid sulfate Chloride Cathode Efficiency (%) 90 15 98 80 95 100 90 95 Plating Constant Ca mm²/amp-s -2 6.73 x 10-2 2.50 x 10-² 7.35 x 10-2 10.6 x 10-2 3.42 x 10-² 10.7 x 10-2 4.21 x 10-² 4.75 x 10-² in ³/amp-min -4 2.47 x 10- 0.92 x 101 2.69 x 101 3.87 x 107 1.25 x 107 3.90 x 107 1.54 x 107 1.74 x 107 4 4 Compiled from [17]. "Most common valence given in parenthesis (); this is the value assumed in determining the plating constant C. For a different valence, compute the new Cby multiplying C value in the table by the most common valence and then dividing by the new valence.
Expert Answer:

Related Book For 

Materials Science and Engineering An Introduction
ISBN: 978-0470419977
8th edition
Authors: William D. Callister Jr., David G. Rethwisch
Posted Date:
Students also viewed these mechanical engineering questions
-
A uniform piece of sheet steel is shaped as in Figure P9.41. Compute the x and y coordinates of the center of mass of the piece. y(cm) 30 20 10 x(cm) 10 20 30 Figure P9.41
-
A cylindrical piece of 4140 steel is to be austenitized and quenched in moderately agitated oil. If the microstructure is to consist of at least 50% martensite throughout the entire piece, what is...
-
A cylindrical piece of 8640 steel is to be austenitized and quenched in moderately agitated oil. If the hardness at the surface of the piece must be at least 49 HRC, what is the maximum allowable...
-
Which of the following activity bases would best be used to allocate setup activity to products? a. Number of inspections b. Direct labor hours c. Direct machine hours d. Number of production runs
-
Should prototyping be used on every systems development project? Why or why not?
-
Jaleh Mehr is the owner of the retail store 151 Jeans. She purchases jeans from a number of manufacturers to bring great style and fit to her customers. Prepare journal entries for March 2023 to...
-
The following balance sheet and income statement data were taken from the financial records of The Dana Point Company: Required 1. Calculate the following ratios for The Dana Point Company for 2015...
-
Rocky owns and operates Balboas Gym located in Philadelphia. The following transactions occur for the month of October: October 2 Receive membership dues for the month of October totaling $8,500....
-
NB: Do NOT wait until the last date provided before you complete the assignment on myUnisa, as the system might be very busy or down for routine maintenance. Required: This assignment consists of...
-
Table B. 4 presents data for 27 houses sold in Erie, Pennsylvania. a. Fit a simple linear regression model relating selling price of the house to the current taxes $\left(x_{1} ight)$. b. Test for...
-
How are melting point and boiling point quantitative properties of a substance? Give examples of how they may be used when describing a substance's quantitative property. For instance, viscosity is a...
-
Willets Coffee Equipment sells European style coffee makers and uses a periodic inventory system. Its inventory records show that at July 1, Willets had 12 units on hand at a cost of $220 each....
-
A memory system has the following performance characteristics: Cache Tag Check time: 1ns Cache Read Time: 1ns Cache Line Size: 64 bytes Memory Access time (Time to start memory operation): 10 ns...
-
You have reached the end of your work with clients, the end of your first year internship, and the end of our class. Reflect on all of these and the breadth of your feelings, thoughts, and...
-
Sufficient audit evidence gathered by the auditor involves which of the following? A) B. The type of evidence obtained B A. The quantity of evidence to be obtained C D. The use of an audit program to...
-
Determine the maximum contribution that can be made to a Keogh plan in each of the following cases. In all instances, the individual is self-employed, and the self-employment tax reduction has...
-
Edward Strong compared the test results of military personnel regarding occupational likes and dislikes with which population who took the same test? a. civilians b. high school graduates c. college...
-
For the following exercises, write the polynomial function that models the given situation. Consider the same rectangle of the preceding problem. Squares of 2x by 2x units are cut out of each corner....
-
If cupric oxide (CuO) is exposed to reducing atmospheres at elevated temperatures, some of the Cu2+ ions will become Cu+. (a) Under these conditions, name one crystalline defect that you would expect...
-
Demonstrate, in a manner similar to Equation 15.4, how vulcanization may occur in a butadiene rubber.
-
Within a cubic unit cell, sketch the following directions: (a) , [1 10] (b) , [1 2 1] (c) , [01 2] (d) , [13 3] (e) , [1 1 1] (f) , [1 22] (g) , [12 3 ] (h) . [1 03]
-
The 2015 annual report of the Procter & Gamble Company (P&G) is available at www.pginvestor.com. After reviewing P&Gs annual report, respond to the following questions: a. When does P&G recognize...
-
Consider a publicly held company whose products you are familiar with. Some examples might include: Access the companys public website and search for its most recent annual report. (Some companies...
-
The 2014 financial statements of LVMH Moet Hennessey-Louis Vuitton S.A.LVMH is a Paris-based holding company and one of the worlds largest and best-known luxury goods companies. As a member-nation of...

Study smarter with the SolutionInn App


