A refrigerator (see Figure below) is used to keep a cold storage space (cold source) at...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
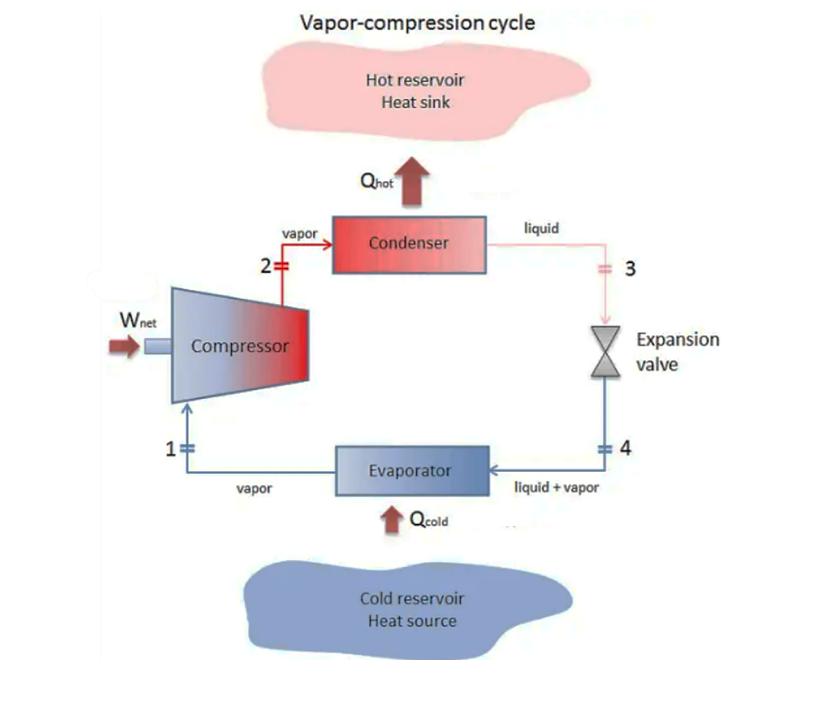
A refrigerator (see Figure below) is used to keep a cold storage space (cold source) at -20 °C (assume the evaporator saturation temperature is lower by 5 °C). To do so, it needs to remove 3.5 kW from the storage space. The temperature outside the system (hot source) is 25 °C (assume the condenser saturation temperature is higher by 10 °C). The cycle uses refrigerant R22. With this refrigerant and under the described operating conditions, the actual coefficient of performance (COP) of the cycle is 2. (Hint: Assume the outlet flow from the condenser is saturated liquid and the outlet flow from the evaporator is saturated vapor. Deal with condenser and evaporator as stream flow with heat transfer.) a) What is the operating pressure at the low-pressure side of the cycle (evaporator)? b) What is the operating pressure at the high-pressure side of the cycle (condenser pressure)? c) What is the quality of the process stream at the INLET to the evaporator in the cycle? d) What is the mass flow rate of R22 needed to achieve the desired amount of cooling? e) Calculate the work required by the compressor in units of kW. f) What is the isentropic efficiency of the compressor? g) What is the rate of entropy generation for each component, with proper comments and recommendations? Wnet 1: 2= vapor Compressor vapor Vapor-compression cycle Hot reservoir Heat sink Qhot Condenser Evaporator Qcold Cold reservoir Heat source liquid liquid + vapor 3 4 Expansion valve A refrigerator (see Figure below) is used to keep a cold storage space (cold source) at -20 °C (assume the evaporator saturation temperature is lower by 5 °C). To do so, it needs to remove 3.5 kW from the storage space. The temperature outside the system (hot source) is 25 °C (assume the condenser saturation temperature is higher by 10 °C). The cycle uses refrigerant R22. With this refrigerant and under the described operating conditions, the actual coefficient of performance (COP) of the cycle is 2. (Hint: Assume the outlet flow from the condenser is saturated liquid and the outlet flow from the evaporator is saturated vapor. Deal with condenser and evaporator as stream flow with heat transfer.) a) What is the operating pressure at the low-pressure side of the cycle (evaporator)? b) What is the operating pressure at the high-pressure side of the cycle (condenser pressure)? c) What is the quality of the process stream at the INLET to the evaporator in the cycle? d) What is the mass flow rate of R22 needed to achieve the desired amount of cooling? e) Calculate the work required by the compressor in units of kW. f) What is the isentropic efficiency of the compressor? g) What is the rate of entropy generation for each component, with proper comments and recommendations? Wnet 1: 2= vapor Compressor vapor Vapor-compression cycle Hot reservoir Heat sink Qhot Condenser Evaporator Qcold Cold reservoir Heat source liquid liquid + vapor 3 4 Expansion valve
Expert Answer:
Answer rating: 100% (QA)
Given Cold storage temperature 20C Evaporator saturation temperature 20C 5C 25C Hot source temperatu... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
The shareholders equity of HS Corporation includes $ 3 8 0 , 0 0 0 of $ 1 par common stock and $ 6 8 0 , 0 0 0 par of 7 % cumulative preferred stock. The board of directors of HS declared cash...
-
A heat pump with refrigerant-134a as the working fluid is used to keep a space at 25°C by absorbing heat from geothermal water that enters the evaporator at 50°C at a rate of 0.065 kg/s and...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Bob drives off in Freds car mistakenly believing that it is his. Is this theft? Explain.
-
Washington County's Board of Representatives is considering the construction of a longer runway at the county airport. Currently, the airport can handle only private aircraft and small commuter jets....
-
Swinn Company manufactures bicycles. Materials are added at the beginning of the production process, and conversion costs are incurred uniformly. Production and cost data for the month of May are as...
-
\(\frac{d^{8}}{d^{17}}\) Simplify the expression.
-
FruTee Corp. makes irrigation sprinkler systems for fruit tree nurseries. FruTees new controller can find only the following partial information for the past year: Compute the unknown values. List...
-
Instructions: Answer the question below Ken and Sam, who prepare their accounts annually to 30 September, are partners in retail business sharing profits and losses in the ratio of 3:2 respectively....
-
From the adjustments columns in Exercise 5-9A, journalize the four adjusting entries, as of December 31, in proper general journal format. Exercise 5-9A Jim Jacobs Furniture Repair Work Sheet...
-
St. John Corporation, a Canadian controlled private corporation has Taxable Income of $250,000 in 2019, 87 percent of which is earned in a Canadian province. The company reported Canadian active...
-
The following is an interesting interview with Ray Kurzweil that explains what the "Singularity" is. It is only two months ago, before the explosion Chat GPT....
-
1. A flywheel rotates at 5.0 revolutions per second when it is brought uniformly to rest in 20 seconds. What is the magnitude of the angular acceleration of the flywheel during this time?
-
Use the two force relationships you just derived with Newton's 2nd law to write an algebraic expression for the coefficient of kinetic friction (u) that depends only on 0.
-
A company has the choice of either selling 1 , 0 0 0 unfinished units as is or completing them. The company could sell the unfinished units as is for $ 4 , 0 0 0 . Alternatively, it could complete...
-
An object moves along a path whose parametric equation, are y= 2t^2 and x= t^3 where x & y are distances travelled in meters & t is time in seconds a.) velocity @ t = 2sec b.) acceleration after 3...
-
kevin and his friends are headed to nyc, to ski at a pricey ski area. The ski club unloads about 1,500 skiers per hour at the top of a diamond slope. The ride from the bottom to the top of the slope...
-
Determine the values of the given trigonometric functions directly on a calculator. The angles are approximate. tan 0.8035
-
Show that: av , ,
-
At winter design conditions, a house is projected to lose heat at a rate of 60,000 Btu/h. The internal heat gain from people, lights, and appliances is estimated to be 6000 Btu/h. If this house is to...
-
Determine the adiabatic saturation temperature of the humid air in Prob. 14-41. Prob. 14-41. Atmospheric air at a pressure of 1 atm and drybulb temperature of 28oC has a wet-bulb temperature of 20oC....
-
List some ways you might obtain job-related experience.
-
Identify sources of information to find out more about the career in which you are interested.
-
Explain your rights as an employee.

Study smarter with the SolutionInn App


