a) The fluorene monomer A and 1,4-dibromo benzene B (Figure Q2) can be copolymerised. What is...
Fantastic news! We've Found the answer you've been seeking!
Question:
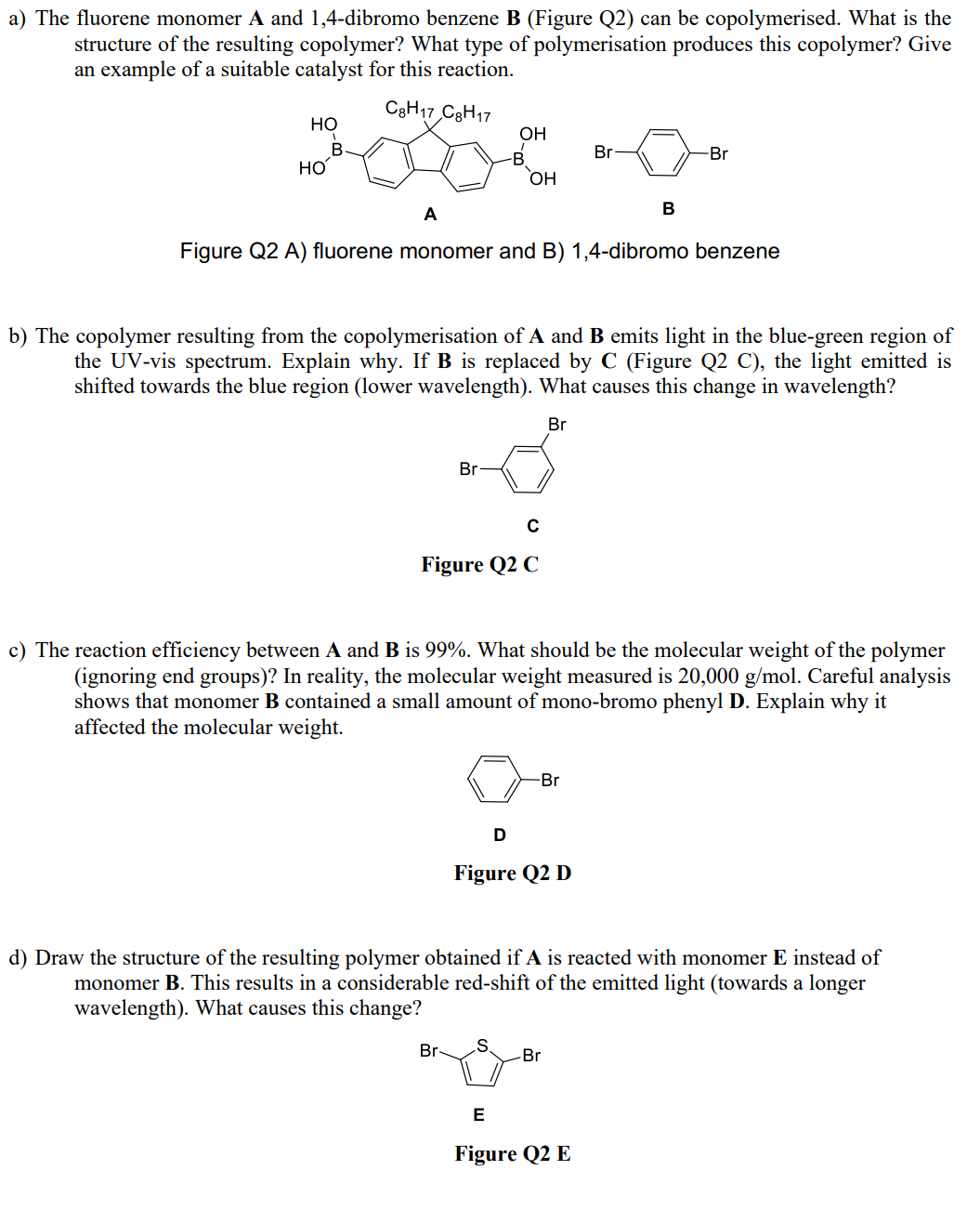
Transcribed Image Text:
a) The fluorene monomer A and 1,4-dibromo benzene B (Figure Q2) can be copolymerised. What is the structure of the resulting copolymer? What type of polymerisation produces this copolymer? Give an example of a suitable catalyst for this reaction. C8H17 C8H17 HO B 20 HO Br OH -B Br- OH A Figure Q2 A) fluorene monomer and B) 1,4-dibromo benzene C Figure Q2 C b) The copolymer resulting from the copolymerisation of A and B emits light in the blue-green region of the UV-vis spectrum. Explain why. If B is replaced by C (Figure Q2 C), the light emitted is shifted towards the blue region (lower wavelength). What causes this change in wavelength? Br D S c) The reaction efficiency between A and B is 99%. What should be the molecular weight of the polymer (ignoring end groups)? In reality, the molecular weight measured is 20,000 g/mol. Careful analysis shows that monomer B contained a small amount of mono-bromo phenyl D. Explain why it affected the molecular weight. -Br Figure Q2 D Br B -Br d) Draw the structure of the resulting polymer obtained if A is reacted with monomer E instead of monomer B. This results in a considerable red-shift of the emitted light (towards a longer wavelength). What causes this change? Br E Figure Q2 E a) The fluorene monomer A and 1,4-dibromo benzene B (Figure Q2) can be copolymerised. What is the structure of the resulting copolymer? What type of polymerisation produces this copolymer? Give an example of a suitable catalyst for this reaction. C8H17 C8H17 HO B 20 HO Br OH -B Br- OH A Figure Q2 A) fluorene monomer and B) 1,4-dibromo benzene C Figure Q2 C b) The copolymer resulting from the copolymerisation of A and B emits light in the blue-green region of the UV-vis spectrum. Explain why. If B is replaced by C (Figure Q2 C), the light emitted is shifted towards the blue region (lower wavelength). What causes this change in wavelength? Br D S c) The reaction efficiency between A and B is 99%. What should be the molecular weight of the polymer (ignoring end groups)? In reality, the molecular weight measured is 20,000 g/mol. Careful analysis shows that monomer B contained a small amount of mono-bromo phenyl D. Explain why it affected the molecular weight. -Br Figure Q2 D Br B -Br d) Draw the structure of the resulting polymer obtained if A is reacted with monomer E instead of monomer B. This results in a considerable red-shift of the emitted light (towards a longer wavelength). What causes this change? Br E Figure Q2 E
Expert Answer:
Answer rating: 100% (QA)
The image contains a chemistry exam question about the copolymerization of monomers the emission of light by copolymers the molecular weight of polyme... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemical engineering questions
-
The company has 200 000 non-redeemable preference shares in issue with a par value R100. Preference dividends are payable annually in arrears. The non-redeemable preference shares are currently...
-
The following functional-group interchange is a useful synthesis of aldehydes. (a) What reagents were used in this chapter for this transformation? Give an example to illustrate this method. (b) This...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
At December 31, 2010, Cafu Co. reported the following information on its statement on financial position. Accounts receivable...............R$960,000 Less: Allowance for doubtful accounts............
-
Bogoda Industries makes all sales on account. Ian Holt, accountant for the company, receives and opens incoming mail. Company procedure requires Holt to separate customer checks from the remittance...
-
The brands debate Under IAS 22, the depletion of equity reserves caused by the accounting treatment for purchased goodwill resulted in some companies capitalising brands on their statements of...
-
Question: Antoine makes a check out to cash and delivers it to Barley. He writes on the back, "Pay to the order of Charlotte." She signs her name. Is this check bearer paper or order paper? Has it...
-
For years The Glass Slipper restaurant has operated in a resort community near a popular ski area of New Mexico. The restaurant is busiest during the first 3 months of the year, when the ski slopes...
-
A non-callable 4-year UK government bond pays semi-annual coupons. The face value is 1000, the annual coupon rate is 6% and the yield to maturity is 4% per year. (a) Show clearly how the current...
-
You are working on a free-form Packet Tracer challenge activity as seen in Figure 1, you have been given the London Railways network.' The purpose of this EMA question is to build upon each of the...
-
draw a use case diagram and sequence diagram for a following scenario: online food ordering and delivery system: Firstly, When the users open the app, they will first see the login interface. If they...
-
Create a class that describes a circle and have the following: 1. Variable Length 2. Variable Wigth 3. Members: a. SetRadius b. GetRadius c. SetCenterX (you set the circle center coordinate xo). d....
-
Lengthy Issue Resolution Delays and Inadequate Notices Burden Taxpayers Who Are Victims of Identity Theft or Whose Returns the IRS Has Flagged for Possible Identity Theft? WHY THIS IS A SERIOUS...
-
Within the intricate tapestry of advanced organizational ecosystems, characterized by their complex network of interrelated stakeholders and intricate decision-making processes, what systemic factors...
-
Employing elementary transformations, find the inverse of the matrix 0 1 2 (i) 1 2 3 3 1 1 1 2 3 1 1 3 3 2 (ii) 2433 1 1 1 1
-
sunland zone corporation expeirenced variances: materials price $ 3 1 0 U , materials quanity $ 1 , 5 0 0 F , labor price $ 7 0 0 F , and total overhead $ 1 , 0 6 0 U . Sales revenue was $ 8 1 , 0 0...
-
How has your academic , athletic, employment, religious, and/or community service experiences molded you into the individual you are today? What adversity have you overcome and how has that...
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
a. Wilson filed his individual tax return on the original due date, but failed to pay $700 in taxes that were due with the return. If Wilson pays the taxes exactly 2 months late, calculate the amount...
-
Jenny earns $34,500 in 2012. Calculate the FICA tax that must be paid by: Jenny: ..............................Soc,Sec. ..................$______________...
-
John Williams (age 42) is a single taxpayer, and he lives at 1324 Forest Dr., Reno, NV 89501. His Social Security number is 555-94-9358. John's earnings and withholdings as the manager of a local...
-
1. The German-American Vocational League was formed in New York during World War II to serve as a propaganda agency for the German Reich. Under U.S. law all foreign agents were required to register....
-
3. David and Fiona Rookard purchased tickets for a trip through Mexico from a Mexicoach office in San Diego. Mexicoach told them that the trip would be safe. It did not tell them, however, that their...
-
2. ETHICS Radio TV Reports (RTV) was in the business of recording, transcribing, and monitoring radio and video programming for its clients. The Department of Defense (DOD) in Washington, D.C., was...

Study smarter with the SolutionInn App


