A process system for the manufacture of ethylene glycol is shown in Figure Q1. Ethylene oxide...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
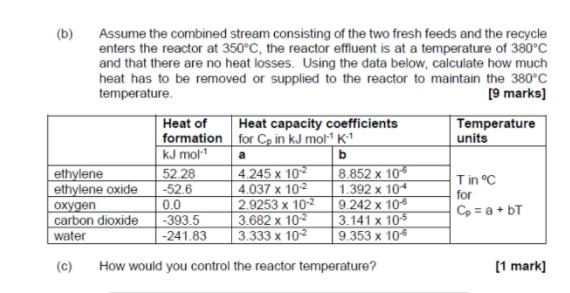
A process system for the manufacture of ethylene glycol is shown in Figure Q1. Ethylene oxide (CaH.O) is produced in the reactor by the following main reaction of catalytic oxidation of ethylene (C2H«): (6)O'HO- (6)o (6)"H) An undesired competing side reaction is the combustion of ethylene to CO2. CaHa(g) + 302(g)- 2CO2 (g) + 2H:O(g) The reactor product is fed to a separation system where water is added to convert ethylene oxide to ethylene glycol. CH,O(g) + H20(1)CaHO2(1) recycle: n, purge: n. water: n. ethylene: net oxygen: 10 kmol/h T 350°C T= 380 °C Separation System Preheater Reactor Q=? ethylene glycol product: neg n, - molar flow rate of x, kmol/h Figure Q1 (a) Produce a process material balance for the production of ethylene glycol based on an oxygen feed rate of 10 kmol h-1. Calculate the molar flowrates of each component in each stream. The following conditions apply: • All oxygen reacts, 90% goes to ethylene oxide, the rest to CO2 and H20. • Ethylene and oxygen feeds are pure. • 20% excess ethylene is fed to the process to ensure process safety. • No reaction of ethylene oxide with water takes place in the reactor. • All the water and ethylene oxide entering the separator is converted to • ethylene glycol. • Gases do not dissolve in ethylene glycol and the separation is assumed to • be ideal, i.e. ethylene glycol, is pure. • There is no water in the recycle, purge and product streams. 1% of gas stream is purged. Assume the combined stream consisting of the two fresh feeds and the recycle enters the reactor at 350°C, the reactor effluent is at a temperature of 380°c and that there are no heat losses. Using the data below, calculate how much heat has to be removed or supplied to the reactor to maintain the 380°C temperature. (b) [9 marks) Heat of formation for Cp in kJ mol K1 kJ mol 52.28 -52.6 Heat capacity coefficients Temperature units a 4.245 x 10 4.037 x 102 2.9253 x 10-2 3.682 x 102 3.333 x 10 8.852 x 10 1.392 x 104 9.242 x 105 3.141 x 105 9.353 x 10 ethylene ethylene oxide Tin °C for Cp = a + bT 0.0 охудеn carbon dioxide -393.5 -241.83 water (c) How would you control the reactor temperature? [1 mark] A process system for the manufacture of ethylene glycol is shown in Figure Q1. Ethylene oxide (CaH.O) is produced in the reactor by the following main reaction of catalytic oxidation of ethylene (C2H«): (6)O'HO- (6)o (6)"H) An undesired competing side reaction is the combustion of ethylene to CO2. CaHa(g) + 302(g)- 2CO2 (g) + 2H:O(g) The reactor product is fed to a separation system where water is added to convert ethylene oxide to ethylene glycol. CH,O(g) + H20(1)CaHO2(1) recycle: n, purge: n. water: n. ethylene: net oxygen: 10 kmol/h T 350°C T= 380 °C Separation System Preheater Reactor Q=? ethylene glycol product: neg n, - molar flow rate of x, kmol/h Figure Q1 (a) Produce a process material balance for the production of ethylene glycol based on an oxygen feed rate of 10 kmol h-1. Calculate the molar flowrates of each component in each stream. The following conditions apply: • All oxygen reacts, 90% goes to ethylene oxide, the rest to CO2 and H20. • Ethylene and oxygen feeds are pure. • 20% excess ethylene is fed to the process to ensure process safety. • No reaction of ethylene oxide with water takes place in the reactor. • All the water and ethylene oxide entering the separator is converted to • ethylene glycol. • Gases do not dissolve in ethylene glycol and the separation is assumed to • be ideal, i.e. ethylene glycol, is pure. • There is no water in the recycle, purge and product streams. 1% of gas stream is purged. Assume the combined stream consisting of the two fresh feeds and the recycle enters the reactor at 350°C, the reactor effluent is at a temperature of 380°c and that there are no heat losses. Using the data below, calculate how much heat has to be removed or supplied to the reactor to maintain the 380°C temperature. (b) [9 marks) Heat of formation for Cp in kJ mol K1 kJ mol 52.28 -52.6 Heat capacity coefficients Temperature units a 4.245 x 10 4.037 x 102 2.9253 x 10-2 3.682 x 102 3.333 x 10 8.852 x 10 1.392 x 104 9.242 x 105 3.141 x 105 9.353 x 10 ethylene ethylene oxide Tin °C for Cp = a + bT 0.0 охудеn carbon dioxide -393.5 -241.83 water (c) How would you control the reactor temperature? [1 mark]
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these computer network questions
-
A system with unity feedback is shown in Figure E5.11. Determine the steady-state error for a step and a ramp input when Figure E5.11 Unity feedback system. 5(s + 8) s(s 1)(s 4)(s 10) G(s) Gs) Yts)
-
A system with a controller is shown in Figure AP5.5. The zero of the controller may be varied. Let α = 0, 10, 100. (a) Determine the steady-state error for a step input r(t) for...
-
A feedback system with positive feedback is shown in Figure AP7.7. The root locus for K > 0 must meet the condition Sketch the root locus for 0 KG(s)-1 / k 360 G(s) Y(s) FIGURE AP7.7 A closed-loop...
-
True or False? Azure files can be accessed from anywhere in the world using a URL that points to the file. True False
-
Imagine that you and several of your classmates own a company that manufactures cheap sunglasses. To lower production costs, you choose to move your factory from your developed country to a more...
-
To stabilize a tree partially uprooted in a storm, cables AB and AC are attached to the upper trunk of the tree and then are fastened to steel rods anchored in the ground. Knowing that the tension in...
-
Plaintiff grounds manager sued a manufacturer, Monsanto, alleging that herbicide use caused his non-Hodgkins lymphoma. The jury awarded the plaintiff \($39.3\) million in compensatory damages and...
-
The marketing department of Metroline Manufacturing estimates that its sales in 2013 will be $1.5 million. Interest expense is expected to remain unchanged at $35,000, and the firm plans to pay...
-
The table below shows the output per labourer per day for Guyana and Jamaica for Coffee and Rice. Coffee Rice Table 1 Output per labourer per day Guyana Jamaica 8 6 10 4 What is the autarky cost of...
-
Excalibur Corporation sells video games for personal computers. The unadjusted trial balance as of December 31, 2024, appears below. December 31 is the company's reporting year-end. The company uses...
-
You lend Jack $18,000 and he agrees to repay you in equal year-end amounts over 5 years. If the interest rate is 9.2% per annum compounded quarterly, the annual repayment will be (rounded to nearest...
-
In May 2012, Honda Motor Scooter India Pvt Ltd (HMSI), the Indian subsidiary of Honda Motor Company Ltd. (HMCL), launched the Dream Yuga motorcycle in the Indian two wheeler market to attract mass...
-
8. A shipper obtains a marine policy covering the shipment of textiles from China to Poland. The declared value of the shipment was $15,000 although the real (market) value of the merchandise was...
-
Would it make sense to buy a house when mortgage rates are 14% and expected inflation is 15%? Explain your answer.
-
Imagine that you are a consultant on organizational development hired to help improve the work-life balance of a company's workforce. You have been asked to evaluate a published study about how...
-
The profile of facilities management is in the process of being enhanced by the Government Soft Landings initiative. In particular, greater emphasis is being placed on end user and building in use...
-
What does quality management in operations management aim to achieve? Cost reductionCustomer satisfactionEmployee motivationMarket expansion
-
Express these numbers in standard notation. a. 2.87 10-8 b. 1.78 1011 c. 1.381 10-23
-
Consider the closed-loop system in Figure P5.21. Determine values of the parameters k and a so that the following specifications are satisfied: (a) The steady-state error to a unit step input is...
-
Flexible-joint robotic arms are constructed of lightweight materials and exhibit lightly damped open-loop dynamics [15]. A feedback control system for a flexible arm. Select K so that the system has...
-
A unity feedback system has a loop transfer function where K = 1 / 2and T = 1. The polar plot for Gc(jÏ)G(jaÏ) is shown in Figure E9.24. Determine whether the system is stable by using the...
-
Cumulative Normal distribution \(\Phi_{(\mu, \sigma)}\) and probability (a) \(X \sim \phi_{(0,1)}\); what is \(P(X \leq 1.43)\) ? (b) \(X \sim \phi_{(0,1)}\); what is \(P(X>1.43)\) ? (c) \(X \sim...
-
Inverse cumulative Normal distribution \(z\) (a) Find \(z_{0.05}\). (b) Find \(z_{0.95}\). (c) Let \(X \sim \phi_{(2,1)}\). Find \(a\) such that \(P(X \leq a)=0.05\). (d) Let \(X \sim \phi_{(2,1)}\)....
-
The Normal approximation (a) A discrete stochastic variable \(X\) has expected value \(\mu_{X}=3\) and \(\sigma_{X}=1.2\). Use the Normal approximation to find \(P(X \leq 4)\). (b) A continuous...

Study smarter with the SolutionInn App


