1. A sample of a concentrated sulfuric acid solution with a density of 1.84 g/cm contains...
Fantastic news! We've Found the answer you've been seeking!
Question:
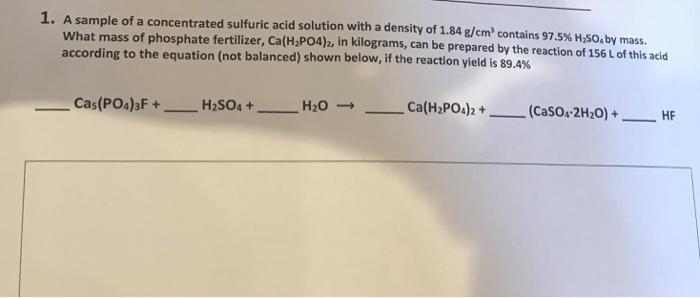
Transcribed Image Text:
1. A sample of a concentrated sulfuric acid solution with a density of 1.84 g/cm³ contains 97.5% H₂SO, by mass. What mass of phosphate fertilizer, Ca(H₂PO4)2, in kilograms, can be prepared by the reaction of 156 L of this acid according to the equation (not balanced) shown below, if the reaction yield is 89.4% Cas(PO4)3F+ H₂SO4 + — H₂O -> Ca(H₂PO4)2 + (CaSO4-2H₂O) + HF 1. A sample of a concentrated sulfuric acid solution with a density of 1.84 g/cm³ contains 97.5% H₂SO, by mass. What mass of phosphate fertilizer, Ca(H₂PO4)2, in kilograms, can be prepared by the reaction of 156 L of this acid according to the equation (not balanced) shown below, if the reaction yield is 89.4% Cas(PO4)3F+ H₂SO4 + — H₂O -> Ca(H₂PO4)2 + (CaSO4-2H₂O) + HF
Expert Answer:
Answer rating: 100% (QA)
2 CasPoub F HSO4 14 HO 36 12804 765042420 First balance Ply then Ca then s... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
A ketone can be prepared from the reaction of a nitrile with a Grignard reagent. Describe the intermediate that is formed in this reaction, and explain how it can be converted to a ketone.
-
Phosphoric acid, H3PO4, can be prepared by the reaction of phosphorus(V) oxide, P4O10, with water. What is H for the reaction involving 1 mol of P4O10? P4O10(s) + 6H2O(l ) 4H3PO4(aq) P,O10(s) +...
-
Gaseous iodine pentafluoride, IF5, can be prepared by the reaction of solid iodine and gaseous fluorine: I2 (s) + 5F2 (g) 2IF5 (g) A 5.00-L flask containing 10.0 g I2 is charged with 10.0 g F2, and...
-
Alert Security Services Co. offers security services to business clients. The trial balance for Alert Security Services Co. has been prepared on the following end-of-period spreadsheet for the year...
-
You have just been told, Because we are going to finance this project with debt, its required rate of return must exceed the cost of debt. Do you agree or disagree? Explain.
-
In a study conducted in New Zealand, Parkin et al. randomly assigned volunteers to either wear socks over their shoes (intervention) or wear usual footwear (control) as they walked downhill on an...
-
The following MINITAB output presents a 95% confidence interval for the mean cholesterol levels for men aged 50 years, and a 95% prediction interval for an individual man aged 50. The units of...
-
In March 2011, an explosion occurred at Kirk Co.'s plant, causing damage to area properties. By May 2011, no claims had yet been asserted against Kirk. Kirk's management and legal counsel, however,...
-
! Required information [The following information applies to the questions displayed below.] Cane Company manufactures two products called Alpha and Beta that sell for $185 and $120, respectively....
-
XYZ is a calendar-year corporation that began business on January 1, 2020. For the year, it reported the following information in its current-year audited income statement. Notes with important tax...
-
Research and Customers had renewed appreciation for hospitality staff. Frontline workers faced health risks and dealt with constantly changing policies. Many people felt sympathy for these...
-
Following statements are given Consolidated Statements of Financial Position - USD ($) $ in Millions Assets Cash and cash equivalents Inventory Other current assets Total current assets Property...
-
A proton (q=e= 1.602x10-19 C, m-1.67x10-27 kg) goes through a small hole in a negatively charged metal plate with an initial velocity of v 7.42x105 m/s at 0=11 degrees above the horizontal. The field...
-
Which one of the metal atoms whose electronic structure given below forms a nitrate of the type M(NO3)2? 37 A. 2.8.0 B. 2.8.1 C. 2.8.2 D. 2.8.3
-
17. The reaction A+B C+D is carried out at 80 C in an isothermal, perfectly stirred tank batch reactor. The reaction rate can be described as second-order, where k-0.058 m kmo1 min. At 80 C, 240 kg...
-
2. Current is flowing through a wire at a constant amperage of 15 coulombs/second. The wire is composed of copper that is 10 cm long with a 2 mm radius. The resulting resistivity of the copper is...
-
XYZ Insurance Corporation. XYZ Insurance Corporation is a leading global insurance organization serving business and individuals with a broad range of insurance products and insurance-related...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
If an average O3 molecule "lives" only 100-200 seconds in the stratosphere before undergoing dissociation, how can O3 offer any protection from ultraviolet radiation?
-
(a) Use data in Appendix C to estimate the boiling point of benzene, C6H6(l). (b) Use a reference source, such as the CRC Handbook of Chemistry and Physics, to find the experimental boiling point of...
-
Each of the following elements is capable of forming an ion in chemical reactions. By referring to the periodic table, predict the charge of the most stable ion of each: (a) Mg, (b) Al, (c) K, (d) S,...
-
Which of the following activities would most likely be performed in the computer department? a. Conversion of information to machine-readable form. b. Initiation of changes to master records. c....
-
For control purposes, which of the following should be organizationally segregated from the computer operations functions? a. Surveillance of CRT messages. b. Data conversion. c. Systems development....
-
What two attributes of controls do tests of controls measure?

Study smarter with the SolutionInn App


