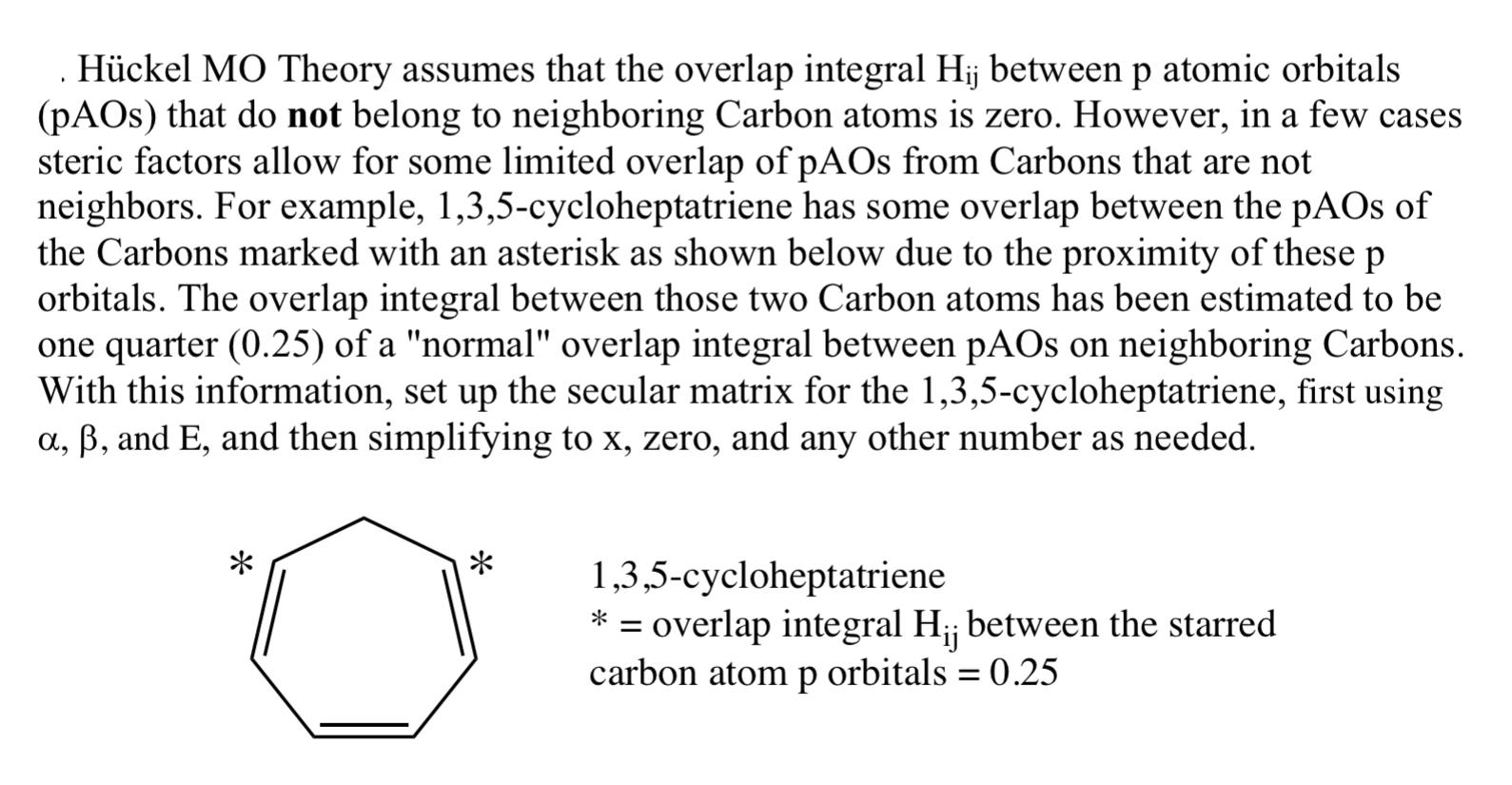
.Hckel MO Theory assumes that the overlap integral Hij between p atomic orbitals (pAOs) that do...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
.Hückel MO Theory assumes that the overlap integral Hij between p atomic orbitals (pAOs) that do not belong to neighboring Carbon atoms is zero. However, in a few cases steric factors allow for some limited overlap of pAOs from Carbons that are not neighbors. For example, 1,3,5-cycloheptatriene has some overlap between the pAOs of the Carbons marked with an asterisk as shown below due to the proximity of these p orbitals. The overlap integral between those two Carbon atoms has been estimated to be one quarter (0.25) of a "normal" overlap integral between pAOs on neighboring Carbons. With this information, set up the secular matrix for the 1,3,5-cycloheptatriene, first using a, ß, and E, and then simplifying to x, zero, and any other number as needed. * * 1,3,5-cycloheptatriene = overlap integral H₁; between the starred carbon atom p orbitals = 0.25 .Hückel MO Theory assumes that the overlap integral Hij between p atomic orbitals (pAOs) that do not belong to neighboring Carbon atoms is zero. However, in a few cases steric factors allow for some limited overlap of pAOs from Carbons that are not neighbors. For example, 1,3,5-cycloheptatriene has some overlap between the pAOs of the Carbons marked with an asterisk as shown below due to the proximity of these p orbitals. The overlap integral between those two Carbon atoms has been estimated to be one quarter (0.25) of a "normal" overlap integral between pAOs on neighboring Carbons. With this information, set up the secular matrix for the 1,3,5-cycloheptatriene, first using a, ß, and E, and then simplifying to x, zero, and any other number as needed. * * 1,3,5-cycloheptatriene = overlap integral H₁; between the starred carbon atom p orbitals = 0.25
Expert Answer:
Answer rating: 100% (QA)
Here is the secular matrix for 135cycloheptatrienefirst using and Eand then simplifying to xzeroand ... View the full answer

Related Book For 

Posted Date:
Students also viewed these general management questions
-
Ethan Properties Philippines, Inc. was registered with the Philippine SEC in 2008 and is engaged in developing and operating for sale, lease, or hold for investment real estate of all kinds,...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Single PlantwideandMultiple Production Department Factory Overhead Rate Methodsand Product Cost Distortion The management of Nova Industries Inc. manufactures gasoline and diesel engines through two...
-
An excited H atom is in a 6d state. (a) Name all the state (n, l) to which the atom is allowed to jump with the emission of a photon. (b) How many different wave-lengths are there (ignoring fine...
-
Sydney Park Fitness Gym has \($500,000\) of 10-year bonds payable outstanding. These bonds had a discount of \($45,000\) at issuance, which was five years ago. The company uses the straight-line...
-
Which theory emphasizes the importance of cooperation and participation in the workplace? A. Chaos Theory. B. Systems Theory. C. Classical Theory. D. Neoclassical Theory.
-
Gates Video Games manufactures video game machines. Market saturation and technological innovations have caused pricing pressures that have resulted in declining profits. To stem the slide in profits...
-
Suppose your firm is considering investing in a project with the cash flows shown below, that the required rate of return on projects of this risk class is 1 2 percent, and that the maximum allowable...
-
Calculate Federal Income Tax Withholding Using Two Methods (pre-2020 Form W-4) For each employee listed, use both the wage-bracket method and the percentage method to calculate federal income tax...
-
Determine the power rating of the motor in an elevator system. The elevator (with a full load) weights 2000 kg and is required to move upward 2 m/sec at constant speed. The lifting mechanism is 80%...
-
Calculate free cash flow to the firm (FCFF) based on the Statement of Cash Flows and other information presented below. Statement of Cash Flows Operating activities Net Income $345 Adjustments...
-
Are there traits that have been identified with successful leaders? Do research in popular business periodicals and identify traits that seem to be associated with managerial success. Are these...
-
Calculate Order level, Minimum level, Maximum level (Normal and Absolute), and EOQ from the following data: Cost per Order: Rs. 10 Carrying cost per unit per year: Rs. 600 Annual requirement :...
-
What do you perceive as the pros and cons of the Electoral College? Is it time for us as a nation to do away with this method of indirect election?
-
es Brett Collins is reviewing his company's investment in a cement plant. The company paid $10,500,000 five years ago to acquire the plant. Now top management is considering an opportunity to sell...
-
The following data are available for Sellco for the fiscal year ended on January 3 1 , 2 0 2 3 : Sales 1 , 6 0 0 units Beginning inventory 5 0 0 units @ $ 4 Purchases, in chronological order 6 0 0...
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
The isomerization of alkenes is catalysed by HCo(CO) 3 and Fig. 25.20 shows the relevant catalytic cycle. (a) HCo(CO) 4 is a catalyst precursor; explain what this means. (b) Give a fuller description...
-
State the types of isomerism that may be exhibited by the following complexes, and draw structures of the isomers: (a) [Co(en) 2 (ox)] + , (b) [Cr(ox) 2 (OH 2 ) 2 ] , (c) [PtCl 2 (PPh 3 ) 2 ], (d)...
-
Suggest likely products for the following reactions, with the stoichiometries stated: (a) B5 H + Br (b) B4 H10 + PF3 (c) 1-BrB5H8 (d) 2-MeB5Hg 298 K KH, 195 K ROH
-
Figure 1-2 shows that organizational culture and the design of an AIS influence one another. What does this imply about the degree to which an innovative system I developed by one company can be...
-
One interesting property of digital assets is that they can be reproduced and dis tributed via the Internet at very little cost. What are some of the implications of having a product with a marginal...
-
Figure 1-2 shows that developments in IT affect both an organizations strategy and the design of its AIS. How can a company determine whether it is spending too much, too little, or just enough on...

Study smarter with the SolutionInn App


