A pyridine-water solution, 50 wt% pyridine, is to be continuously and countercurrently extracted at the rate...
Fantastic news! We've Found the answer you've been seeking!
Question:
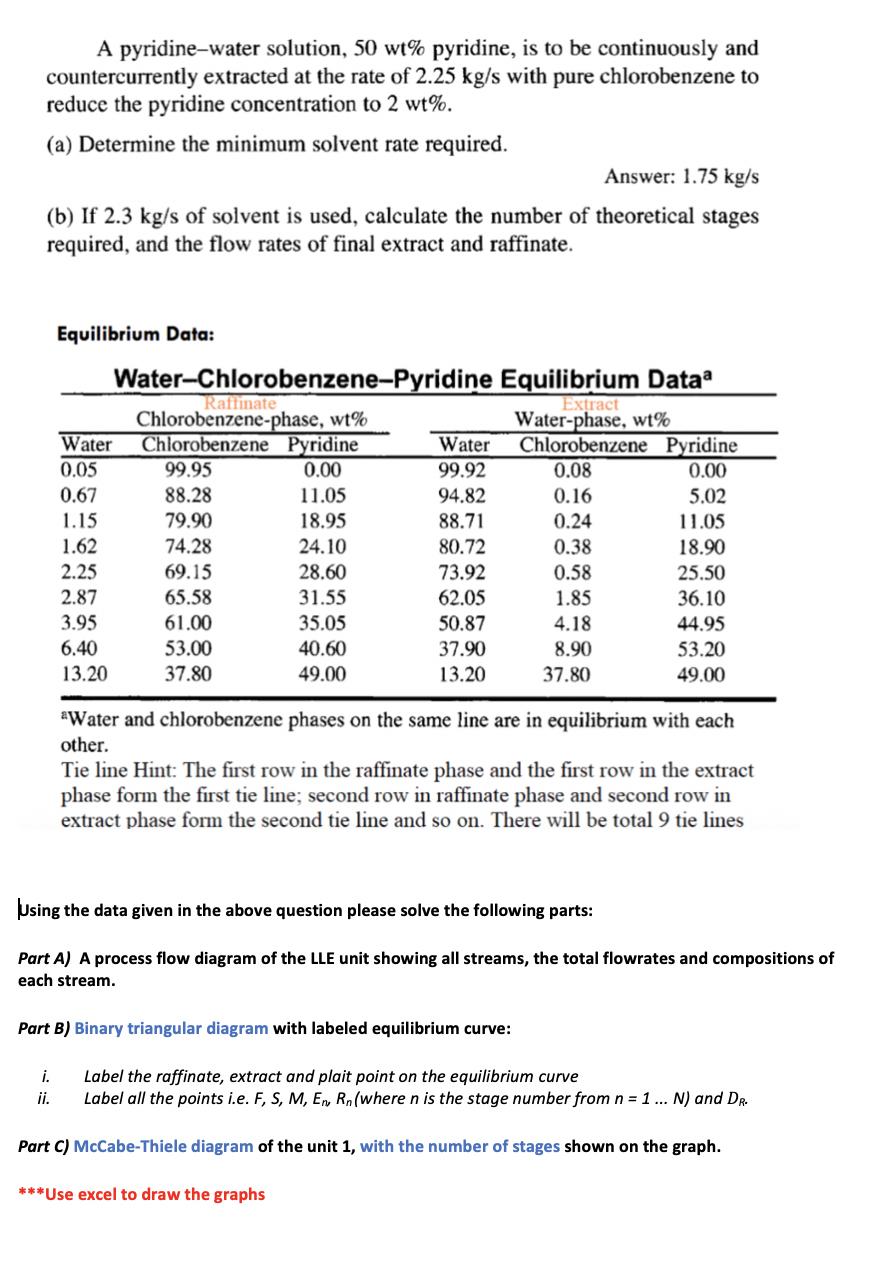
Transcribed Image Text:
A pyridine-water solution, 50 wt% pyridine, is to be continuously and countercurrently extracted at the rate of 2.25 kg/s with pure chlorobenzene to reduce the pyridine concentration to 2 wt%. (a) Determine the minimum solvent rate required. Answer: 1.75 kg/s (b) If 2.3 kg/s of solvent is used, calculate the number of theoretical stages required, and the flow rates of final extract and raffinate. Equilibrium Data: Water-Chlorobenzene-Pyridine Equilibrium Dataa Raffinate Chlorobenzene-phase, wt% Extract Water-phase, wt% Water Chlorobenzene Pyridine Water Chlorobenzene Pyridine 0.05 99.95 0.00 99.92 0.08 0.00 0.67 88.28 11.05 94.82 0.16 5.02 1.15 79.90 18.95 88.71 0.24 11.05 1.62 74.28 24.10 80.72 0.38 18.90 2.25 69.15 28.60 73.92 0.58 25.50 2.87 65.58 31.55 62.05 1.85 36.10 3.95 61.00 35.05 50.87 4.18 44.95 6.40 53.00 40.60 37.90 8.90 53.20 13.20 37.80 49.00 13.20 37.80 49.00 *Water and chlorobenzene phases on the same line are in equilibrium with each other. Tie line Hint: The first row in the raffinate phase and the first row in the extract phase form the first tie line; second row in raffinate phase and second row in extract phase form the second tie line and so on. There will be total 9 tie lines Using the data given in the above question please solve the following parts: Part A) A process flow diagram of the LLE unit showing all streams, the total flowrates and compositions of each stream. Part B) Binary triangular diagram with labeled equilibrium curve: i. Label the raffinate, extract and plait point on the equilibrium curve ii. Label all the points i.e. F, S, M, En Rn (where n is the stage number from n = 1 ... N) and DR. Part C) McCabe-Thiele diagram of the unit 1, with the number of stages shown on the graph. ***Use excel to draw the graphs A pyridine-water solution, 50 wt% pyridine, is to be continuously and countercurrently extracted at the rate of 2.25 kg/s with pure chlorobenzene to reduce the pyridine concentration to 2 wt%. (a) Determine the minimum solvent rate required. Answer: 1.75 kg/s (b) If 2.3 kg/s of solvent is used, calculate the number of theoretical stages required, and the flow rates of final extract and raffinate. Equilibrium Data: Water-Chlorobenzene-Pyridine Equilibrium Dataa Raffinate Chlorobenzene-phase, wt% Extract Water-phase, wt% Water Chlorobenzene Pyridine Water Chlorobenzene Pyridine 0.05 99.95 0.00 99.92 0.08 0.00 0.67 88.28 11.05 94.82 0.16 5.02 1.15 79.90 18.95 88.71 0.24 11.05 1.62 74.28 24.10 80.72 0.38 18.90 2.25 69.15 28.60 73.92 0.58 25.50 2.87 65.58 31.55 62.05 1.85 36.10 3.95 61.00 35.05 50.87 4.18 44.95 6.40 53.00 40.60 37.90 8.90 53.20 13.20 37.80 49.00 13.20 37.80 49.00 *Water and chlorobenzene phases on the same line are in equilibrium with each other. Tie line Hint: The first row in the raffinate phase and the first row in the extract phase form the first tie line; second row in raffinate phase and second row in extract phase form the second tie line and so on. There will be total 9 tie lines Using the data given in the above question please solve the following parts: Part A) A process flow diagram of the LLE unit showing all streams, the total flowrates and compositions of each stream. Part B) Binary triangular diagram with labeled equilibrium curve: i. Label the raffinate, extract and plait point on the equilibrium curve ii. Label all the points i.e. F, S, M, En Rn (where n is the stage number from n = 1 ... N) and DR. Part C) McCabe-Thiele diagram of the unit 1, with the number of stages shown on the graph. ***Use excel to draw the graphs
Expert Answer:
Answer rating: 100% (QA)
valuey So by putting 1105 y 1105 ty 2 00L vales in the ... View the full answer

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these general management questions
-
A stream of nitrogen flowing at the rate of 2 kg s-1 and a stream of hydrogen flowing at the rate of 0.5 kg s-l mix adiabatically in a steady-flow process. If the gases are assumed ideal, what is the...
-
Water at 68F flows at the rate of 628 cfs (ft3/sec) through 100 feet of horizontal corrugated metal pipe 10 ft in diameter. Determine the friction factor and flow regime (i.e., laminar, critical...
-
Question 5 An FSK system transmits binary data at the rate of 3 Mbps. During transmission, white Gaussian noise of with power spectral density (No/2) of 10-20 watts per hertz is added to the signal....
-
Your friend recently attended a local mail fraud trial. In your conversation about the case, she described the cross-examination of the expert witness as follows: After his counsels questioning was...
-
A manager wants to assign tasks to workstations in order to achieve an hourly output rate of four units. The department uses a working time of 56 minutes per hour. a. Assign the tasks shown in the...
-
What is the present value, when interest rates are 7.5 percent, of a $50 payment made every year forever?
-
Identify the most appropriate approach and then design. Provide a rationale as to why this approach and design would be most appropriate. Develop a hypothetical research scenario that would...
-
Nadal Inc. has two temporary differences at the end of 2008. The first difference stems from installment sales, and the second one results from the accrual of a loss contingency. Nadals accounting...
-
Solve the linear programming problem by the method of corners. (There may be more than one correct answer.) C=5x+by Minimize subject to 4x + y 42 2x+ y 30 x+3y 2 30 x20, y 20 The minimum is C 52.5...
-
In 1879, A.A. Michelson measured the velocity of light in air using a modification of a method proposed by the French physicist Foucault. Twenty of these measurements are in table 6E.27 (the value...
-
The management team is interested in knowing what potential vulnerabilities exist for my environment. Take this opportunity to review common vulnerabilities specific to my environment and provide the...
-
Calculate y' and y" for the equation y - x = 2. Express y' in terms of x and y. (Use symbolic notation and fractions where needed.) y' = y" Express y" in terms of x and y. (Use symbolic notation and...
-
4) Find the antiderivative of: [(8x-9x + 4) dx
-
Find f'(x) if f(x) = f'(x) = 4x + 6 x +7
-
Find the gradient of the function at the given point. w = x tan(y + z), (5,4,-2) Vw(5, 4, -2) = =
-
f For the functions f(x)=3-x and g(x)=x + 2x63, find f+g, f-g, fg, and g function. (f+g)(x) = (Simplify your answer.) Determine the domain for each
-
One month Tom rented 12 movies and 2 video games for a total of $29. The next month he rented 3 movies and 5 video games for a total of $32. Find the rental cost for each movie and each video game. 2...
-
When the concentration of a strong acid is not substantially higher than 1.0 10-7 M, the ionization of water must be taken into account in the calculation of the solution's pH. (a) Derive an...
-
Consider an air-standard cycle for the turbojet power plant shown in Fig. 8.13. The temperature and pressure of the air entering the compressor are 1 bar and 30oC. The pressure ratio in the...
-
Use the Souve/Redlieh/Kwong equation to calculate the molar volumes of saturated liquid and saturated vapor for the substance and conditions given by one of the ports of Pb. 3.38 and compare results...
-
A steel casting weighing 2 kg has an initial temperature of 500oC; 40 kg of water initially at 25oC is contained in a perfectly insulated steel tank weighing 5 kg. The casting is immersed in the...
-
Obtain a copy of the ACAS Code of Practice 1: Disciplinary and Grievance Procedures (2009). Use this, and our suggestions in this chapter, to rewrite or modify either of the procedures you used for...
-
One of the disadvantages of the IEEET1 exciter is following a fault the terminal voltage does not necessarily return to its prefault value. Using PowerWorld Simulator case Problem 12_3 determine the...
-
Rework Example 13.6 if the source impedance at the sending end of line \(\mathrm{A}\) is \(\mathrm{Z}_{\mathrm{G}}=\mathrm{Z}_{\mathrm{A}} / 4=100 \Omega\), and the receiving end of line...

Study smarter with the SolutionInn App


