A student titrated a 10.00 mL sample of vinegar with 0.187 M NaOH. The student analyzed...
Fantastic news! We've Found the answer you've been seeking!
Question:
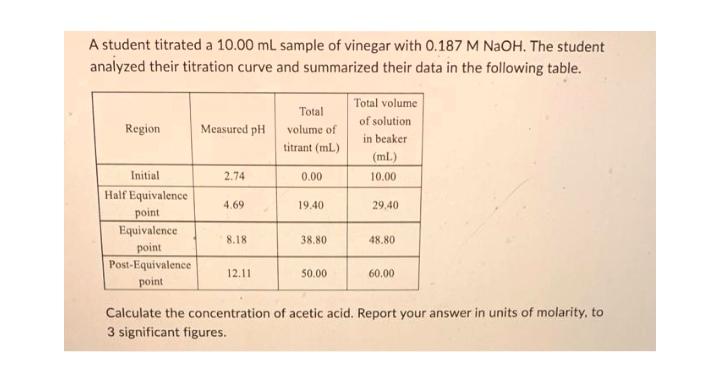
Transcribed Image Text:
A student titrated a 10.00 mL sample of vinegar with 0.187 M NaOH. The student analyzed their titration curve and summarized their data in the following table. Region Initial Half Equivalence point Equivalence point Post-Equivalence point Measured pH 2.74 4.69 8.18 12.11 Total volume of titrant (ml) 0.00 19.40 38.80 50.00 Total volume of solution in beaker (ml) 10.00 29.40 48.80 60,00 Calculate the concentration of acetic acid. Report your answer in units of molarity, to 3 significant figures. A student titrated a 10.00 mL sample of vinegar with 0.187 M NaOH. The student analyzed their titration curve and summarized their data in the following table. Region Initial Half Equivalence point Equivalence point Post-Equivalence point Measured pH 2.74 4.69 8.18 12.11 Total volume of titrant (ml) 0.00 19.40 38.80 50.00 Total volume of solution in beaker (ml) 10.00 29.40 48.80 60,00 Calculate the concentration of acetic acid. Report your answer in units of molarity, to 3 significant figures.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The labor demand and supply data in the following table relate to a single occupation. Use them to answer the questions that follow. Base your answers on the taste-for-discrimination model. a. Plot...
-
Fill in the missing data in the following table for amonopolist. Quantity Total Revenue Marginal Revenue Demand Elastic or Inelastic? Price $11 10 6 10 2
-
Plot the income and consumption data in the following table on a graph, with income on the X-axis. Does the data indicate a positive or negative relationship between income and consumption? Average...
-
Do laws provide a complete guide to ethical behavior? Can an activity be legal but not ethical?
-
Any successful project plan must contain nine key elements. List these items and briefly describe the composition of each.
-
Which of the six cases shown in the figure has the largest absolute value of the scalar product of the vectors A and B? (a) (b) (c) (d) (f) (e)
-
Prove Green's identity \(\int_{a}^{b}(u \mathcal{L} v-v \mathcal{L} u) d x=\left.\left[p\left(u v^{\prime}-v u^{\prime} ight) ight] ight|_{a} ^{b}\) for the general Sturm-Liouville operator...
-
Yang Corporation wholesales repair products to equipment manufacturers. On May 1, 2014, Yang Corporation issued $20,000,000 of 10-year, 9% bonds at a market (effective) interest rate of 7%, receiving...
-
Gifts Galore Inc. borrowed $1.95 million from National City Bank. The loan was made at a simple annual interest rate of 15% a year for 3 months. A 20% compensating balance requirement raised the...
-
You send daily emails about a variety of topics for a shoe store. Your open rates are low, but sale announcements perform best. Which two marketing actions would most likely increase open rates?...
-
Why is Specifying System Constraints and Intervention Inputs important in healthcare? Discuss.
-
Agro International has a foreign subsidiary, which requires $200K cash every month. It uses a wire service for the cash transfer. The cost of wire transfer is $190. In order to send cash to its...
-
discuss the role of computational fluid dynamics (CFD) in process modeling, including its applications in simulating complex fluid behavior, heat and mass transfer phenomena, and reactor design...
-
You have successfully passed the pre-employment assessment and have been invited to attend an interview. You have been informed that during the interview you will be asked to discuss how you would...
-
You make two savings bank deposits, $4,000 today, and $2,400 in 2 years time. Your bank pays an interest rate of 2%. What will your balance be (from these two deposits only) after 5 years (3 years...
-
Problem 3. A University of Massachusetts study found that married women who work outside the home do about one hour less of housework per week for every $7500 they earn outside the home. Assume that...
-
1. Marian Hobby Store pays a dividend of $0.35 per share per quarter. The next dividend will be paid to shareholders of record on Friday, December 20, 2013. The company has 3 million shares...
-
Why do markets typically lead to an efficient outcome for buyers and sellers?
-
(a) Explain the difference between solubility and solubilityproduct constant. (b) Write the expression for the solubility-product constant for each of the following ionic compounds:MnCO3, Hg(OH)2,...
-
Methanol (CH3OH) can be made by the reaction of CO with H2: (a) Use thermochemical data in Appendix C to calculate (Ho for this reaction. (b) To maximize the equilibrium yield of methanol, would you...
-
The standard enthalpies of formation of ClO and ClO2 are 101 and 102 kJ/mol, respectively. Using these data and the thermodynamic data in Appendix C, calculate the overall enthalpy change for each...
-
A prestigious investment bank designed a new security that pays a quarterly dividend of \($10\) permanently. What is the price of the security if the stated annual interest rate is 12 percent,...
-
Calculate the present value of \($5,000\) in 12 years at a stated annual interest rate of 10 percent, compounded quarterly.
-
Bank America offers a stated annual interest rate of 4.1 percent, compounded quarterly, while Bank USA offers a stated annual interest rate of 4.05 percent, compounded monthly. In which bank should...

Study smarter with the SolutionInn App



