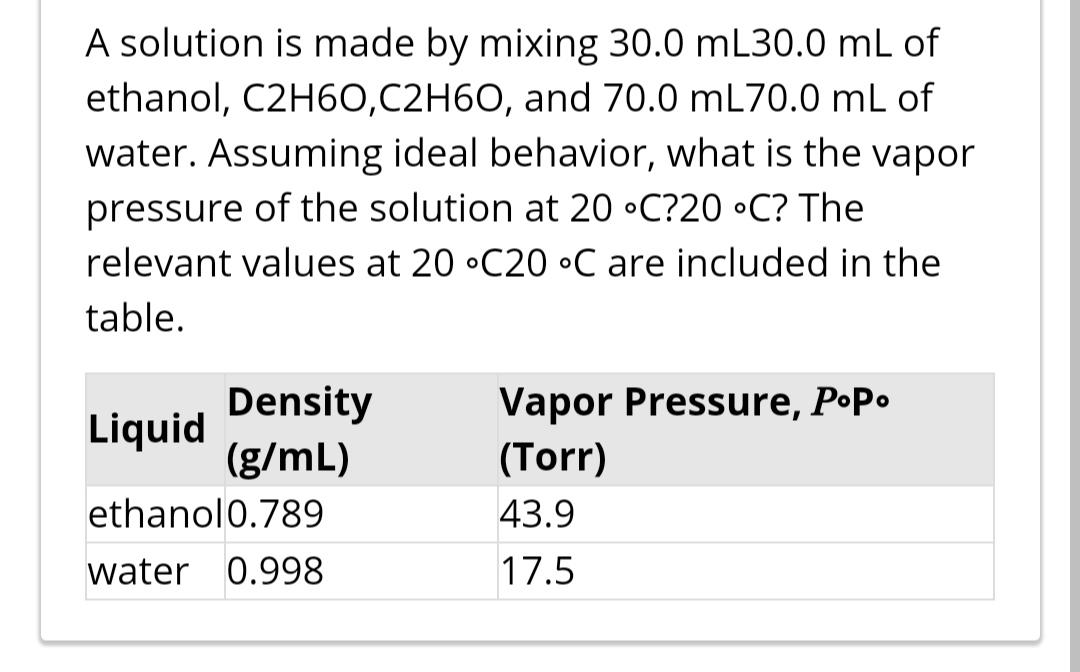
A solution is made by mixing 30.0 mL30.0 mL of ethanol, C2H60,C2H6O, and 70.0 mL70.0 mL...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A solution is made by mixing 30.0 mL30.0 mL of ethanol, C2H60,C2H6O, and 70.0 mL70.0 mL of water. Assuming ideal behavior, what is the vapor pressure of the solution at 20 •C?20 •C? The relevant values at 20 •C20 •C are included in the table. Density (g/mL) Vapor Pressure, P•P. Liquid (Torr) ethanol0.789 43.9 water 0.998 17.5 A solution is made by mixing 30.0 mL30.0 mL of ethanol, C2H60,C2H6O, and 70.0 mL70.0 mL of water. Assuming ideal behavior, what is the vapor pressure of the solution at 20 •C?20 •C? The relevant values at 20 •C20 •C are included in the table. Density (g/mL) Vapor Pressure, P•P. Liquid (Torr) ethanol0.789 43.9 water 0.998 17.5
Expert Answer:
Answer rating: 100% (QA)
Volume of ethanol 30 mL and density 0789 gmL so mass ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
A solution is made by mixing 50 mL of 2.0 M K2HPO4 and 25 mL of 2.0 M KH2 PO4. The solution is diluted to a final volume of 200 mL. What is the pH of the final solution?
-
A solution is made by mixing 5.00 102 mL of 0.167 M NaOH with 5.00 102 mL of 0.100 M CH3COOH. Calculate the equilibrium concentrations of H1, CH3COOH, CH3COO-, OH2, and Na+.
-
A solution is made by mixing 33.8 g of CH3OH in 50.0 g of H2O. What is the mole fraction of CH3OH in the solution?
-
Distance between Cars (Refer to Example 6.) At 9:00 A.M. car A is traveling north at 50 miles per hour and is located 50 miles south of car B. Car B is travel- ing west at 20 miles per hour. (a) Let...
-
What is the molarity of Na+ in a solution of NaCl whose salinity is 5.6 if the solution has a density of 1.03 g/mol?
-
In 2012, 15.9% of all U.S. families had incomes below the poverty level, as reported by the U.S. Census Bureau in American Community Survey. During that same year, of 400 randomly selected Wyoming...
-
John is considering the purchase of a lot. He can buy the lot today and expects the price to rise to $\$ 15,000$ at the end of 10 years. He believes that he should earn an investment yield of $10 \%$...
-
The Trotter Trust has the receipts and expenditures listed below for the current year. Assume the Uniform Act governs an items classification as principal or income. The trustees fee is charged...
-
Each question below is 1 point each, for a total of 3 extra credit points. There is no time limit on this. 1. What are free cash flows? 2. Explain the difference between a company's operating cash...
-
1. In cell E2, enter a formula using the HLOOKUP function to determine a staff member?s base salary (which is based on the number of years of service). a. Use a structured reference to look up the...
-
All Corporation's unadjusted trial balance includes the following balances (assume normal balances): Accounts Receivable Allowance for Doubtful Accounts a. $15,000 b. $36,000 Bad debts are estimated...
-
Find the derivative of the function. f (u) = u2+10 f '(u) =
-
Refer to the given figure, an isosceles triangle with AB = BC If angle B measures 60, then find the measures of angle A and angle C. A.. Su The measure of angle A is The measure of angle C is
-
An Atwood's Machine is a counterbalance machine used for lifting heavy objects.A picture of one is below.If we hang a 250 kg mass on both sides of the machine, what is the tension in the string? m m2
-
An ocean liner is cruising 15 knots in the direction S70W. A sailing vessel is moving at 6 knots in the direction S40E. Find the velocity and direction of the liner relative to the sailboat. use high...
-
Determine the simple interest. (Assume 360 days in a year.) p=$607, r=0.053% per day, t=6 months The simple interest on $607 at 0.053% per day for 6 months is $ (Round to the nearest cent.)
-
2016 ACC Expert Consensus Decision Pathway on the Role of Non-Statin Therapies and the 2017 focused update of the 2016 ACC Expert Consensus Decision Pathway on the Role of Non-Statin Therapies for...
-
List four items of financial information you consider to be important to a manager of a business that has been operating for a year.
-
(a) In polyvinyl chloride shown in Table 12.5, which bonds have the lowest average bond enthalpy? (b) When subjected to high pressure and heated polyvinyl chloride converts to diamond. During this...
-
Based on data in Table 8.2, estimate (within 30 kJ / mol) the lattice energy for (a) LiBr, (b) CsBr, (c) CaCl2. TABLE 8.2 Lattice Energies for Some Ionic Compounds Lattice Energy (kJ/mol) Lattice...
-
Arrange the bonds in each of the following sets in order of increasing polarity: (a) C-F, O-F, Be-F (b) O-Cl, S-Br, C-P (c) C-S, B-F, N-O
-
Consider an experiment that selects a cell phone camera and records the recycle time of a flash (the time taken to ready the camera for another flash). The possible values for this time depend on the...
-
Suppose that the recycle times of two cameras are recorded. The extension of the positive real line \(R\) is to take the sample space to be the positive quadrant of the plane \[ S=R^{+} \times R^{+}...
-
As in Example 2.1, camera recycle times might use the sample space \(S=R^{+}\), the set of positive real numbers. Let \[ E_{1}=\{x \mid 10 \leq x <12\} \quad \text { and } \quad E_{2}=\{x \mid 11 0\}...

Study smarter with the SolutionInn App


