A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as...
Fantastic news! We've Found the answer you've been seeking!
Question:
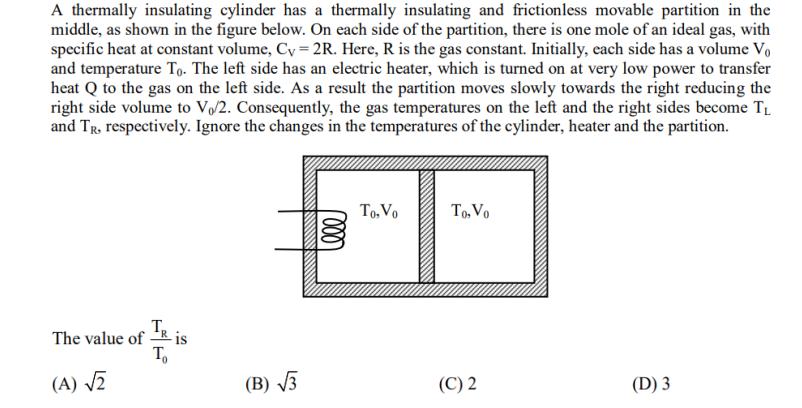
Transcribed Image Text:
A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, Cy=2R. Here, R is the gas constant. Initially, each side has a volume Vo and temperature To. The left side has an electric heater, which is turned on at very low power to transfer heat Q to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to V/2. Consequently, the gas temperatures on the left and the right sides become TL and Tr. respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition. TR To The value of - (A) √2 -is (B)√√√3 Lele To, Vo To, Vo (C) 2 (D) 3 A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, Cy=2R. Here, R is the gas constant. Initially, each side has a volume Vo and temperature To. The left side has an electric heater, which is turned on at very low power to transfer heat Q to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to V/2. Consequently, the gas temperatures on the left and the right sides become TL and Tr. respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition. TR To The value of - (A) √2 -is (B)√√√3 Lele To, Vo To, Vo (C) 2 (D) 3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
One mole of an ideal gas with heat capacity Cv goes through a process in which its entropy S depends on T as S = a/T, where a is a constant. The gas temperature varies from T1 to T2. Find: (a) The...
-
One mole of an ideal gas with the adiabatic exponent y goes through a polytropic process as a result of which the absolute temperature of the gas increases T-fold. The polytropic constant equals n....
-
One mole of an ideal gas with CP = (7/2) R and CV = (5/2) R expands from P1 = 8 bar and T1 = 600 K to P2 = 1 bar by each of the following paths: (a) Constant volume: (b) Constant temperature; (c)...
-
PWX Inc. has the following information for its years ended June 30: Required: Calculate the accounts receivable turnover and average collection period for 20X3 and 20X2. Comment on the trend. What...
-
The pretax operating incomes of Pop Corporation and Son Corporation, its 70 percent-owned subsidiary, for 2011 are as follows (in thousands): ADDITIONAL INFORMATION1. Pop received $280,000 dividends...
-
Discuss risk-management strategies that firms use to hedge commodity price risk.
-
Calculate \(43+18 \times 15\).
-
Condensed financial data of Sinjh Inc. follow. Additional information: 1. New plant assets costing $80,000 were purchased for cash during the year. 2. Old plant assets having an original cost of...
-
All employees are paid on a weekly basis and according to their W-4 are all married and do not request any additional withholding (Use the tables that start on page 13 of the Pub 15-T that is posted...
-
1. What is the cost of debt for Sunrise Bakery? 2. What is their cost of equity? 3. What is the WACC? 4. Which cost of capital should be used to evaluate the feasibility of the oven purchase? 5....
-
The Platte Corporation issues a 5-year note payable on January 1, Year 1 for $5,000. The interest rate is 5% and the annual payment of $1,156, due each December 31, includes both interest and...
-
Monty Inc. had service revenue of $742000, operating expenses of $396000, and interest expense of $28800 for the year ended December 31, 2024. The company has a 30% income tax rate. Assuming Monty...
-
The owner of Britten's Egg Farm wants to estimate the mean number of eggs laid per chicken. A sample of 16 chickens shows they laid an average of 18 eggs per month with a standard deviation of 5 eggs...
-
a. Benefits for employees are forms of payment other than a salary or hourly wage. The following are just a few examples of specific employee benefits: paid time off, health insurance, tuition...
-
A producer of door frame chin-up bars has determined that the revenue from the production and sale of x units is given by R(x)=50x0.04x2 dollars, where 0x1000 . For what interval of production is the...
-
Valber Company is considering eliminating its Phone division. The company allocates fixed costs based on sales. If the Phone division is dropped, all of its variable costs are avoidable, and $158,000...
-
The bulbs that LV stocks in their warehouse contain toxic substances if they are broken. Provide a brief overview to management on the roles and responsibilities that LV has specific to Dangerous...
-
The ultimate goal of Google, Bing, and other consumer search engines is to provide users with search listings that contain useful information on the topic of their search. What recommendations would...
-
Light that has a wavelength of 668 nm passes through a slit 6.73 10-6 m wide and falls on a screen that is 1.85 m away. What is the distance on the screen from the center of the central bright...
-
A dish of lasagna is being heated in a microwave oven. The effective area of the lasagna that is exposed to the microwaves is 2.2 10-2 m2. The mass of the lasagna is 0.35 kg, and its specific heat...
-
The preparation of homeopathic remedies involves the repeated dilution of solutions containing an active ingredient such as arsenic trioxide (As2O3). Suppose one begins with 18.0 g of arsenic...
-
Explain how a shell company could be used to engage in a pass-through billing scheme.
-
What is meant by the cockroach theory of fraud?
-
The FBI estimates that fraud in the United States is annually what amount? a. \(\$ 652\) billion b. \(\$ 1\) trillion c. \(\$ 300\) billion d. \(\$ 400\) billion e. Some other amount

Study smarter with the SolutionInn App



