. A stream consisting of a mixture of N2 and water vapor, at 98.0 C and 5200.0...
Fantastic news! We've Found the answer you've been seeking!
Question:
 .
.
Transcribed Image Text:
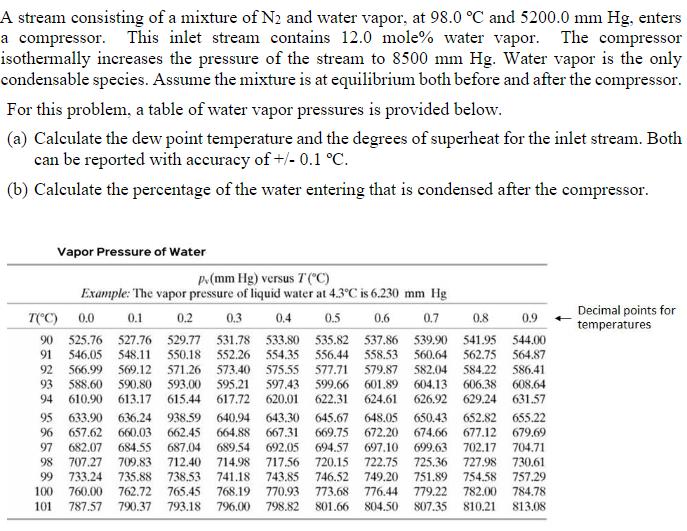
A stream consisting of a mixture of N2 and water vapor, at 98.0 °C and 5200.0 mm Hg, enters a compressor. This inlet stream contains 12.0 mole% water vapor. The compressor isothermally increases the pressure of the stream to 8500 mm Hg. Water vapor is the only condensable species. Assume the mixture is at equilibrium both before and after the compressor. For this problem, a table of water vapor pressures is provided below. (a) Calculate the dew point temperature and the degrees of superheat for the inlet stream. Both can be reported with accuracy of +/- 0.1 °C. (b) Calculate the percentage of the water entering that is condensed after the compressor. Vapor Pressure of Water p.(mm Hg) versus T("C) Example: The vapor pressure of liquid water at 4.3°C is 6.230 mm Hg 0.1 0.3 0.9 Decimal points for temperatures T(*C) 0.0 0.2 0.4 0.5 0.6 0.7 0.8 90 525.76 527.76 529.77 531.78 548,11 533.80 535.82 537.86 539.90 541.95 544.00 91 546.05 550.18 552.26 554.35 556.44 558.53 560.64 562.75 564.87 575.55 597.43 617.72 620.01 582.04 604.13 626.92 629.24 631.57 584.22 586.41 606.38 608.64 92 566.99 569.12 571.26 573.40 577.71 579.87 93 588.60 94 610,90 593.00 615.44 595.21 599.66 622.31 601.89 624,61 590.80 613.17 938.59 662.45 640.94 643.30 667.31 648.05 650.43 674.66 697.10 699.63 725.36 751.89 779.22 652.82 655.22 95 657.62 633.90 636.24 645.67 669.75 96 660.03 664.88 672.20 677.12 679.69 692.05 717.56 702.17 727.98 730.61 754.58 757.29 97 682.07 684.55 687.04 689.54 694.57 704.71 707.27 709,83 735.88 762.72 712.40 714,98 741.18 768,19 98 720,15 722.75 99 733.24 738.53 743.85 746.52 749.20 765.45 770,93 798.82 100 760.00 773.68 776.44 782.00 784.78 101 787.57 790.37 793.18 796.00 801.66 804.50 807.35 810.21 813.08 A stream consisting of a mixture of N2 and water vapor, at 98.0 °C and 5200.0 mm Hg, enters a compressor. This inlet stream contains 12.0 mole% water vapor. The compressor isothermally increases the pressure of the stream to 8500 mm Hg. Water vapor is the only condensable species. Assume the mixture is at equilibrium both before and after the compressor. For this problem, a table of water vapor pressures is provided below. (a) Calculate the dew point temperature and the degrees of superheat for the inlet stream. Both can be reported with accuracy of +/- 0.1 °C. (b) Calculate the percentage of the water entering that is condensed after the compressor. Vapor Pressure of Water p.(mm Hg) versus T("C) Example: The vapor pressure of liquid water at 4.3°C is 6.230 mm Hg 0.1 0.3 0.9 Decimal points for temperatures T(*C) 0.0 0.2 0.4 0.5 0.6 0.7 0.8 90 525.76 527.76 529.77 531.78 548,11 533.80 535.82 537.86 539.90 541.95 544.00 91 546.05 550.18 552.26 554.35 556.44 558.53 560.64 562.75 564.87 575.55 597.43 617.72 620.01 582.04 604.13 626.92 629.24 631.57 584.22 586.41 606.38 608.64 92 566.99 569.12 571.26 573.40 577.71 579.87 93 588.60 94 610,90 593.00 615.44 595.21 599.66 622.31 601.89 624,61 590.80 613.17 938.59 662.45 640.94 643.30 667.31 648.05 650.43 674.66 697.10 699.63 725.36 751.89 779.22 652.82 655.22 95 657.62 633.90 636.24 645.67 669.75 96 660.03 664.88 672.20 677.12 679.69 692.05 717.56 702.17 727.98 730.61 754.58 757.29 97 682.07 684.55 687.04 689.54 694.57 704.71 707.27 709,83 735.88 762.72 712.40 714,98 741.18 768,19 98 720,15 722.75 99 733.24 738.53 743.85 746.52 749.20 765.45 770,93 798.82 100 760.00 773.68 776.44 782.00 784.78 101 787.57 790.37 793.18 796.00 801.66 804.50 807.35 810.21 813.08
Expert Answer:
Answer rating: 100% (QA)
Degree of superheeat is the extra temperature in addition to saturation temperature Part 1 At inlet ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder,? Ronald W. Rousseau,? Lisa G. Bullard
Posted Date:
Students also viewed these chemical engineering questions
-
An equimolar mixture of carbon dioxide and water vapor at 1 atm and 60oC enter a dehumidifying section where the entire water vapor is condensed and removed from the mixture, and the carbon dioxide...
-
An equimolar mixture of carbon monoxide and water vapor at 1 atm and 298 K enters a reactor operating at steady state. The equilibrium mixture, composed of CO2, CO, H2O(g) and H2, leaves at 2000 K....
-
A stream consisting of 44.6 mole% benzene and 55.4% toluene is fed at a constant rate to a process unit that produces two product streams, one a vapor and the other a liquid. The vapor flow rate is...
-
Find a vector parametrization of the intersection of the surfaces x 2 + y 4 + 2z 3 = 6 and x = y 2 in R 3 .
-
Agents owe a fiduciary duty to their principals. What are the requirements of that duty?
-
Wisteria World is a theme park operated by an audit client of Salmon Partners. It is located on the Gold Coast and, due to its proximity to the ocean, the rides are prone to rust and corrosion....
-
The October 1, bank statement of Blakes Hamburger just arrived from First State Bank. To prepare the bank reconciliation, you gather the following additional data: a.The following checks are...
-
A 2.20.kg hoop 1.20 m in diameter is rolling to the right without slipping on a horizontal floor at a steady 3.00 rad/s. (a) How fast is its center moving? (b) What is the total kinetic energy of the...
-
Apple is planning to incorporate new software for the price of $ 5 0 0 0 0 0 today in order to produce a new line of the iPod. The new iPod will be ready for sale in 1 year. If the new software will...
-
Pascal's Identity states that 1 (0)-(3))+(7) k 1 k = k =o (3) k=0 1 Use Pascal's identity to prove that the sum of each row of Pascal's triangle is twice the sum of the previous row. = 2n Please do...
-
1. Explain diversity in a company. Explain how diversity benefits an organization? 2. What demographics lead to and are examples of diversity in an organization. Explain 3. the differences between...
-
Why is expediting required on construction jobs?
-
Describe the processes by which a state case can make its way to the U.S. Supreme Court (see the "Warren McCleskey's Odyssey" box).
-
Explain the effect on project indirect costs when a job is lengthened and when it is shortened.
-
Describe the basis of management-by-exception in construction.
-
What is the intended use for each of the following cost models? a. Project budget b. Project cost breakdown or schedule of values c. Progress estimates d. Change order estimates e. Project cash flow...
-
What do the authors say is sociologically significant about gaslighting? Why do they say psychological study of gaslighting is insufficient?
-
Solve each equation. x 3 - 6x 2 = -8x
-
Nitrogen is bubbled through a liquid mixture that initially contains Equimolar amounts of benzene and toluene. The system pressure is 3 atm and the temperature is 80C. The nitrogen flow rate is 10.0...
-
The little-known rare earth element nauseum (atomic weight = 172) has the interesting property of being completely insoluble in everything but 12-year-old bourbon. This curious fact was discovered in...
-
As part of a design calculation, you must evaluate an enthalpy change for an obscure organic vapor that is to be cooled from 1800C to 150C in a heat exchanger. You search through all the standard...
-
Describe the ways in which the scope of many audits in the early 1900 s was significantly different from what it is today.
-
What is the difference between statutory law and common law?
-
Name and define four standards of proof.

Study smarter with the SolutionInn App


