(a) What is the % composition of the vapor in equilibrium with a boiling liquid that has...
Fantastic news! We've Found the answer you've been seeking!
Question:
(a) What is the % composition of the vapor in equilibrium with a boiling liquid that has a composition of 30% benzene and 70% toluene.
b). If a sample of vapor is found to have a composition of equal parts benzene and toluene, what is the composition of the boiling liquid that produced this vapor?

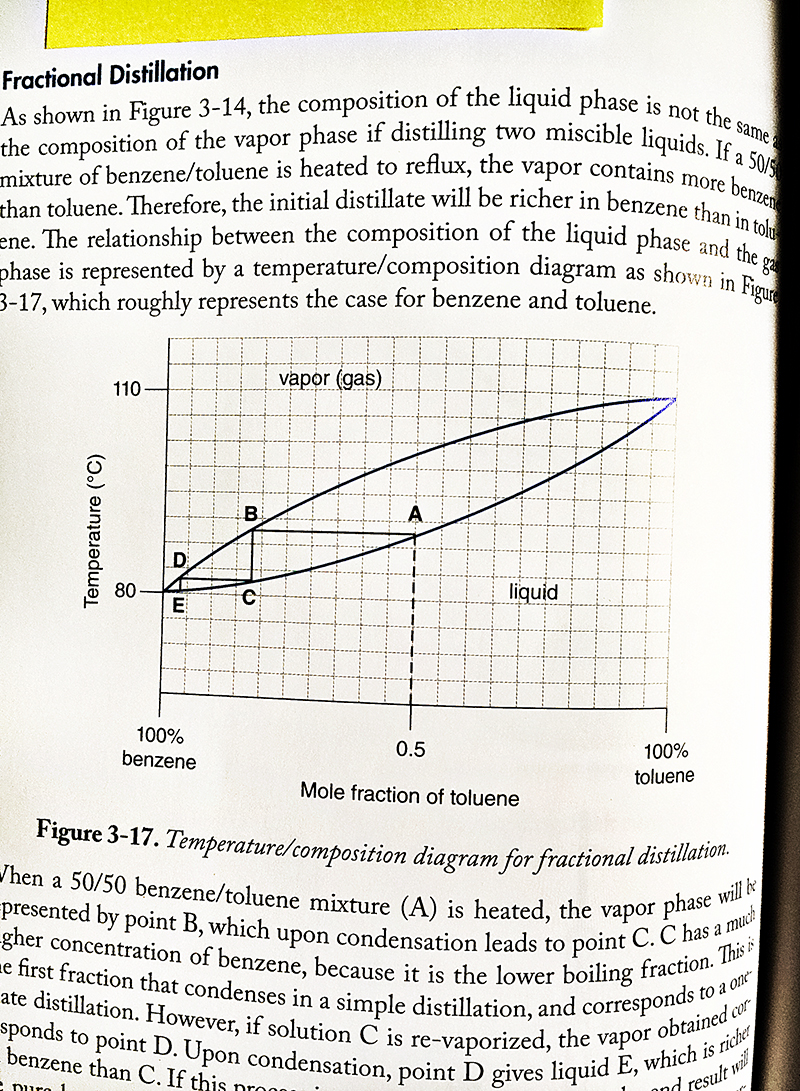
Transcribed Image Text:
Post-Lab Questions 1. Refer to MtC Figure 3-17 to answer the following questions. Note that each vertical line in the graph grid equals 5%, and the scale runs both ways for benzene and toluene: 100% benzene → 0% benzene 0% toluene → 100% toluene a. What is the % composition of the vapor in equilibrium with a boiling liquid that has a composition of 30% benzene and 70% toluene? b. If a sample of vapor is found to have a composition of equal parts benzene and toluene, what is the composition of the boiling liquid that produced this vapor? Fractional Distillation As shown in Figure 3-14, the composition of the liquid phase is not the same a the composition of the vapor phase if distilling two miscible liquids. If a 50/5 than toluene. Therefore, the initial distillate will be richer in benzene than in tolu mixture of benzene/toluene is heated to reflux, the vapor contains more benzene ene. The relationship between the composition of the liquid phase and the ga phase is represented by a temperature/composition diagram as shown in Figure 3-17, which roughly represents the case for benzene and toluene. Temperature (°C) 110 D E 100% benzene B vapor (gas) A 0.5 liquid Mole fraction of toluene 100% toluene Figure 3-17. Temperature/composition diagram for fractional distillation. When a 50/50 benzene/toluene mixture (A) is heated, the vapor phase will b gher concentration of benzene, because it is the lower boiling fraction. This is presented by point B, which upon condensation leads to point C. C has a much e first fraction that condenses in a simple distillation, and corresponds t ate distillation. However, if solution C is re-vaporized, the vapor obtained cor sponds to point D. Upon condensation, point D gives liquid E, which is richer to a one benzene than C. If this pron e Duro 1 and result will Post-Lab Questions 1. Refer to MtC Figure 3-17 to answer the following questions. Note that each vertical line in the graph grid equals 5%, and the scale runs both ways for benzene and toluene: 100% benzene → 0% benzene 0% toluene → 100% toluene a. What is the % composition of the vapor in equilibrium with a boiling liquid that has a composition of 30% benzene and 70% toluene? b. If a sample of vapor is found to have a composition of equal parts benzene and toluene, what is the composition of the boiling liquid that produced this vapor? Fractional Distillation As shown in Figure 3-14, the composition of the liquid phase is not the same a the composition of the vapor phase if distilling two miscible liquids. If a 50/5 than toluene. Therefore, the initial distillate will be richer in benzene than in tolu mixture of benzene/toluene is heated to reflux, the vapor contains more benzene ene. The relationship between the composition of the liquid phase and the ga phase is represented by a temperature/composition diagram as shown in Figure 3-17, which roughly represents the case for benzene and toluene. Temperature (°C) 110 D E 100% benzene B vapor (gas) A 0.5 liquid Mole fraction of toluene 100% toluene Figure 3-17. Temperature/composition diagram for fractional distillation. When a 50/50 benzene/toluene mixture (A) is heated, the vapor phase will b gher concentration of benzene, because it is the lower boiling fraction. This is presented by point B, which upon condensation leads to point C. C has a much e first fraction that condenses in a simple distillation, and corresponds t ate distillation. However, if solution C is re-vaporized, the vapor obtained cor sponds to point D. Upon condensation, point D gives liquid E, which is richer to a one benzene than C. If this pron e Duro 1 and result will
Expert Answer:
Answer rating: 100% (QA)
Solution a 65 Benzene and 35 Toluene Go up wards from the point of 30 Benz... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A freshly isolated sample of 90Y was found to have an activity of 9.8 105 disintegrations per minute at 1:00 p.m. on December 3, 2003. At 2:15 p.m. on December 17, 2003, its activity was...
-
What is the composition of the FASB's membership?
-
Is the composition of state legislatures in the House of Representatives related to the specific state? Use = 0.05. Perform the following steps. a. State the hypotheses and identify the claim. b....
-
solve the following instance of the 0/1 kropsack problem using dynamic programming approach. Assume that the knapsack capacity is 9. Item 1 3 4 weight 6 2 5 7 Benef $8 $4 $6 $10
-
Is it possible for an adiabatic liquid-vapor separator to separate wet steam at 100 psia and 90 percent quality, so that the pressure of the outlet streams is greater than 100 psia?
-
Find an equation of the line satisfying the conditions. If possible, for each line write a formula for a linear function whose graph is the line. Horizontal, passing through (-5, 6)
-
Bristol-Myers Squibb (BMS) Company reports that it maintains a \($2\) billion, five-year revolving line of credit with a syndicate of lenders. The company disclosed in its annual report that: The...
-
Mark Albin, Roland Peters and Sam Ramsey invested $ 164,000, $ 98,400 and $ 65,600, respectively, in a partnership. During its first calendar year, the firm earned $ 270,000. Required Prepare the...
-
Blossom Company's standard labor cost of producing one unit of Product DD is 3.00 hours at the rate of $10.00 per hour. During August, 40,000 hours of labor are incurred at a cost of $10.10 per hour...
-
Janet just graduated from college, has a job she's scheduled to begin in 3 months and has decided to treat herself to 6 weeks of travel across South America before she buckles down and starts working...
-
A new product line for BMW could be a line of electric vehicles. Research and answer the following questions for your marketing plan. Questions: Who are your competitors? What features distinguish...
-
Why is it hard to reuse traditional model in the world of changing requirements.
-
List some of the related design patterns used in formulating the trust stable pattern.
-
List three test cases to test the class members of the trust pattern.
-
List some of the testing patterns that can be applied for testing the trust stable analysis pattern.
-
List some of the lessons learnt from the use of the stable analysis pattern for trust.
-
Characteristic FSA HRA HSA Unspent account balances can be carried over from year to year Account balances are funded only by the employer If you leave your employer, the funds remain with the...
-
Velshi Printers has contracts to complete weekly supplements required by fortysix customers. For the year 2018, manufacturing overhead cost estimates total $600,000 for an annual production capacity...
-
Calculate the molality of each of the following solutions: (a) 14.3 g of sucrose (C12H22O11) in 676 g of water, (b) 7.20 moles of ethylene glycol (C2H6O2) in 3546 g of water.
-
A 250-mL glass bottle was filled with 242 mL of water at 20C and tightly capped. It was then left outdoors overnight, where the average temperature was - 5C. Predict what would happen. The density of...
-
A solution of 1.00 g of anhydrous aluminum chloride, AlCl3, in 50.0 g of water freezes at 21.11C. Does the molar mass determined from this freezing point agree with that calculated from the formula?...
-
Hardy Instruments, Inc., is a manufacturer of various measuring and controlling instruments. Financial statement data for the firm are as follows: Required a. Using the given data, calculate ratios 1...
-
You are analyzing the performance of Jackson Corporation, a manufacturer of personal care products, for the most recent year. The following data are taken from the firm's latest annual report: In...
-
Past quarterly data are presented below for Company company's fiscal year ends on the Saturday nearest to January 31. The other company is the Gillette Company prior to its acquisition by Procter \&...

Study smarter with the SolutionInn App


