A motorist applies the two forces shown to loosen a lug nut. The direction cosines of...
Fantastic news! We've Found the answer you've been seeking!
Question:
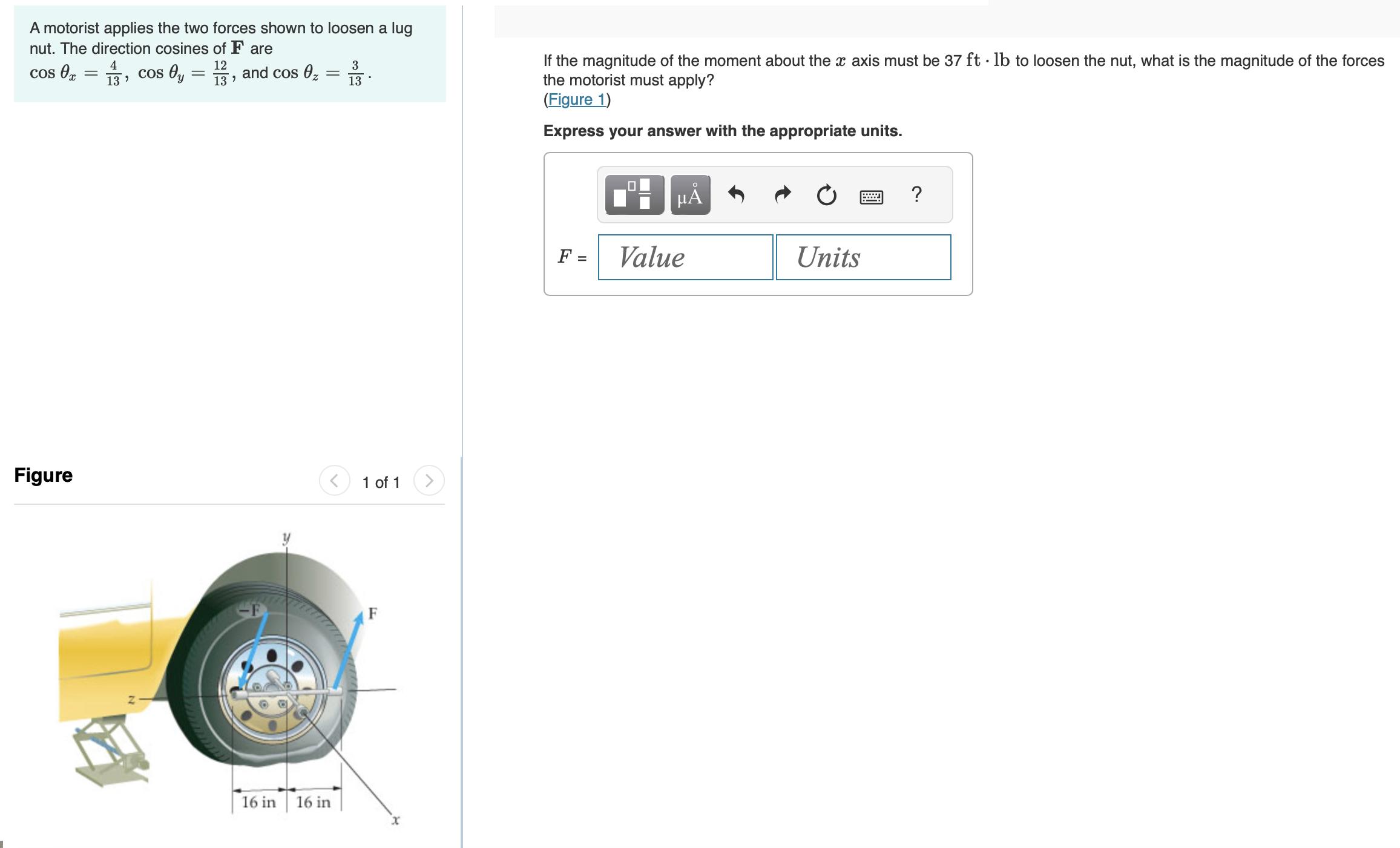
Transcribed Image Text:
A motorist applies the two forces shown to loosen a lug nut. The direction cosines of F are 12 13⁹ 4 cos 0x = 13, cos Oy - = Figure and cos 0₂ = 16 in 16 in 3 13 1 of 1 F If the magnitude of the moment about the x axis must be 37 ft· lb to loosen the nut, what is the magnitude of the forces the motorist must apply? (Figure 1) Express your answer with the appropriate units. F = 0 μĂ Value Units A motorist applies the two forces shown to loosen a lug nut. The direction cosines of F are 12 13⁹ 4 cos 0x = 13, cos Oy - = Figure and cos 0₂ = 16 in 16 in 3 13 1 of 1 F If the magnitude of the moment about the x axis must be 37 ft· lb to loosen the nut, what is the magnitude of the forces the motorist must apply? (Figure 1) Express your answer with the appropriate units. F = 0 μĂ Value Units A motorist applies the two forces shown to loosen a lug nut. The direction cosines of F are 12 13⁹ 4 cos 0x = 13, cos Oy - = Figure and cos 0₂ = 16 in 16 in 3 13 1 of 1 F If the magnitude of the moment about the x axis must be 37 ft· lb to loosen the nut, what is the magnitude of the forces the motorist must apply? (Figure 1) Express your answer with the appropriate units. F = 0 μĂ Value Units
Expert Answer:

Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Posted Date:
Students also viewed these accounting questions
-
Assuming = 1, determine the distribution factor specified in the AASHTO LRFD Bridge design specifications for the moment in the exterior and interior beams for the bridge shown below. The bridge has...
-
Old Office Building Karas Investments owns a small office building in Windhoek, from which the company operates. The Windhoek office was purchased on 1 March 2019. Karas Investments decided to move...
-
The case provides select financial information on GM for the years 2019 to 2021. For each item below, calculate the percentage of rate of annual growth (or decline) from 2019 to 2020 and 2020 to...
-
In a closed economy, prices are constant/fixed. Behaviour is characterised by the following relationships. C=0.9375 (Y-T), T=10 and I=50-250i Money demand Md=Y-100i The supply of money and government...
-
Reconsider the Wyndor Glass Co. case study introduced in Section 2.1. Suppose that Bill Tasto (Wyndor's vice president for manufacturing) now has found a way to provide a little additional production...
-
a. Describe fully what will occur if a plant cell is placed in a solution that has a higher water potential than the cell. Use the following terms in your answer. cell wall, freely permeable,...
-
99 percent per month. In 2014, Boling resolved his suit against the gas can manufacturer. Shortly thereafter, Prospect sent Boling a Schedule of Purchases, asserting that Boling owed Prospect...
-
The Edelman Gem Company, a small jewelry manufacturer, has been successful and has enjoyed a good growth trend. Now Edelman is planning to go public with an issue of common stock , and it faces the...
-
Provide three comparisons between hierarchical, network and relational database models. You can present your answer in the table. (b) The relational database model is the most widely used database....
-
Myles Etter and Crystal Santori are partners who share in the income equally and have capital balances of $210,000 and $62,500, respectively. Etter, with the consent of Santori, sells one-third of...
-
Instructions: Visit the following URL for additional information on the Federal Emergency Management Agency (FEMA), the National Incident Management System (NIMS), and the Incident Command System...
-
What is the theoretical yield, in g, of PbCl2 when 11.7 g of NaCl is reacted with 361 mL of 0.359 M Pb(NO3)2 according to the equation below? Pb(NO3)2(aq) + 2 NaCl(s) PbCl2(s) + 2 NaNO3(aq)
-
The cost of equity for rosental corporation is 13.61%. the expected return on the market =12.5%, the risk free rate is 1.4%, inflation rate =3.3%. what is the company equity beta?
-
Create a project draft that provides the cost for developing the project, securing permissions, costs for preparations, costs to implement (paper, time, incentives, thank-you cards, etc.), costs to...
-
Briefly, explain the Uniform Commercial Code. Is it enforceable in all 50 states of the United States?
-
Discuss some of the benefits of using global citizenship principles when communicating across cultures for a professional organization. The organization type could be anything from a nonprofit...
-
make the journal neccessary entities for the transaction: (so the solution to the question is sequential) 1.Company exported 28.000 USD Merchandise, Rate was 8,05 TL/USD. The account will be closed...
-
Proposals have been made to ?sail? spacecraft to the outer solar system using the pressure of sunlight, or even to propel interstellar spacecraft with high-powered, Earth-based lasers. Sailing...
-
Which acid and base react to give an aqueous solution of K 2 SO 4 ? Write a balanced equation for this neutralization reaction.
-
An aqueous solution that has an H 3 O + concentration of 10 8 M is (a) Acidic. (b) Basic. (c) Neutral. (d) There is so little H 3 O + that the question cannot be answered.
-
Creating a cation from a neutral atom (a) Always takes energy, called the ionization energy. (b) Always requires the removal of an electron from the atom. (c) Never requires the addition of an...
-
Distinguish between (a) a branch, (b) a subsidiary, and (c) a joint venture.
-
What purposes are normally served by bilateral investment protection treaties?
-
What is meant by collateral security?

Study smarter with the SolutionInn App


