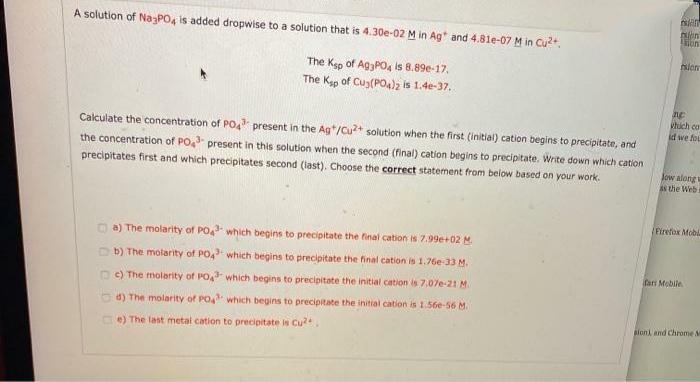
A solution of Na-PO, is added dropwise to a solution that is 4.30e-02 M in Ag...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A solution of Na-PO, is added dropwise to a solution that is 4.30e-02 M in Ag and 4.81e-07 M in Cu2+. nlon The Keo of AgPO, is 8.89e-17, The Kap of Cus(PO4)z is 1.4e-37. which co id we fo Calculate the concentration of PO, present in the Ag*/Cu+ solution when the first (initial) cation begins to precipitate, and the concentration of PO present in this solution when the second (final) cation begins to precipitate. Write down which cation precipitates first and which precipitates second (iast). Choose the correct statement from below based on your work. Jow alongs AS the Web (Firefax Mobl a) The molarity of PO which begins to precipitate the final cation is 7.99e+02 M. O b) The molarity of PO, which begins to precipitate the final cation is 1.76e-33 M. fari Mobile O e) The molarity of PO which begins to precipitate the initial cation is 7.07e-21 M. d) The molarity of PO which begins to precipitate the initial cation is 1.56e-56 M. loni and Chrome M Ce) The last metal cation to precipitate is Cu2. A solution of Na-PO, is added dropwise to a solution that is 4.30e-02 M in Ag and 4.81e-07 M in Cu2+. nlon The Keo of AgPO, is 8.89e-17, The Kap of Cus(PO4)z is 1.4e-37. which co id we fo Calculate the concentration of PO, present in the Ag*/Cu+ solution when the first (initial) cation begins to precipitate, and the concentration of PO present in this solution when the second (final) cation begins to precipitate. Write down which cation precipitates first and which precipitates second (iast). Choose the correct statement from below based on your work. Jow alongs AS the Web (Firefax Mobl a) The molarity of PO which begins to precipitate the final cation is 7.99e+02 M. O b) The molarity of PO, which begins to precipitate the final cation is 1.76e-33 M. fari Mobile O e) The molarity of PO which begins to precipitate the initial cation is 7.07e-21 M. d) The molarity of PO which begins to precipitate the initial cation is 1.56e-56 M. loni and Chrome M Ce) The last metal cation to precipitate is Cu2.
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Write a college-level reflective essay based on your work or life experience. The essay must contain the correct number of subtopics and real-world experiences with sufficient examples of your own...
-
Based on your work in the previous three problems, a. Determine a seasonally adjusted linear trend forecasting model. Compare this model with an unadjusted linear trend model. Use both models to...
-
Based on your work experience, write a report titled Work Experience Report. The report should include profile of the organization and your plan (with aims, objective and targets) for the project....
-
Junes Garden Supplies purchases merchandise on account from Lawn Supplies, Inc. The list price is $1,600, with a trade discount of 25 percent, terms 1/10, n/30. Junes Garden Supplies pays the amount...
-
Name this molecule. C-c=C-C-C -C
-
Air flows in a frictionless, adiabatic duct at M = 0.6 and P 0 = 500 kPa. The cross-sectional area of the duct is 6 cm 2 and the mass flow rate is 0.5 kg/s. If the area of the duct near the exit is...
-
Comparative statements of financial position as at 31 December 2025 and 2024 and the statement of financial performance for 2025 for C. Dion are set out below. Required (a) Prepare a statement of...
-
Faced with three young children who were always complaining Im bored, Ann Newton was looking for an interesting afterschool activity. Finding nothing available in the community, she decided to solve...
-
A dog training business began on December 1. The following transactions occurred during its first month. December 1 Receives $22,000 cash as an owner investment in exchange for common stock. December...
-
Kate Collins has opened Fontina and Stuff, a wholesale grocery and cheese company. The following transactions occurred in February: Required 1. Journalize the transactions. Beginning balances of...
-
Friends (Pty) Ltd is a retailer of industrial machinery. The machinery sold by Friends (Pty) Ltd is purchased from local manufacturers. In recent years, Friends has also started manufacturing...
-
How can pension plans and mutual funds help solve the global warming problem?
-
What are the investment grade ratings used by S&P.
-
What are the advantages of expected shortfall over value at risk?
-
Explain how machine learning is changing the way lending decisions are made. What are the risks in relying on machine learning algorithms for these decisions?
-
Outline the basic steps in promotion planning.
-
What is the output of the following C++ code? (4) vector intList (5); int num = 1; for (auto &p : intList) { if (num % 2 == num % 3) p = 2 * num; else } p = 3* num; num = p-2; for (auto p: intList)...
-
You have accepted the engagement of auditing the financial statements of the C. Reis Company, a small manufacturing firm that has been your auditee for several years. Because you were busy writing...
-
Acetonitrile (CH3CN) is a polar organic solvent that dissolves a wide range of solutes, including many salts. The density of a 1.80 M LiBr solution in acetonitrile is 0.826 g/cm3. Calculate the...
-
The compound chloral hydrate, known in detective stories as knockout drops, is composed of 25.52%C, 1.83% H, 64.30% Cl, and 19.35% O by mass and has a molar mass of 165.4g/mol. (a) What is the...
-
Calculate the pH of a solution made by adding 2.50 g of lithium oxide (Li2O) to enough water to make 1.500 L of solution.
-
Calculate the double-declining balance depreciation schedule for a \($1,000\) item that will last four years. What is the estimated salvage value?
-
If an interest rate is currently 6% and a lending institution announces a 25 basis point increase, what percentage increase does this represent?
-
Calculate the straight-line and sum-of-years-digits depreciation schedules for a \($450\) video camera that will have a salvage value of \($50\) after five years of use.

Study smarter with the SolutionInn App


