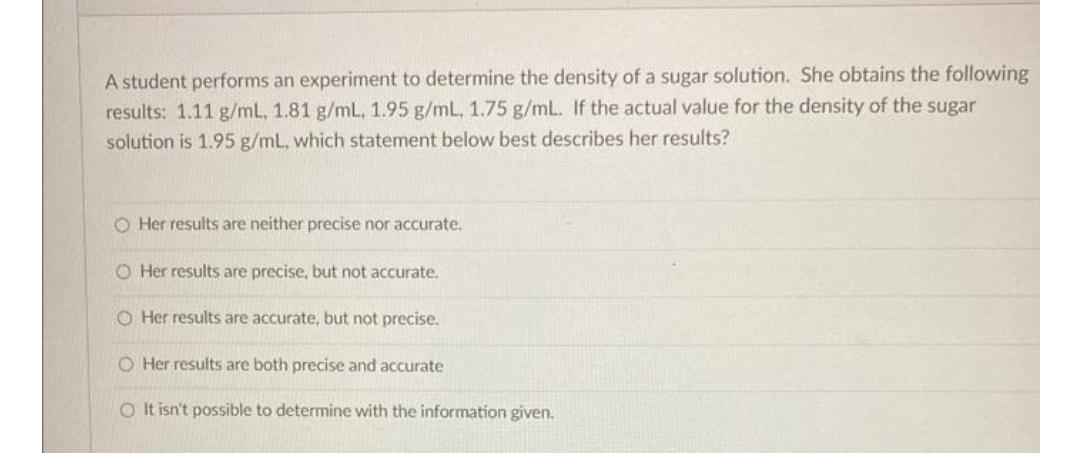
A student performs an experiment to determine the density of a sugar solution. She obtains the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A student performs an experiment to determine the density of a sugar solution. She obtains the following results: 1.11 g/mL, 1.81 g/mL, 1.95 g/mL, 1.75 g/mL. If the actual value for the density of the sugar solution is 1.95 g/mL, which statement below best describes her results? O Her results are neither precise nor accurate. O Her results are precise, but not accurate. O Her results are accurate, but not precise. O Her results are both precise and accurate O It isn't possible to determine with the information given. A student performs an experiment to determine the density of a sugar solution. She obtains the following results: 1.11 g/mL, 1.81 g/mL, 1.95 g/mL, 1.75 g/mL. If the actual value for the density of the sugar solution is 1.95 g/mL, which statement below best describes her results? O Her results are neither precise nor accurate. O Her results are precise, but not accurate. O Her results are accurate, but not precise. O Her results are both precise and accurate O It isn't possible to determine with the information given.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
A plant physiologist conducted an experiment to determine whether mechanical stress can retard the growth of soybean plants. Young plants were randomly allocated to two groups of 13 plants each....
-
The radius of a solid sphere is measured to be (6.50 0.20) cm, and its mass is measured to be (1.85 0.02) kg. Determine the density of the sphere in kilograms per cubic meter and the uncertainty in...
-
An experiment to determine the convection coefficient associated with airflow over the surface of a thick stainless steel casting involves insertion of thermocouples in the casting at distances of 10...
-
Find f. f(x) = f'(x) = 12x + X x > 0, f(1) = -4
-
The perpetuity with growth formula invokes several assumptions. Which one seems least plausible?
-
Question Drawing on the topic of measuring and reporting CSR, assume you are a CSR consultant and one of your small-firm clients wants to begin producing a CSR report in the hopes that their firm...
-
Presented below are selected financial data from the The Coca-Cola Company 2015 annual report. Using the ratio definitions from Exhibit 4.6, calculate the following ratios: receivable turnover,...
-
As of the end of June, the job cost sheets at Racing Wheels, Inc., show the following total costs accumulated on three custom jobs. Job 102 was started in production in May and the following costs...
-
A clothing company has a glut of jeans in its warehouse. They have let the market decide the best price to reduce the inventory. Which approach can be used to allow this to happen? a. Bargains b....
-
Is it alright for non-native speaker of English to use a sound from his/her language as a substitute for an English sound that is nonexistent in his/her language? Why or why not? Explain your answer...
-
I. Fill out the blanks below for 5 points each. (Notice that reading Textbook Pages 212 213 helps you to complete the fill-in questions below.) ) is the interest rate that would exist on a riskless...
-
Which parking deck topping system should be considered during the structural design of a building?
-
Which technique involves attackers mimicking normal user behavior to remain undetected by an IDS ?
-
You have been tasked by your manager to teach your workgroup about innovation and organizational change as part of a pilot business development initiative. If the pilot with your group is successful,...
-
Which method can help in detecting encrypted malicious traffic?
-
At this time your team will need to make a communication plan such as a weekly schedule. For example, the team may choose to have one initial live chat during this week to set up a communication plan...
-
1 Critically evaluate the importance of the media in influencing partners, opinion formers, stakeholders, customers and the public at large. 2 Critically assess domestic, national and international...
-
Feller Company purchased a site for a limestone quarry for $100,000 on January 2, 2019. It estimate that the quarry will yield 400,000 tons of limestone. It estimates that its retirement obligation...
-
The structures of another class of high-temperature ceramic superconductors are shown below. a. Determine the formula of each of these four superconductors. b. One of the structural features that...
-
What is the valence electron configuration for Group 4A elements? Group 4A contains two of the most important elements on earth. What are they, and why are they so important? How does metallic...
-
Write formulas for the following compounds. a. Sodium oxide b. Sodium peroxide c. Potassium cyanide d. Copper(II) nitrate e. Silicon tetrachloride f. Lead(II) oxide g. Lead(IV) oxide h. Copper(I)...
-
Schedule of Cost of Goods Sold Sydney Company reported the following amounts for October: Use Excel to prepare a schedule of cost of goods sold for October. Beginning raw materials inventory....
-
For each of the following unrelated companies, compute the cost of goods manufactured and the cost of goods sold: A B C Selling expenses Factory insurance. Ending finished goods inventory....
-
The following transactions occurred during January 2019 for Richards Manufacturing Company: Jan. 5 Acquired $4,500 of materials on account that will be used to produce product for resale. 11...

Study smarter with the SolutionInn App



