A solid metal M can form two oxides, MO and M304. The metal can exist in equilibrium
Fantastic news! We've Found the answer you've been seeking!
Question:
A solid metal M can form two oxides, MO and M304. The metal can exist in equilibrium with one of these oxides at different temperatures. Determine which of the two oxides is in equilibrium with the metal at room temperature and the maximum temperature at which this oxide is in equilibrium with M.
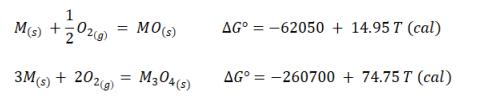
Related Book For 

University Physics with Modern Physics
ISBN: 978-0321501219
12th Edition
Authors: Hugh D. Young, Roger A. Freedman, Lewis Ford
Posted Date:




